Clear Sky Science · en
Change in the chondroitin/dermatan structure in distal lung tissue from COPD patients
Why this matters for people with COPD
Chronic obstructive pulmonary disease (COPD) is usually described as a problem of blocked airways and damaged air sacs. But behind those familiar symptoms lies a hidden scaffolding of sugars and proteins that helps hold lung tissue together and control inflammation. This study looks closely at that scaffolding in human lungs and shows that its sugar chains are chemically reworked as COPD progresses. Understanding these subtle changes could open up new ways to slow down damage in the lungs rather than just treating symptoms.
The lung’s hidden support network
Our lungs are built around an intricate support network called the extracellular matrix. It is made of proteins, like collagen, and special molecules called proteoglycans that carry long sugar chains known as glycosaminoglycans (GAGs). These GAGs, including chondroitin sulfate, dermatan sulfate, and heparan sulfate, help determine how stiff or elastic the tissue is and how it interacts with growth factors and enzymes that break down matrix. In COPD, previous work has shown that the overall mix of matrix proteins changes. Here, the researchers asked a more detailed question: even if the main protein backbones stay similar, do the attached sugar chains change their composition and chemical decoration as disease worsens?
Comparing healthy, smoker, and COPD lungs
The team analyzed lung tissue taken from non-smokers, smokers without COPD, and patients with moderate and severe COPD. Using sensitive chemical methods, they measured the total amount and fine structure of GAGs in the distal parts of the lungs, where gas exchange happens. They also re-examined existing protein and gene activity datasets from similar patients. While the core proteoglycan proteins themselves did not show large differences in the small protein dataset, the total amount of GAGs was higher in COPD, especially in more advanced disease. The most abundant GAGs were chondroitin and dermatan sulfates, and these were already elevated in patients with moderate COPD compared with healthy controls.
Chemical tweaks to sugar chains in diseased lungs
Looking beyond total levels, the scientists zoomed in on how individual sugar building blocks within GAG chains were modified. In severe COPD, heparan sulfate showed more sulfate groups added at specific positions, a change that can alter how it binds signaling molecules. Even more striking were the changes in chondroitin and dermatan sulfate. Sugars carrying a particular type of sulfate group at the 4-position became more common across COPD stages, and a portion of these sugars were converted into a slightly different form (iduronic acid) that is known to influence how the matrix interacts with enzymes and growth factors. These structural fingerprints matched a rise in the activity of several genes that build and modify these chains, most notably CHST11, which helps place the 4-sulfate group.
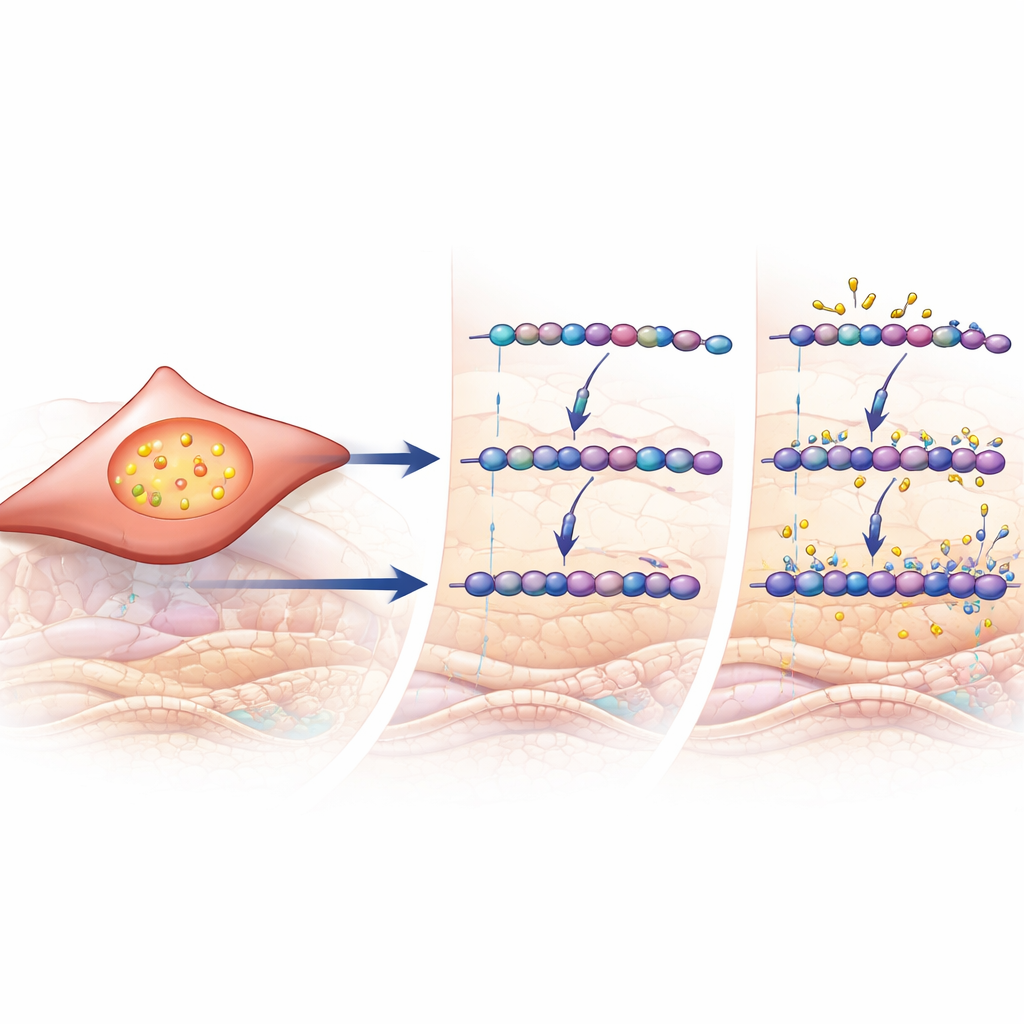
A signaling switch that drives remodeling
To find out what might be switching on these sugar-modifying enzymes, the researchers turned to large-scale gene data and cell experiments. In patient lung samples, genes turned up in COPD were enriched for those known to respond to the signaling molecule TGF-β, a key driver of scarring and remodeling in many tissues. The expression of CHST11 and several related enzymes tracked with a computed TGF-β activity score. In cultured human lung fibroblast cells, direct exposure to TGF-β increased production of chondroitin/dermatan sulfate, boosted the specific 4-sulfated sugars seen in patient lungs, and raised CHST11 levels. Together, these findings connect a well-known remodeling signal to the precise chemical reshaping of GAG chains observed in COPD lungs.
What this means for future treatments
For people living with COPD, these results suggest that the lung’s scaffolding is not just breaking down; it is being chemically rewired in a disease-stage-specific way. The added sulfate groups and altered sugar forms in chondroitin/dermatan and heparan sulfate chains are likely to change how the matrix binds enzymes, growth factors, and inflammatory molecules, potentially feeding into ongoing tissue damage and scarring. Because these changes are linked to specific enzymes such as CHST11 and to TGF-β signaling, they point to new, more precise drug targets: instead of broadly blocking inflammation, future therapies might aim to gently rebalance how lung sugar chains are built and modified, with the goal of stabilizing the tissue and slowing COPD progression.
Citation: Alsafadi, H.N., Nybom, A., Wagner, D. et al. Change in the chondroitin/dermatan structure in distal lung tissue from COPD patients. Sci Rep 16, 9721 (2026). https://doi.org/10.1038/s41598-026-44120-4
Keywords: COPD, lung matrix, glycosaminoglycans, extracellular remodeling, TGF-beta signaling