Clear Sky Science · en
Access to β-acetoxyselenides, perhydroindolones and anticancer piperidinones via blue LED-driven selenylation of unsaturated carboxamides
Light Powered Chemistry for Future Cancer Drugs
Chemists and biologists have joined forces to use simple blue LED light to build new selenium based molecules that show promise as future anticancer drug candidates. By fine tuning this light driven chemistry, they can steer the same starting materials toward very different products and then test which ones most effectively slow the growth of cancer cells in the lab.
Shining Blue Light on New Molecules
The study centers on a family of organic molecules called carboxamides, which can be thought of as flexible “backbones” that are easy to modify. The researchers exposed these starting materials to blue LED light in the presence of selenium containing reagents and common acids such as acetic and formic acid. Rather than using harsh conditions or toxic additives, the reactions run in air and rely on visible light, making them relatively mild and straightforward to perform. By adjusting the type of amide and the solvent, the team discovered that the same ingredients could be coaxed into forming different products, ranging from open chain molecules to compact ring shaped structures.
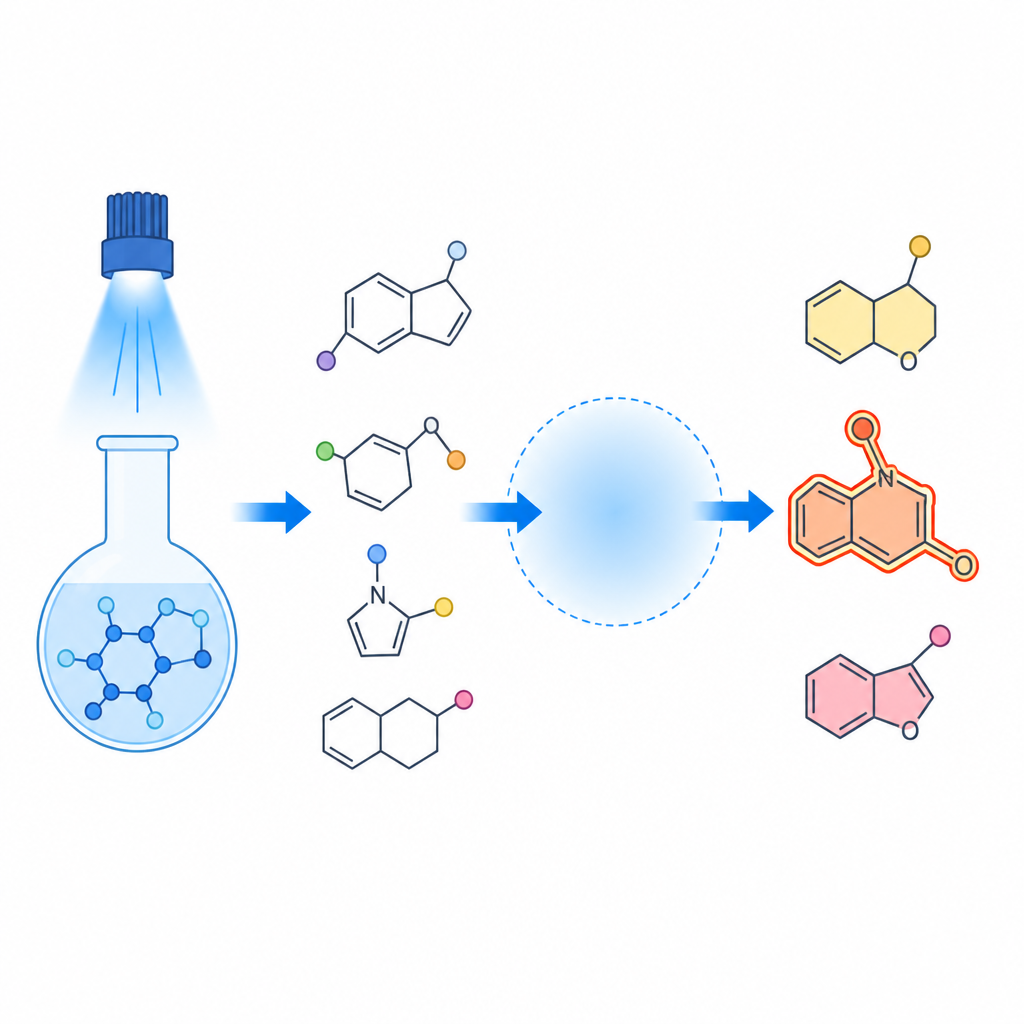
Steering Reactions Like Traffic
A key insight of the work is that the reaction behaves like a small traffic network at the atomic level. When blue light hits the selenium reagent, it splits and generates highly reactive fragments that quickly add to the carbon–carbon double bond in the starting amide. What happens next depends on the “environment” around that bond: the nature of the nitrogen side group, the strength and type of acid, and whether certain salts are present. In some cases, the acid helps an acetate group jump in, giving three component products known as beta acetoxyselenides. In other situations, the amide bends around and bites its own tail, closing into five or six membered rings known as perhydroindolones and piperidinones, while carrying a selenium atom in a defined position.
How Oxygen and Subtle Forces Shape the Outcome
The team probed how this light driven chemistry really works by repeating the reactions under different conditions. When they removed oxygen from the mixture, the process stopped, showing that air plays an essential role in activating the selenium reagent. Adding a common radical trap did not halt product formation, which suggests that only the very first step involves short lived radical species before the system settles into a more orderly ionic process. The balance between different products turned out to be highly sensitive to small changes. A stronger acid shifted the reaction away from oxygen based closure and toward nitrogen based ring formation, while a salt containing a weakly interacting anion helped push stubborn substrates into forming six membered lactam rings. Together, these tests led to a detailed proposal for how the reaction progresses through charged intermediates and why certain paths are favored.
From Test Tube to Cancer Cells
Among the many selenium containing piperidinones produced, one compound, labeled 12ea, stood out in biological tests. The researchers exposed several human cancer cell lines, including cervical and colorectal cancer cells, to increasing amounts of each compound and measured how strongly they reduced cell growth. Compound 12ea inhibited growth at low micromolar concentrations in multiple cancer lines while affecting normal lung fibroblasts less strongly, suggesting some degree of selectivity. Follow up experiments showed that treated cancer cells did not simply die by uncontrolled damage; instead, they underwent programmed cell death, or apoptosis. Markers of this process included activation of executioner enzymes called caspases, changes in membrane features detected by fluorescent probes, and formation of DNA damage foci inside cell nuclei.
Testing in 3D Tumor Like Clusters
To better mimic real tumors, the team grew the cancer cells as three dimensional spheroids, which are compact clusters that reproduce some of the oxygen and nutrient gradients found in the body. When these spheroids were treated with the lead compound 12ea, their growth slowed dramatically and the clusters became smaller and less dense over several days. Fluorescent staining revealed strong signals for apoptotic cell death throughout the spheroids at higher doses, showing that the compound can penetrate and remain active in a more realistic tumor like setting. This is important, because many drugs that work well on flat cell layers lose much of their effect in 3D models.
What This Could Mean for Future Treatments
Overall, the study delivers two main messages for non specialists. First, it demonstrates that simple blue LED light and judicious choice of conditions can give chemists fine control over how molecules assemble, allowing them to build complex selenium containing rings in a cleaner and more sustainable way. Second, it identifies one of these products, compound 12ea, as a selective and potent killer of certain cancer cells that acts through controlled cell death and remains effective in 3D tumor models. While this molecule is far from becoming a medicine and has not yet been tested in animals or people, it provides a well defined starting point for designing better analogs and exploring how selenium based chemistry can be harnessed in future anticancer therapies.
Citation: Vaskevych, A., Maciejewska, N., Mysiak, A. et al. Access to β-acetoxyselenides, perhydroindolones and anticancer piperidinones via blue LED-driven selenylation of unsaturated carboxamides. Sci Rep 16, 14963 (2026). https://doi.org/10.1038/s41598-026-44112-4
Keywords: organoselenium chemistry, blue LED selenylation, piperidinone anticancer, apoptosis in cancer cells, 3D tumor spheroids