Clear Sky Science · en
Analysis of immune-related alterations in blood and spinal cord of canine degenerative myelopathy, a spontaneous model of amyotrophic lateral sclerosis
Why a Dog Disease Matters to People
Many older dogs, especially Pembroke Welsh Corgis, can develop a fatal spine disease called degenerative myelopathy. This condition slowly robs them of the ability to walk and eventually to breathe. Strikingly, it closely resembles amyotrophic lateral sclerosis (ALS) in humans, also known as Lou Gehrig’s disease. Because dogs naturally develop this problem and share a key gene mutation with some ALS patients, studying them can reveal how nervous system damage and the body’s defenses interact in a real-world setting, beyond what can be seen in engineered mouse models.
Looking at Blood and Spine Together
The researchers set out to answer a central question: does the body’s immune system help drive this dog disease, or does it mainly react to nerve damage that is already under way? To explore this, they examined both the blood and spinal cords of dogs with degenerative myelopathy, and compared them with healthy dogs and dogs suffering from another spinal problem, intervertebral disc herniation, which compresses the spinal cord from the outside. They measured the activity of genes related to inflammation in blood cells and in spinal cord tissue, and used glowing antibody stains to see which immune and support cells were present in the spinal cord and how active they were.
Signals of Immune Alarm in the Blood
In the blood of affected dogs, the team found signs that certain white blood cells were switched into an “alert” state. Genes linked to a molecular alarm system known as the inflammasome, which helps produce inflammatory messengers, were more active than in control animals. At the same time, a gene for an immune “brake” molecule that tends to calm inflammation was also more active, suggesting the body was trying to keep this alarm under control. Routine blood tests, however, looked normal: white cell counts and a common inflammation marker called C-reactive protein remained within typical limits. This pattern points to a subtle, tightly regulated immune activation rather than a full-blown, body-wide inflammatory flare.
Who Enters the Spinal Cord, and Who Stays Out
The next issue was whether activated blood cells actually flood into the spinal cord, as happens in some mouse models of ALS. Using cell markers that distinguish resident brain immune cells from newcomers arriving from the blood, the scientists saw only a handful of likely immigrant cells in each entire cross-section of affected spinal cord. In other words, there was clear evidence that some blood-derived cells did enter the nervous tissue, but in very small numbers. At the same time, they saw increased production of a chemical signal in the spinal cord that normally attracts such cells from the bloodstream, indicating that the tissue itself was calling for help even if only a few responders arrived.
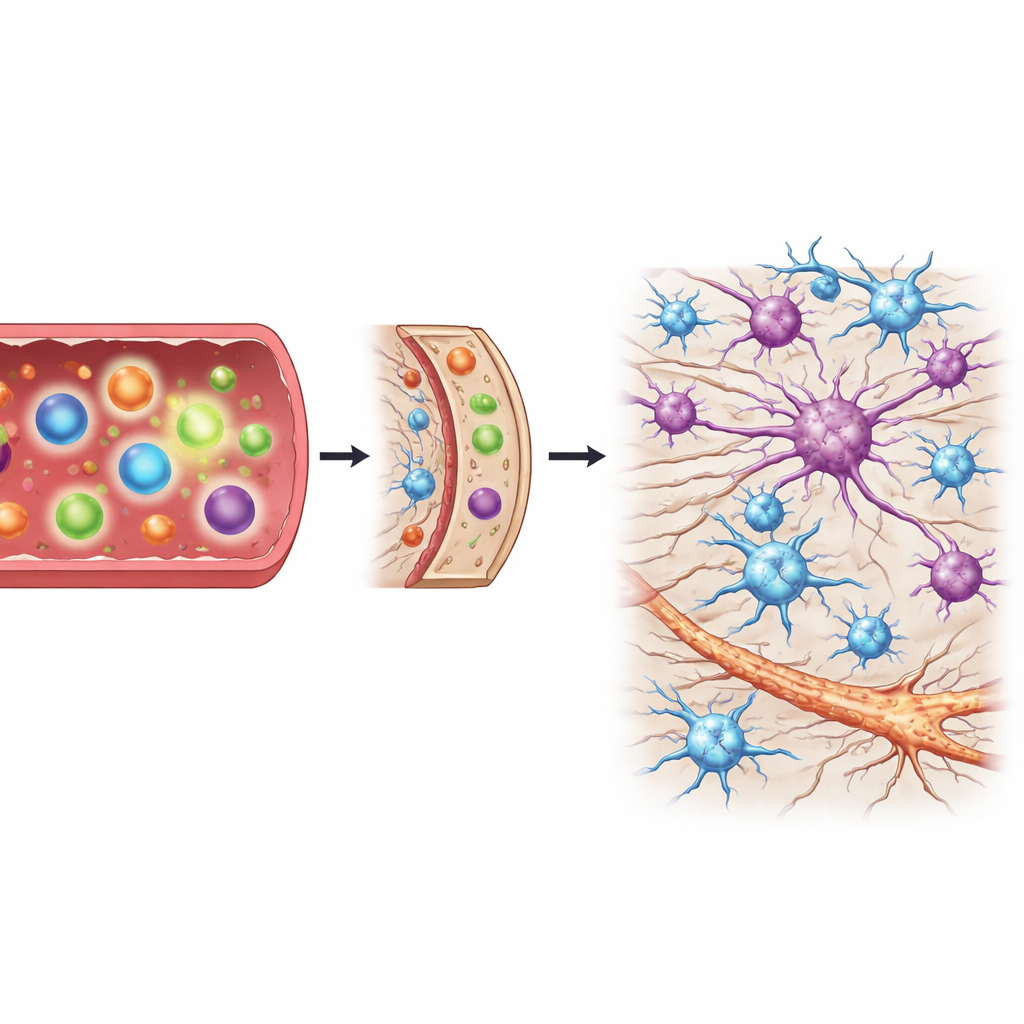
Support Cells React as Damage Worsens
The spinal cord is packed not only with nerve fibers but also with support cells called astrocytes and microglia, which can either protect or harm neurons depending on their state. By grouping spinal cord samples according to how badly they were damaged—from none, to mild, to moderate, to severe—the team could track how these support cells changed over time. As degeneration advanced, genes that mark both harmful and protective forms of astrocytes rose sharply, and the chemokine that calls in immune cells became more active. In contrast, markers for activated microglia did not show strong, early increases when damage was still mild. This timing suggests that the big wave of astrocyte activation and immune signaling appears after nerve injury has already taken hold, rather than at the very beginning.
What This Means for Dogs and People
Taken together, the results paint a picture in which degenerative myelopathy involves a quiet but complex immune shift: blood cells are primed, the spinal cord sends distress signals, and local support cells react strongly as damage accumulates. Yet the actual invasion of immune cells from the blood into the spinal cord remains limited, and dramatic inflammatory changes show up mainly in later stages. For dog owners, this means that by the time obvious signs of disease appear, immune changes are more a response to nerve loss than its original cause. For human medicine, these naturally occurring dog cases suggest that in ALS-like diseases, dialing down inflammation may still be helpful, but therapies that focus solely on blocking immune cells from entering the nervous system are unlikely to stop the disease at its root. Instead, treatments may need to target the underlying nerve vulnerability while carefully modulating, rather than simply shutting off, the body’s immune response.
Citation: Yokota, S., Kobatake, Y., Yoshida, K. et al. Analysis of immune-related alterations in blood and spinal cord of canine degenerative myelopathy, a spontaneous model of amyotrophic lateral sclerosis. Sci Rep 16, 13494 (2026). https://doi.org/10.1038/s41598-026-43838-5
Keywords: degenerative myelopathy, amyotrophic lateral sclerosis, neuroinflammation, canine model, immune response