Clear Sky Science · en
Exosome-mediated delivery of miRNA-1290 inhibitor enhances JNK-dependent tissue-resident memory T cell immunity in prostate cancer
Why this research matters
For many men, prostate cancer is a slow-burning threat that can suddenly become life‑ending once it stops responding to standard treatments. This study explores a fresh way to turn the body’s own immune system back against the disease, using tiny natural delivery packets from cells to carry a genetic "off switch" directly into tumors. The work points to a future in which prostate cancer might be controlled not only by drugs that hit the tumor itself, but also by carefully re‑arming the immune cells that live inside it.
A small genetic switch with big impact
At the heart of the study is a short piece of genetic material called miRNA‑1290. Such microRNAs act as fine‑tuners of many genes at once and can either encourage or restrain cancer. Earlier work had linked high miRNA‑1290 levels to aggressive tumors, but its role in prostate cancer and its value as a target for therapy were still unclear. The authors first confirmed that miRNA‑1290 is indeed elevated in mouse prostate tumors. They then designed a matching "inhibitor" that binds to miRNA‑1290 and blocks its action, with the hope that shutting it down would slow tumor growth and change how immune cells behave inside the cancer.
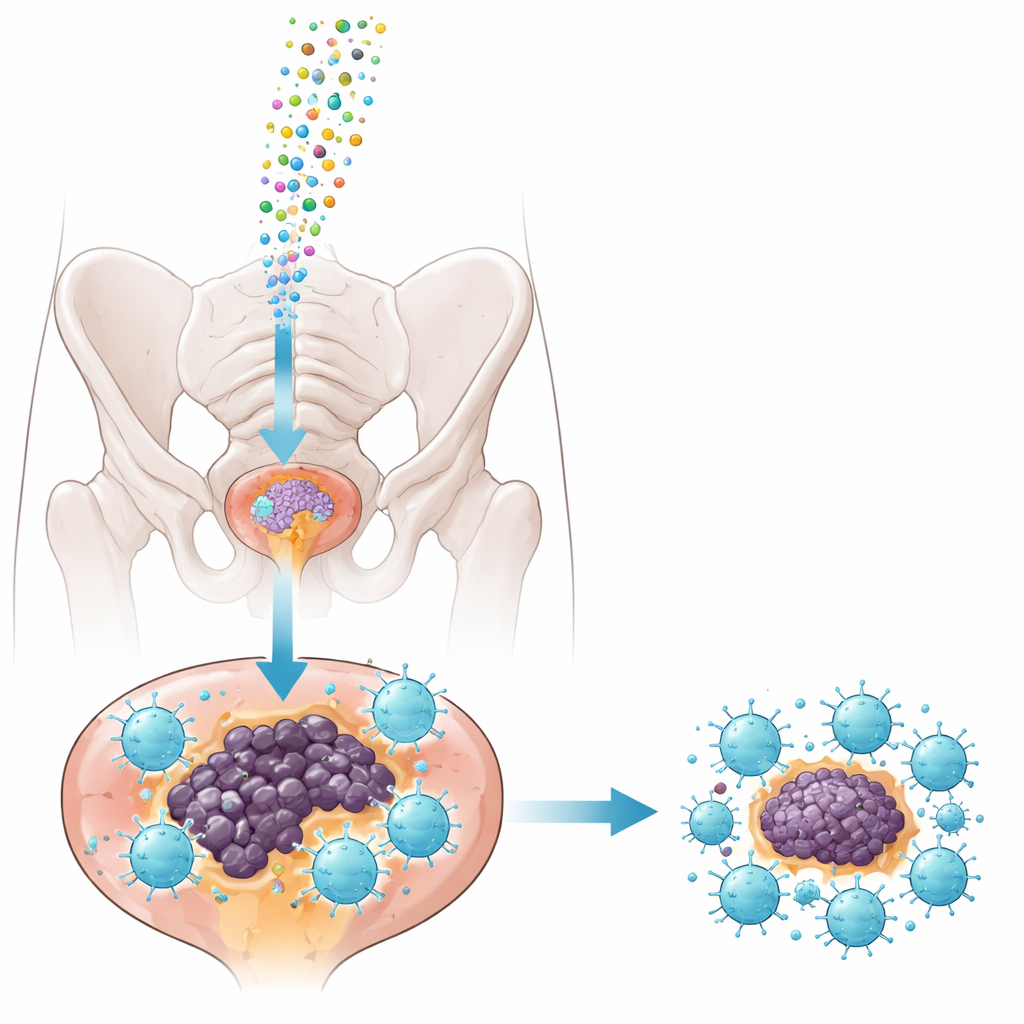
Tiny couriers: exosomes as delivery vehicles
To get the inhibitor safely into tumors, the team turned to exosomes—nanoscale bubbles naturally released by cells that can carry RNA cargo. They harvested exosomes from a mouse prostate cancer cell line, carefully purified them, and loaded them with either the miRNA‑1290 inhibitor or a harmless control sequence. Tests showed that the exosomes were uniform in size, kept their usual surface markers, and were readily taken up by tumor tissue when injected directly into prostate cancers in a well‑established mouse model (TRAMP mice). Importantly, the treatment did not disturb liver and kidney function or body weight in healthy animals, suggesting that the approach is relatively safe at the doses used.
Slower tumors and stronger local immunity
When tumor‑bearing mice received exosomes carrying the miRNA‑1290 inhibitor, their cancers grew more slowly, their body weight stabilized or improved, and they lived longer than control animals that received empty or control‑loaded exosomes. The inhibitor sharply reduced miRNA‑1290 levels in the tumor itself but not in blood or major organs, indicating that the effect was highly localized. To understand what was happening inside the tumors, the researchers profiled thousands of individual immune cells using single‑cell RNA sequencing and confirmed the findings with flow cytometry. They found a striking rise in overall T cells, in killer CD8 T cells, and especially in a special subset called tissue‑resident memory T cells (Trm), which take up long‑term residence in tissues and can respond rapidly to danger. Gene activity patterns in these cells showed enrichment of inflammatory and adaptive immune pathways, consistent with a more aggressive anti‑tumor response.
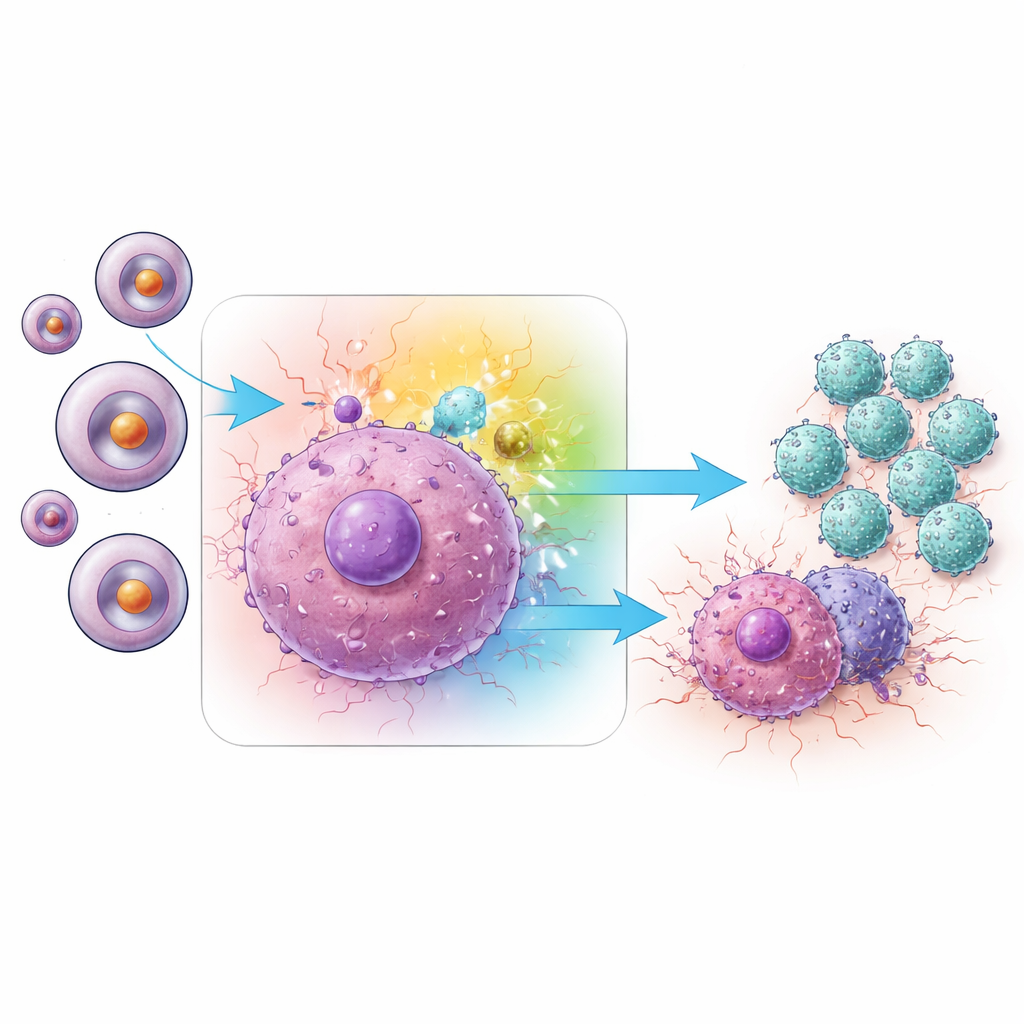
How one signaling route powers memory T cells
Diving deeper, the team asked which internal signaling routes in Trm cells were responsible for this heightened activity. Their analyses pointed to the MAPK network, and particularly to one branch known as JNK. In tumors treated with the miRNA‑1290 inhibitor, Trm cells showed higher levels of activated (phosphorylated) JNK compared with controls. At the same time, the tumors contained more of several pro‑inflammatory messenger proteins, including IFN‑γ and TNF‑α, which are classic weapons used by CD8 T cells to kill cancer cells. When the researchers selectively blocked different MAPK branches, they found that shutting down JNK—but not two related branches, ERK or p38—strongly reduced production of these key cytokines. Silencing JNK with small interfering RNA in Trm cells had the same dampening effect, firmly linking JNK activity to the boosted immune attack triggered by miRNA‑1290 inhibition.
What this could mean for future care
Taken together, the results show that packaging a miRNA‑1290 inhibitor into exosomes and injecting it into prostate tumors can slow cancer growth while awakening a powerful, long‑lasting local immune response centered on tissue‑resident memory T cells and their JNK signaling. Although the experiments were done in mice and used direct tumor injections—which may not be practical for all patients—the work offers a clear proof of principle. It suggests that miRNA‑1290 is both a driver of prostate cancer and a lever for reshaping the tumor’s immune environment, and that exosome‑based RNA medicines could become a new class of therapies that help the immune system hold prostate cancer in check.
Citation: Liang, B., Zou, S. Exosome-mediated delivery of miRNA-1290 inhibitor enhances JNK-dependent tissue-resident memory T cell immunity in prostate cancer. Sci Rep 16, 13472 (2026). https://doi.org/10.1038/s41598-026-43719-x
Keywords: prostate cancer, microRNA therapy, exosomes, tumor immune response, tissue-resident memory T cells