Clear Sky Science · en
A deep-SVM hybrid framework with enhanced EEG feature engineering and SHAP-based explainability for Alzheimer’s classification
Why brain wave tests could spot dementia earlier
Dementia, including Alzheimer’s disease and frontotemporal dementia, is rising worldwide, yet today’s diagnostic tools are often expensive, invasive, or hard to repeat regularly. This study explores whether simple brain wave recordings, taken with an electroencephalogram (EEG) cap, can help doctors distinguish between different types of dementia and healthy aging. The researchers also focus on making the computer’s decisions understandable to clinicians, so it is not just accurate, but also transparent and trustworthy.
Listening to the brain without surgery
EEG measures tiny electrical signals from the scalp as groups of brain cells fire together. These signals change as dementia progresses: faster rhythms tend to weaken, slower rhythms become stronger, and communication between brain regions can break down. Unlike brain scans or spinal taps, EEG is non-invasive, relatively inexpensive, and can be repeated as often as needed. That makes it an appealing candidate for large-scale screening and routine follow-up, provided that we can reliably decode the complex, noisy patterns hidden in the waves.
Blending human insight with machine learning
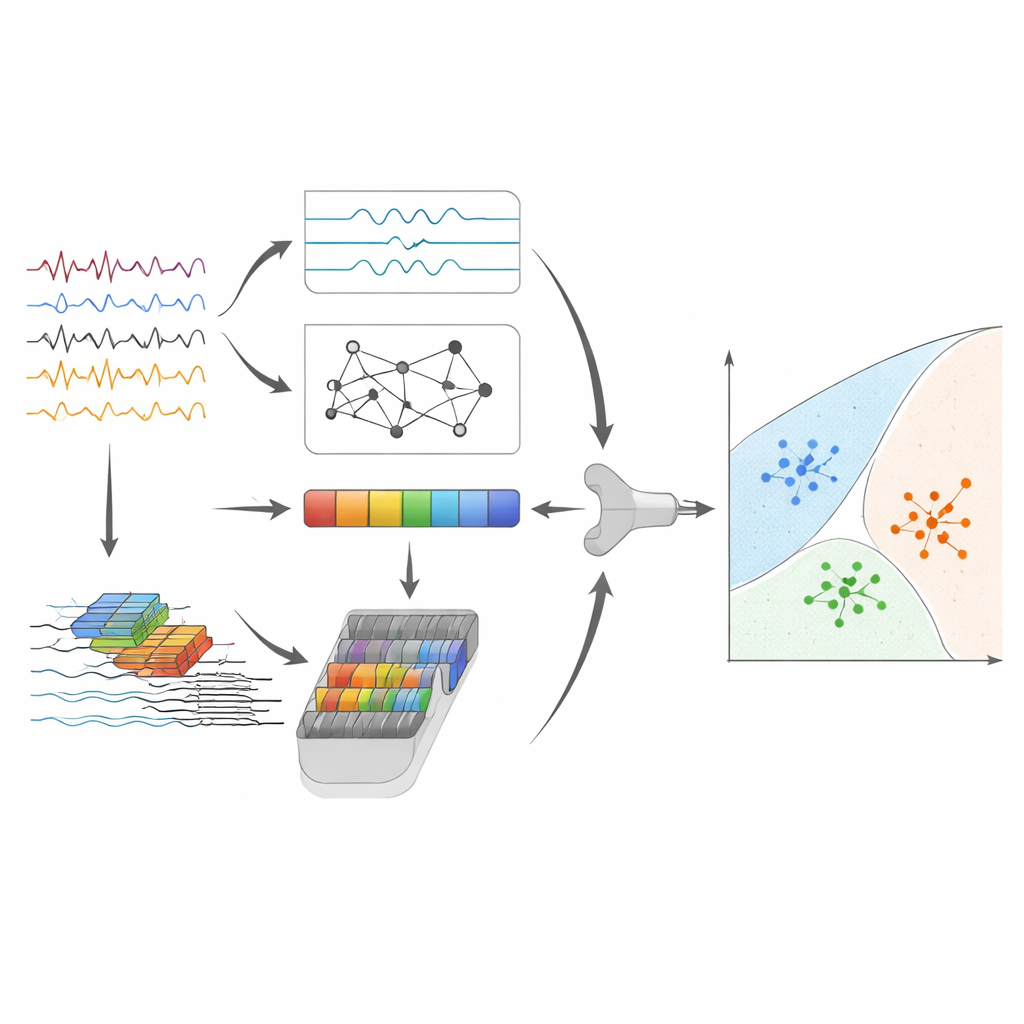
The team designed a hybrid computer system that combines two kinds of information from each 4‑second EEG segment. First, they engineered “handcrafted” features that neuroscientists already know are linked to dementia, such as how strong different frequency bands are (slow delta and theta, faster alpha and beta), how unpredictable the spectrum is, and how well left and right brain regions stay in sync in the alpha band. Second, they trained a compact one‑dimensional convolutional neural network to automatically learn useful temporal patterns directly from the cleaned signals. These two feature streams are standardized, fused into a single representation, and then compressed into a smaller set of components to avoid redundancy.
Making careful use of limited patient data
The main dataset consisted of resting EEG from 88 adults—people with Alzheimer’s disease, people with frontotemporal dementia, and cognitively normal volunteers—collected under consistent recording conditions. Because the number of volunteers is modest, the researchers took special care to avoid overestimating performance. They split the data by person, not by individual EEG segments, so a given participant’s brain waves never appeared in both training and testing. Within each training split, they balanced the classes using a controlled oversampling method, normalized the features, and tuned model settings such as how many principal components to keep and how strongly to regularize the final classifier. This nested evaluation design sharply reduces the risk of “leakage” and overly optimistic results.
High accuracy and insights into what the model sees
After feature fusion and compression, the final step uses a support vector machine, a classic algorithm that draws separating boundaries between groups of points in feature space. On the main dataset, this hybrid pipeline correctly distinguished among Alzheimer’s disease, frontotemporal dementia, and normal aging for about 95% of EEG segments, with equally strong performance across the three groups. To test how well the approach might hold up in new situations, the authors also applied it, without major redesign, to brain waves from the same people recorded with eyes open, and to a completely independent dataset recorded with different equipment and demographics. While performance naturally dipped, the system still showed meaningful discrimination, suggesting it is capturing disease-related patterns rather than quirks of a single study.
Opening the black box for clinicians
Beyond accuracy, a central aim of the work is explainability: doctors need to see why an algorithm leans toward a particular diagnosis. The researchers used a method called SHAP to estimate how much each feature “pushes” the model toward or away from a given class. By grouping these contributions into simple units—such as specific frequency bands and brain regions—they produced summaries that resemble traditional EEG reports. Patterns that boosted Alzheimer’s or frontotemporal dementia decisions included stronger slow theta activity over frontal areas, weaker alpha rhythms in posterior regions, and reduced alpha-band coordination between the two hemispheres, which match decades of clinical EEG findings.
What this means for patients and clinics
In practical terms, this study shows that a carefully designed EEG-based system can help separate two major dementia types from each other and from healthy aging with high reliability, while also explaining its reasoning in familiar physiological terms. If validated on larger, multi-center cohorts, such tools could support earlier, more precise diagnosis using affordable equipment, and could be integrated into clinic-friendly dashboards that show risk estimates and the brain-wave features driving them. Rather than replacing clinicians, the approach is intended to act as a second pair of eyes—highlighting subtle changes in brain activity that may signal when further investigation or closer follow-up is warranted.
Citation: Akbar, F., Alkhrijah, Y., Usman, S.M. et al. A deep-SVM hybrid framework with enhanced EEG feature engineering and SHAP-based explainability for Alzheimer’s classification. Sci Rep 16, 13001 (2026). https://doi.org/10.1038/s41598-026-43431-w
Keywords: EEG dementia diagnosis, Alzheimer’s disease, frontotemporal dementia, explainable AI, brain waves