Clear Sky Science · en
Neuroinflammatory crosstalk between microglia and astrocytes increases viral replication in an iPSC-derived model of CNS HIV infection
Why Brain Inflammation in HIV Still Matters
With modern HIV treatment, many people now live long, healthy lives and can keep the virus at undetectable levels in their blood. Yet a stubborn problem remains: a large share of people living with HIV still experience memory trouble, slower thinking, or difficulty concentrating, even while on effective therapy. This study asks a key question for patients and clinicians alike: if the virus is mostly controlled, why does brain inflammation—and the mental fog that can come with it—so often persist?
The Brain’s Support Cells Under the Spotlight
Instead of focusing on neurons, the nerve cells that carry signals, the researchers turned their attention to two “support” cell types in the brain: microglia and astrocytes. Microglia act like the brain’s resident immune cells, while astrocytes help maintain a healthy environment for neurons. The team used human stem-cell technology to grow lab-made versions of these cells—called induced microglia and induced astrocytes—from donor skin cells. This approach allowed them to closely mimic human brain biology without needing brain tissue from patients.
First, they confirmed that their lab-grown microglia behaved like genuine microglia: they had the right shape, expressed hallmark genes, and carried the receptors that HIV uses to enter cells. These microglia could be infected by HIV and allowed the virus to replicate, though not as efficiently as in blood-derived immune cells known as macrophages. Unlike macrophages, the microglia mounted only a weak inflammatory response to infection, producing few of the classic alarm molecules that usually accompany a strong antiviral reaction.
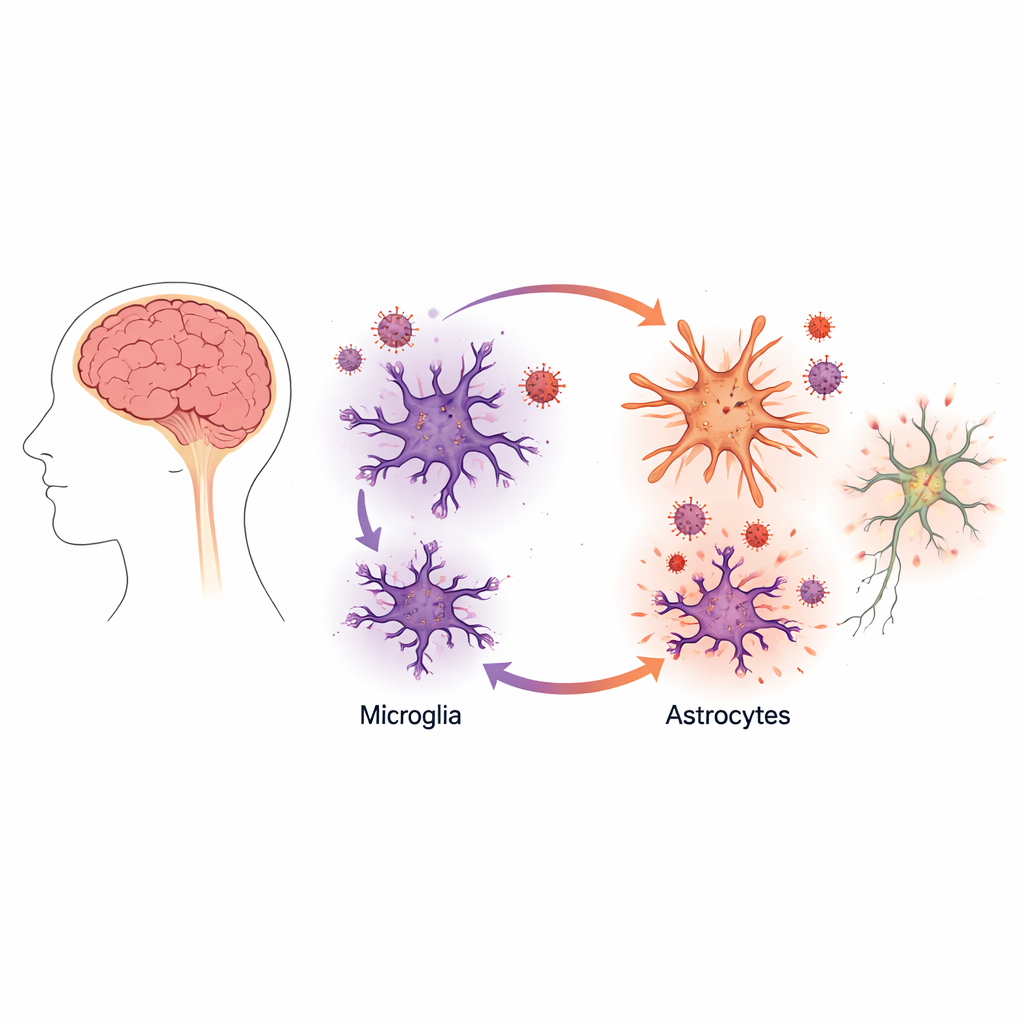
A Surprising Boost in Viral Growth
The picture changed dramatically when the scientists combined microglia with astrocytes in the same dish. In these mixed cultures, viral production by microglia shot up by about tenfold, even though the astrocytes themselves did not become productively infected. Careful imaging and measurements of viral proteins showed that HIV was still replicating mainly in the microglia. The presence of astrocytes, however, made those infected microglia far more active virus factories.
To understand why, the team examined the chemical signals released into the culture fluid. When astrocytes were exposed to material from infected microglia, they shifted into a reactive, inflammatory state that resembles changes seen in many brain diseases. These astrocytes began releasing higher levels of inflammatory messengers, including a molecule called tumor necrosis factor alpha (TNFα), which has long been linked to HIV activation.
How Inflammatory Signals Turn Up the Viral Volume
The researchers then asked whether TNFα was actually helping HIV replicate in microglia. When they added purified TNFα to infected microglia grown alone, viral output rose significantly. Blocking TNFα in mixed microglia–astrocyte cultures with an antibody drug reduced virus levels, though not all the way back to baseline, suggesting that other inflammatory factors also contribute. Further experiments pointed to a master switch inside microglia called NF-kB, a protein complex that moves into the cell nucleus and turns on many immune and stress-related genes, including those that drive HIV transcription.
Using a small molecule that blocks NF-kB activation, the team was able to cut the heightened viral replication in mixed cultures down to the level seen in microglia alone. This same treatment also reduced TNFα release. Imaging showed that, during infection, NF-kB shifted into the nucleus more strongly in microglia surrounded by astrocytes than in microglia alone, and this shift was reversed when the NF-kB blocker was present. Together, these findings outline a feedback loop: infected microglia disturb their surroundings, astrocytes respond by releasing inflammatory signals like TNFα, and those signals in turn push microglia to crank up HIV production via NF-kB.
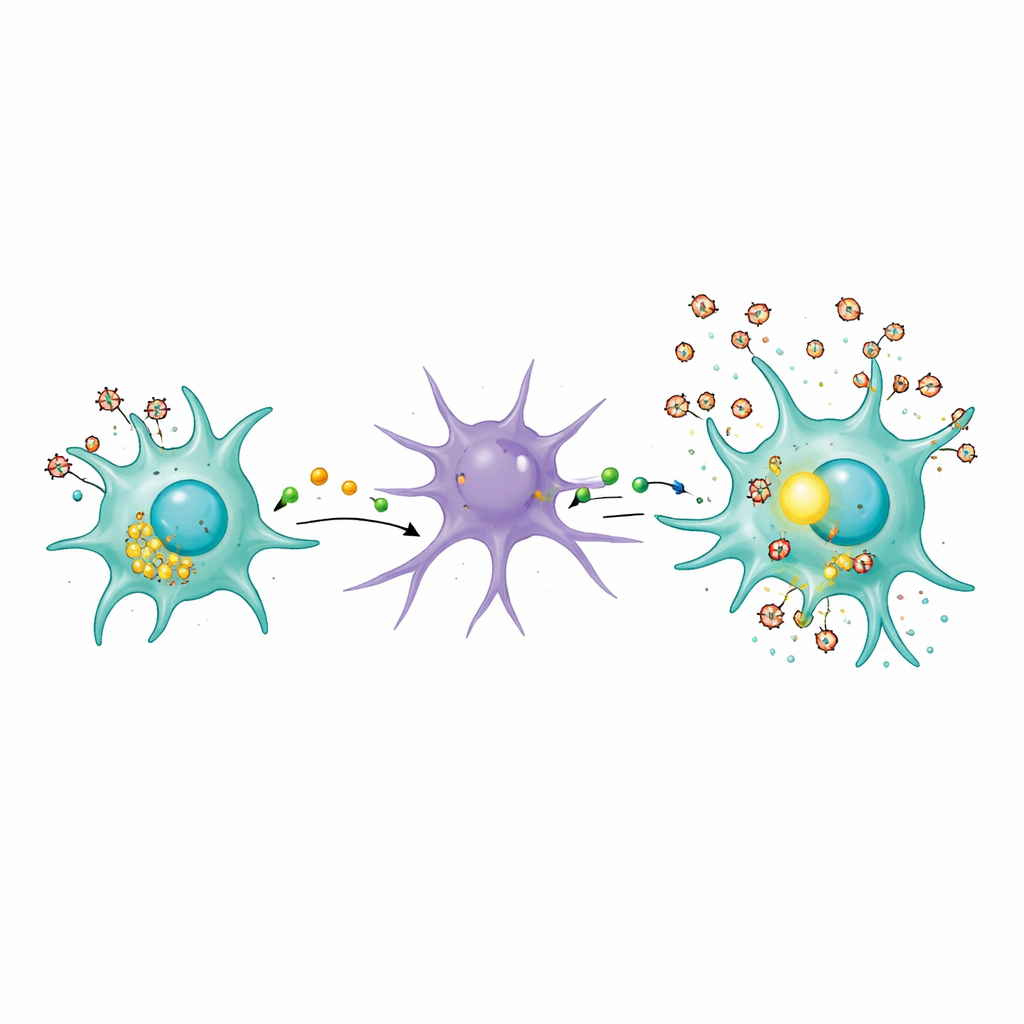
Lysosomes, Cellular Garbage, and a Toxic Neighborhood
The study also probed how infected microglia might initially rile up astrocytes. The authors focused on lysosomes, the cell’s waste-disposal and recycling centers. In infected microglia, lysosomes shifted toward the cell surface, and markers of their contents, such as an enzyme called cathepsin B, appeared in the surrounding fluid. This suggests that HIV infection prompts microglia to dump partially digested material into their environment. Even heat-inactivated virus—unable to replicate but still carrying viral proteins—was enough to disturb this lysosomal traffic. Such leakage can help create a chronically irritating neighborhood for nearby cells, nudging astrocytes into a harmful reactive state that feeds back into the inflammatory cycle.
What This Means for People Living With HIV
To a non-specialist, the main message is that the brain problems seen in many people living with HIV may not simply reflect how much virus is present, but how brain support cells talk to each other. In this work, infected microglia and reactive astrocytes form a vicious circle: subtle viral activity disrupts microglial waste handling, this agitates astrocytes, astrocytes release inflammatory signals like TNFα, and those signals drive microglia to make more virus. Even when standard drug therapy keeps overall viral levels low, this local loop could help sustain inflammation and slow, ongoing brain injury. The findings highlight new therapeutic angles—such as targeting TNFα, NF-kB signaling, or abnormal lysosome release—that might one day help protect thinking and memory in people whose HIV is otherwise well controlled.
Citation: Gesualdi, J., Prah, J., Solomon, S. et al. Neuroinflammatory crosstalk between microglia and astrocytes increases viral replication in an iPSC-derived model of CNS HIV infection. Sci Rep 16, 13047 (2026). https://doi.org/10.1038/s41598-026-43248-7
Keywords: HIV neurocognitive impairment, microglia, astrocytes, brain inflammation, TNF alpha