Clear Sky Science · en
Effect of alkyl chain length on the corrosion inhibition performance of 2-thioxo-2,3-dihydroquinazolin-4(1H)-one derivatives for carbon steel in HCl solution
Protecting Everyday Metals from Hidden Damage
From bridges and buildings to cars and household appliances, carbon steel quietly holds up much of modern life. Yet this workhorse metal has a weakness: in acidic environments, such as those used to clean industrial equipment or process oil and gas, it can dissolve away surprisingly fast. This study explores whether two small, easy-to-make organic molecules can act like invisible shields on steel, slowing this silent damage and extending the lifetime of vital infrastructure.
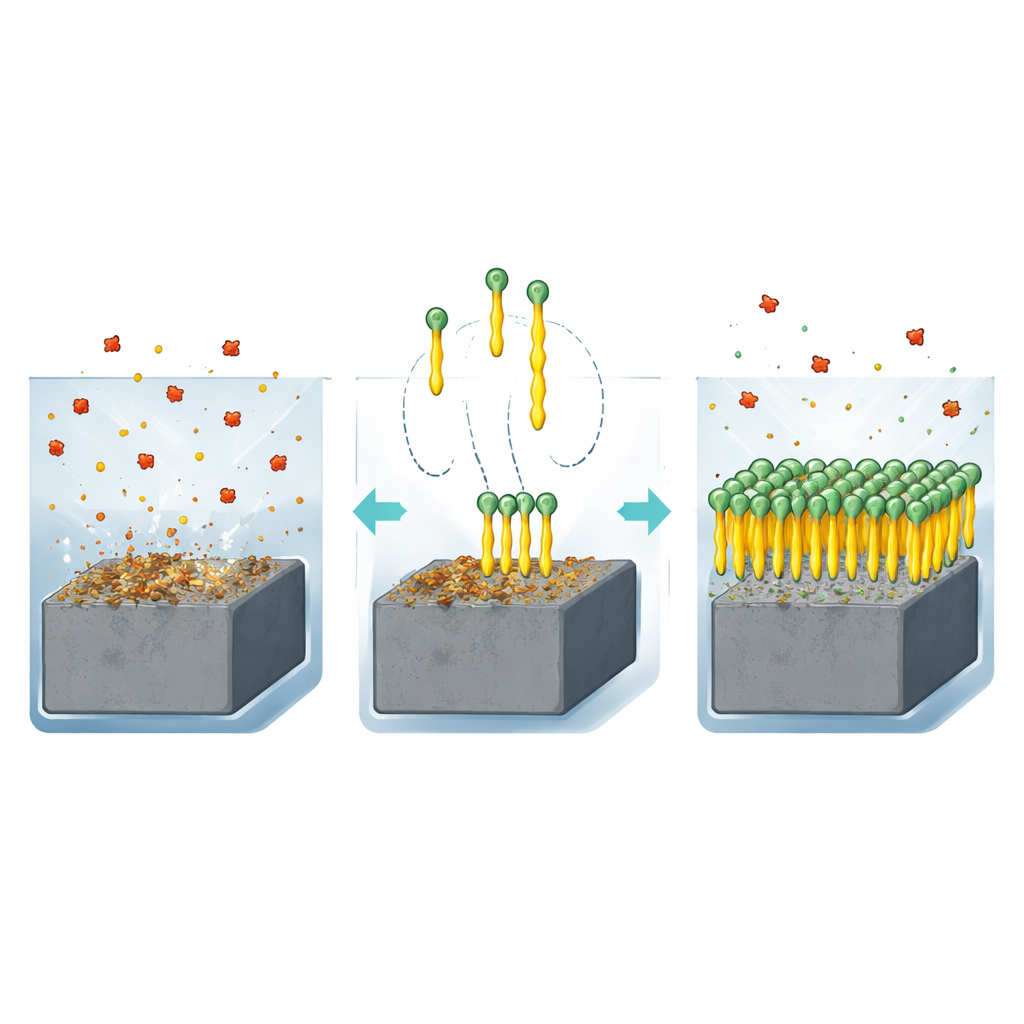
Why Steel Needs a Chemical Bodyguard
When carbon steel sits in an acidic liquid such as hydrochloric acid, atoms on its surface react and peel off into the solution. Over time, this corrosion can thin pipes, weaken tanks, and compromise safety. One of the most practical ways to fight this process is to add tiny amounts of special molecules called inhibitors to the liquid. These molecules attach to the steel surface and form a protective film that blocks further attack. The authors focused on a family of compounds known as quinazolinones, which contain nitrogen, sulfur, and oxygen atoms that can cling strongly to metal. They designed two very simple versions, identical except for the length of a single carbon chain: a short methyl group (Q‑C1) and a longer butyl group (Q‑C4).
Testing Two Small Molecules as Steel Shields
To see how well these compounds defend steel, the team immersed carbon steel samples in strong hydrochloric acid with and without the inhibitors. By carefully weighing the samples before and after exposure, they could tell how much metal had dissolved. They also used electrochemical tests, which track how easily electrical charge moves across the steel–solution interface, giving a sensitive measure of corrosion rate. Across a range of concentrations, both molecules significantly reduced the loss of steel, with the longer‑chain Q‑C4 reaching nearly 89% protection at its best conditions. However, the protective effect weakened at higher temperatures, suggesting that the inhibitor film becomes less stable as heat encourages the molecules to detach from the surface.
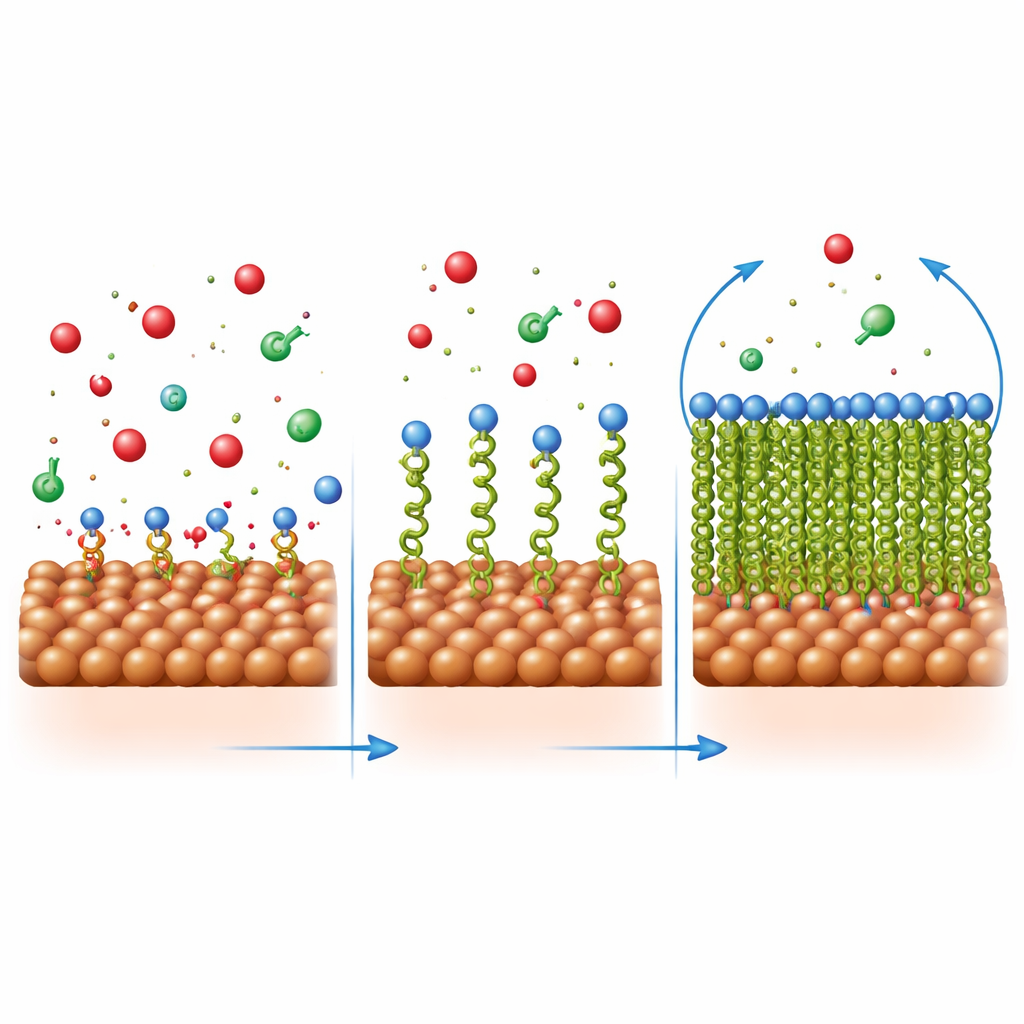
How the Protective Film Forms and Holds Together
The researchers probed the way these molecules sit on steel by analyzing how coverage changes with concentration. The pattern followed a simple “one layer” model, meaning each inhibitor molecule occupies a distinct spot on the surface rather than stacking up in thick piles. Calculations of energy changes showed that the attachment involves both physical attraction (through charge differences) and chemical bonding (through sharing electrons with the metal). Microscopic images supported this picture: bare steel in acid developed pits and cracks, while steel in inhibited acid appeared much smoother, covered by a more uniform layer. Elemental analysis and infrared spectroscopy confirmed that the organic molecules were indeed present on the steel surface and that their bonding groups had interacted with iron atoms.
Why the Longer Chain Works Better
Although both molecules share the same “head” that grips the steel, their “tails” behave differently. The longer butyl chain in Q‑C4 is more water‑repelling than the short methyl chain in Q‑C1. Once the head group anchors to the metal, this tail helps build a thicker, more complete film that keeps the acidic solution at arm’s length. Computer simulations using density functional theory backed this up: Q‑C4 was calculated to have a slightly stronger ability to donate electrons and a softer electronic structure, which promotes stronger interaction with the metal. Together, these traits help Q‑C4 pack more tightly and form a more effective barrier, leaving fewer exposed spots where corrosion can start.
What This Means for Real‑World Steel Protection
In plain terms, the study shows that very simple, low‑cost organic molecules can give carbon steel substantial protection in harsh acidic environments, and that tweaking something as modest as the length of a carbon chain can make a noticeable difference. The longer‑tailed inhibitor forms a denser, more resistant coating, cutting corrosion far more than its shorter cousin. While protection drops at higher temperatures, the work offers a clear roadmap: by adjusting molecular structure, especially chain length, engineers can design tailored inhibitors that are easier to make, effective at low doses, and suitable for large‑scale use in industry to keep steel structures safer and longer‑lasting.
Citation: El-Maksoud, S.A.A., Fathalla, W., Saleh, M.S. et al. Effect of alkyl chain length on the corrosion inhibition performance of 2-thioxo-2,3-dihydroquinazolin-4(1H)-one derivatives for carbon steel in HCl solution. Sci Rep 16, 10982 (2026). https://doi.org/10.1038/s41598-026-40197-z
Keywords: carbon steel corrosion, corrosion inhibitors, quinazolinone compounds, acidic environments, surface protection