Clear Sky Science · en
Microtubules in the axon are GDP bound but adopt a stable GTP-like expanded state
How Cell Highways Shape Brain Wiring
Inside every nerve cell runs a network of microscopic tubes that act like highways, carrying signals and supplies over long distances. For the brain to work, especially as nerve cells grow long axons, these highways must be both sturdy and finely tuned. This study peeks at these tubes inside living-like human neurons at near‑atomic detail and finds that axonal "roadways" use an unexpected strategy to stay stable—one that could help explain how neurons develop, how traffic along axons is controlled, and why certain brain drugs work the way they do.
Tiny Tubes That Keep Neurons Going
Microtubules are hollow cylinders built from repeating protein building blocks and run through nearly all our cells. In neurons they are especially crucial: bundled into tight parallel arrays, they give axons their shape and serve as tracks for motor proteins that haul cargo between the cell body and distant synapses. Test‑tube experiments have long suggested a simple rule: when the building blocks carry a GTP molecule, the tube wall adopts an “expanded” shape and is stable; after GTP is broken down to GDP, the wall compacts and the tube becomes fragile and prone to fall apart. But no one had ever seen, at atomic resolution, what these microtubules look like inside a real human neuron.
Seeing Axonal Highways Atom by Atom
The authors grew human neurons from induced pluripotent stem cells and encouraged them to form spherical clusters that extended long axons across special electron‑microscope grids. Using state‑of‑the‑art cryo‑electron microscopy and tomography, they recorded thousands of images of microtubules within intact axons frozen in a near‑natural state. By computationally averaging many views, they achieved a 2.7‑ångström reconstruction—fine enough to distinguish the two related tubulin proteins, pinpoint the bound nucleotides, and even resolve ordered water molecules and metal ions. They could therefore determine not only how far apart the building blocks sit along each protofilament, but also exactly which chemical state those blocks were in.
A Stable Lattice That Breaks the Usual Rules
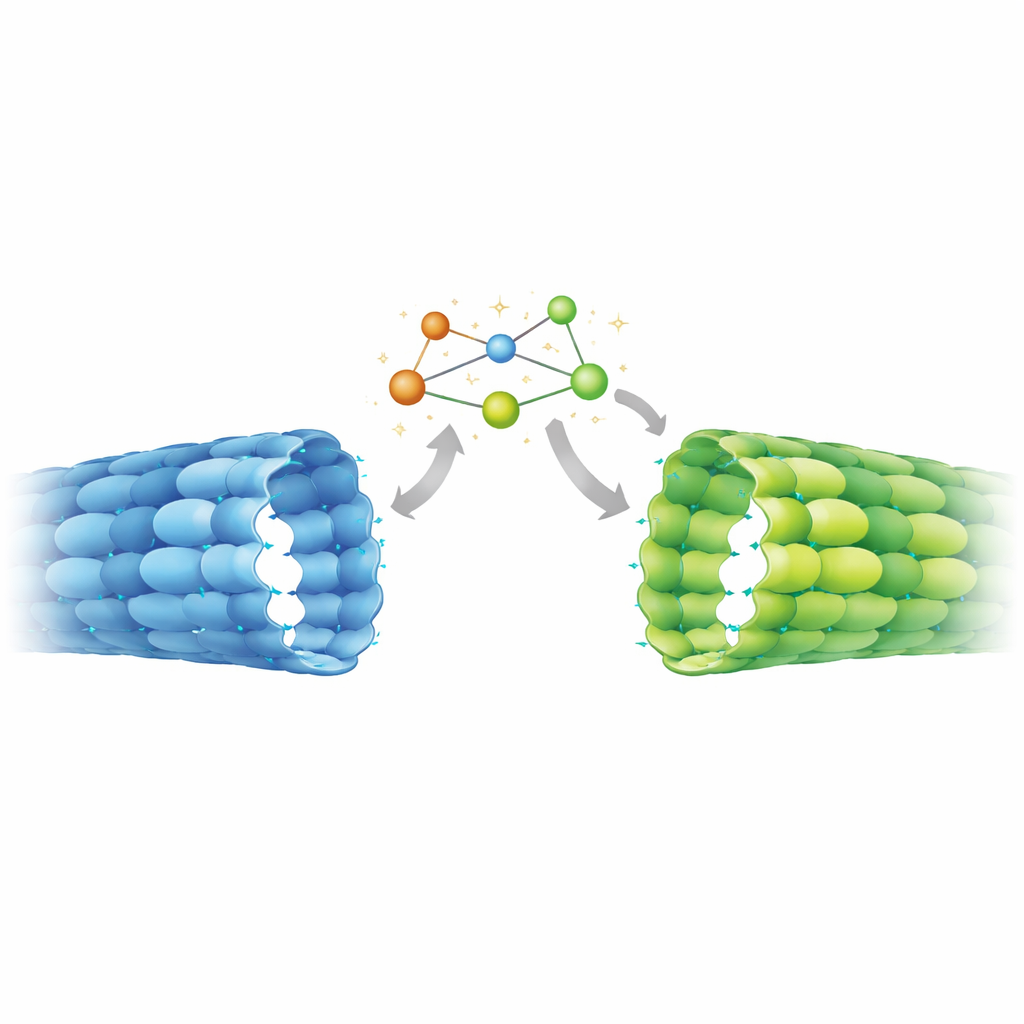
The high‑resolution structure delivered a surprise. In axonal microtubules, the exchangeable site in β‑tubulin clearly held GDP, indicating that GTP had been hydrolyzed as expected. Yet the spacing between tubulin pairs along the tube was expanded, matching what had previously been seen only in GTP‑like, stable lattices made in vitro. Detailed comparison with known structures revealed a hybrid state: regions of the protein that sense the presence of the γ‑phosphate behaved like classic GDP microtubules, while elements farther away—especially a mobile segment called the T5 loop at the junction between neighboring dimers—adopted the expanded, GTP‑like arrangement. A chain of aromatic amino acids appears to relay movements from this loop to neighboring contacts, locking the axonal lattice in a stretched, stable configuration despite its GDP chemistry.
Switching States as Neurons Grow Up
To find out whether this unusual geometry was a special feature of mature axons, the team measured lattice spacing in other contexts using cryo‑electron tomography and power‑spectrum analysis. In undifferentiated stem cells, microtubules showed more compact spacing, closely matching GDP‑microtubules assembled in the test tube. In contrast, microtubules within axons of differentiated neurons displayed expanded spacing, similar to the axonal atomic structure and to in‑vitro lattices stabilized by non‑hydrolyzable GTP mimics or the drug Taxol. This shift from compact to expanded geometry therefore seems to be part of the neuronal differentiation program, coinciding with the need to stabilize long axonal processes and reorganize them into dense bundles.
Implications for Brain Function and Therapy
This work shows that axonal microtubules are chemically in a GDP state yet structurally in a GTP‑like expanded form, stabilized by an internal mechanical switch. That means motors, regulatory proteins, and enzymes that decorate microtubules in neurons interact with a lattice type not faithfully mimicked by many standard lab preparations. Because these effectors can themselves favor expanded or compact spacing—and some chemical modifications to tubulin prefer expanded lattices—the discovered state likely feeds into a feedback loop that tunes cargo transport and microtubule longevity during brain development. The findings also hint that drugs like Taxol, which are known to expand the lattice, may promote axon growth in part by nudging microtubules toward the same stable conformation that naturally appears as neurons wire up.
Citation: Zehr, E.A., Sun, S., Sarbanes, S.L. et al. Microtubules in the axon are GDP bound but adopt a stable GTP-like expanded state. Nat Struct Mol Biol 33, 631–640 (2026). https://doi.org/10.1038/s41594-026-01787-7
Keywords: microtubules, axon, neuronal differentiation, cryo-electron microscopy, cytoskeleton