Clear Sky Science · en
AND logic nanoparticle for precision immunotherapy of metastatic cancers
Smarter Cancer Treatments That Know Where to Turn On
Many of today’s most promising cancer therapies work by rallying the immune system to recognize and destroy tumors. Yet these powerful drugs can also inflame healthy organs, causing serious side effects. This study describes a "smart" nanoparticle that behaves more like a tiny logic circuit than a simple drug capsule: it turns on a potent immune pathway only when it detects two hallmarks of aggressive tumors at once. The goal is simple but ambitious—hit widespread, hard‑to‑treat metastases while sparing the rest of the body.
Why Turning On Immunity Everywhere Is a Problem
Metastatic cancer, in which tumor cells seed distant organs like the lungs, causes the vast majority of cancer deaths. One attractive target for new drugs is a cellular alarm system known as the STING pathway, which helps immune cells sense danger and launch antiviral- and anti‑tumor defenses. When switched on in the right place, STING can wake up dormant immune responses and help T cells attack cancer. But when triggered too broadly or too strongly, it can exhaust or even kill immune cells and inflame healthy tissues. Earlier small‑molecule STING stimulators in clinical trials produced modest benefits but substantial side effects, in part because they lacked precise control over where and in which cells they acted.
Building a Tiny AND Gate for Tumor Signals
To solve this, the researchers designed nanoparticles that operate like an electronic AND logic gate: they release their STING‑activating cargo only if two separate tumor conditions are present at the same time. The particles are made from a pH‑sensitive polymer that assembles into micelles—nanosized spheres stable in the bloodstream’s neutral environment. A small‑molecule STING agonist is chemically tethered to this polymer through a linker that is broken down under low‑oxygen conditions. Many solid tumors, especially metastatic "immune‑cold" ones, are simultaneously more acidic and more hypoxic than normal tissues. Only in this combined setting does the particle disassemble and shed its drug payload. Test‑tube experiments confirmed the full truth table of an AND gate: without acidity, without hypoxia, or with only one of the two, almost no drug was released; with both, release became rapid and nearly complete. 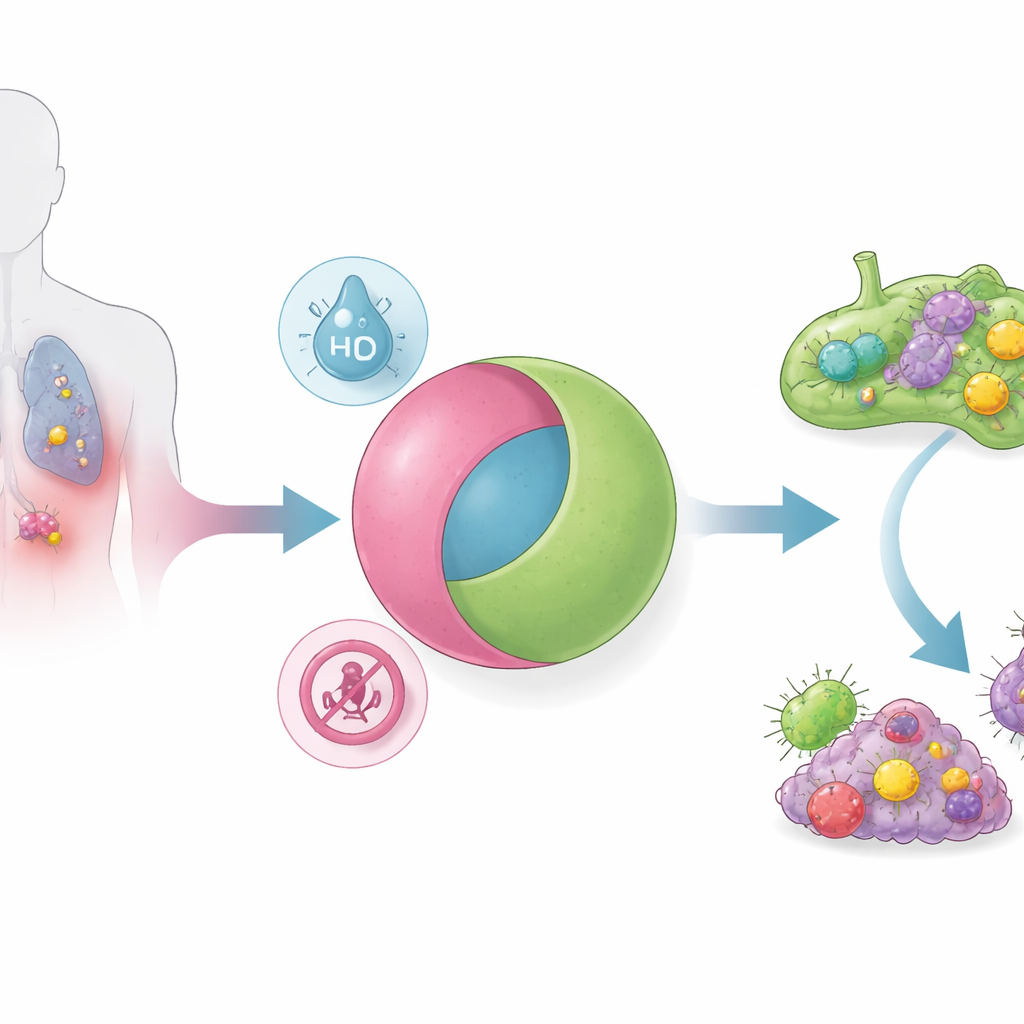
Directing the Spark to the Right Immune Cells
In mice, these AND‑gated particles circulated for hours without clumping and preferentially accumulated in the spleen, lymph nodes and lung metastases. Flow‑cytometry analyses showed that they were taken up mainly by key immune sentinels—dendritic cells and macrophages—rather than by T cells or by the cancer cells themselves. Under hypoxic conditions, dendritic cells naturally raised levels of an enzyme that snips the oxygen‑sensitive linker, further sharpening the tumor‑biased response. Once activated inside these cells, the particles drove strong production of interferon‑β and other alarm signals, but only when the local environment was both acidic and oxygen‑poor, helping confine STING activation to diseased tissues.
From Logic Gate to Tumor Control in Mice
The authors then tested their lead formulation, termed PHM nanoparticles, in several aggressive mouse cancer models, including lung metastases from Lewis lung carcinoma, triple‑negative breast cancer, and melanoma. A single intravenous dose greatly reduced the number of visible lung tumor nodules compared with free drug or with particles that responded to only one stimulus. In head‑to‑head comparisons that also tracked liver and kidney injury markers and inflammatory cytokines, the AND‑gated design delivered the best balance of strong tumor control and low systemic toxicity. Detailed immune studies revealed why: the treatment relied on an intact STING pathway, on a specialized dendritic cell subset (cDC1), and on killer CD8 T cells. In treated tumors, these dendritic cells showed high levels of activated STING and were often seen in direct contact with clusters of CD8 T cells, which had acquired potent tumor‑killing and memory features. 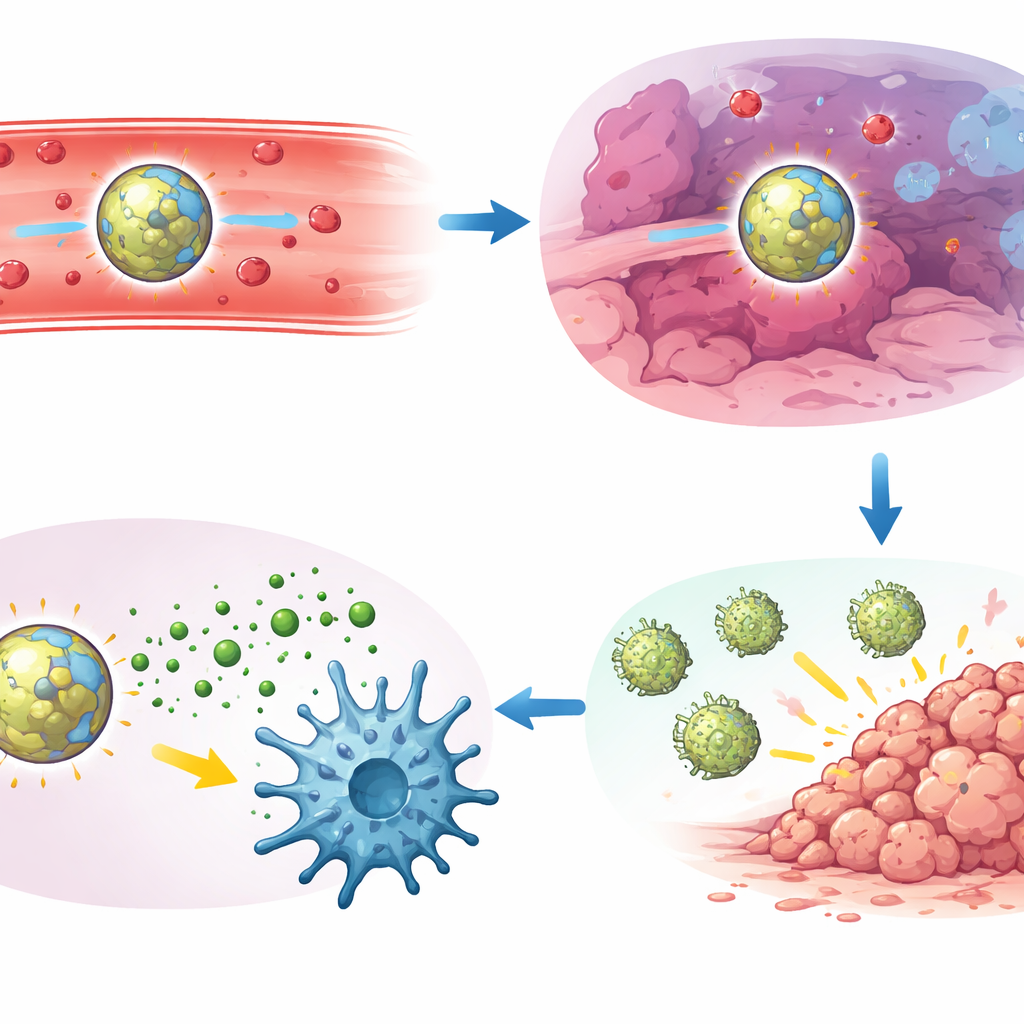
Lasting Protection and Partnership with Other Therapies
Beyond shrinking existing metastases, the nanoparticles helped train the immune system for the long haul. Mice that survived initial treatment could resist a later re‑challenge with the same cancer cells, indicating durable immune memory. In melanoma models, combining the particles with an immune checkpoint inhibitor further extended survival, suggesting that precise STING activation can synergize with existing immunotherapies. In a harsh breast cancer model, the particles alone markedly cut metastases and improved survival, though their combination with checkpoint blockade offered no extra gain, highlighting that optimal pairings may depend on tumor type.
What This Could Mean for Future Cancer Care
To a non‑specialist, the key message is that this work turns a powerful but blunt immune switch into something more discriminating. By building a molecular AND gate that listens for both acidity and low oxygen—conditions common in dangerous tumors but uncommon in most healthy tissues—the authors created a nanoparticle that can patrol the whole body yet ignite strong immune activity mainly where metastases lurk. While still at the animal‑study stage, this strategy could help make future cancer immunotherapies both safer and more effective, and the same logic‑based design could be adapted to other drugs and disease signals beyond cancer.
Citation: Ye, S., Chen, S., Basava, V. et al. AND logic nanoparticle for precision immunotherapy of metastatic cancers. Nat. Nanotechnol. 21, 606–616 (2026). https://doi.org/10.1038/s41565-026-02130-3
Keywords: cancer immunotherapy, nanoparticles, STING pathway, tumor microenvironment, metastasis