Clear Sky Science · en
Long-range chemical signalling in vivo is regulated by mechanical signals
How Touch Shapes Growing Brains
As a brain takes shape, billions of young nerve cells must navigate long, twisting paths to find their correct partners. For decades, scientists have known that these journeys are guided by chemical signals that spread like invisible scents through tissue. This study reveals a surprising partner to those chemical cues: the physical stiffness of the brain itself. By carefully probing developing frog brains, the authors show that how firm or soft the tissue is can switch distant chemical signals on or off, ultimately steering growing nerve fibers along the right routes.
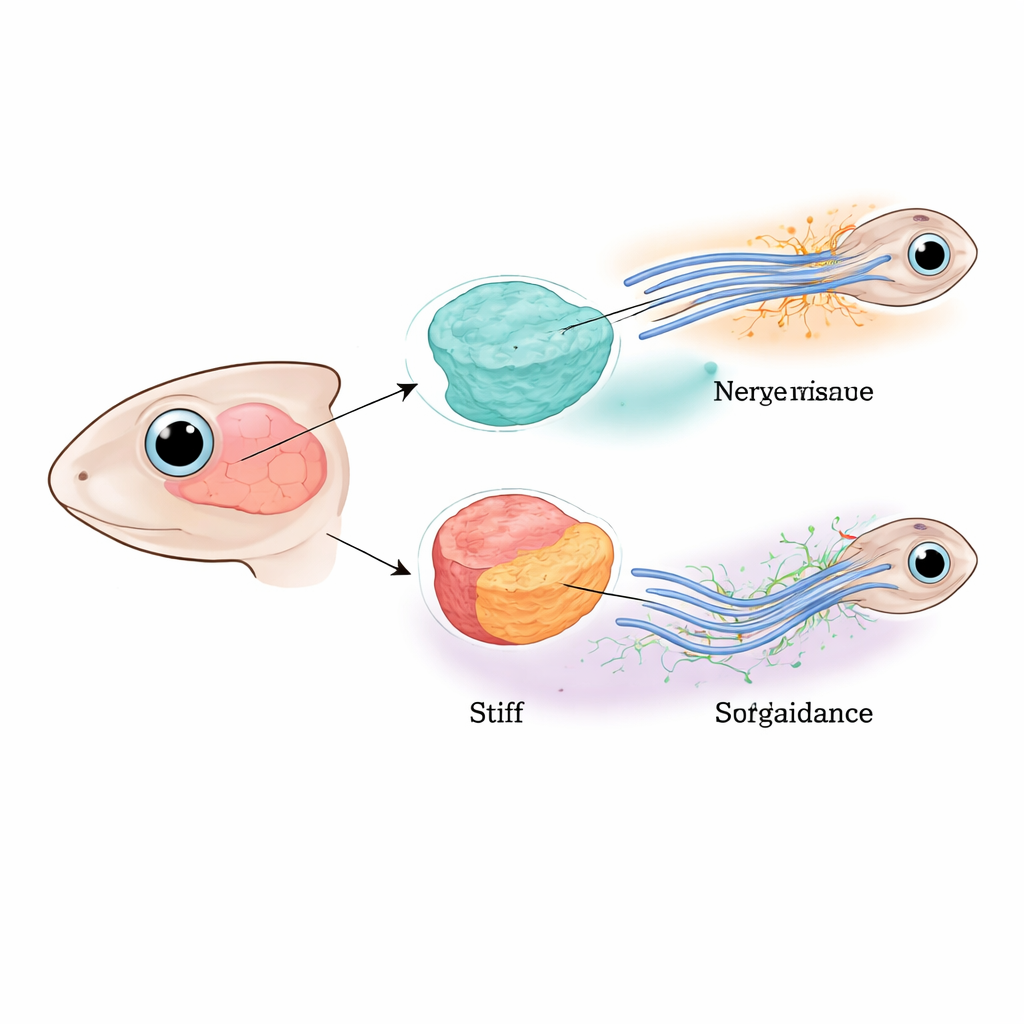
Guiding Nerves Across the Young Brain
The researchers focused on the optic pathway of young African clawed frogs, where nerve fibers from the eye must cross the brain and execute a precise turn before reaching their target region. Earlier work showed that these fibers are steered both by diffusing chemical repellents and by a gradual change in tissue stiffness along their route. Here, the team teased apart the roles of nerve cells themselves and the surrounding brain tissue by selectively reducing a key “force sensor” protein called Piezo1 in either the nerve fibers, the neighboring brain cells, or both. When Piezo1 was missing everywhere, many fibers stalled, split apart, or veered off course, confirming that proper mechanical sensing is essential for wiring the brain.
When the Brain Softens, Long-Distance Signals Fade
Next, the authors asked how the surrounding brain tissue contributes to this guidance system. When they removed Piezo1 only from the nerve fibers, the tissue’s stiffness remained normal, but pathfinding errors still occurred, indicating that the fibers could no longer feel their mechanical environment properly. Strikingly, when Piezo1 was removed from the brain tissue instead, the tissue became almost twice as soft, and guidance errors became even more frequent—even though the nerve fibers still contained Piezo1. In these softened regions, the levels of two known long-range guidance molecules, Slit1 and Semaphorin3A, dropped sharply at both the RNA and protein level. This showed that the brain’s physical state was not just passively sensed by cells; it actively controlled how much of these diffusing chemical cues were produced.
Cell Adhesion as a Hidden Mechanical Lever
What makes Piezo1-deficient brain tissue softer? The team ruled out simple explanations such as fewer cells or floppier individual cells. Instead, they found that major proteins that glue neighboring brain cells together—NCAM1 and N-cadherin—were reduced when Piezo1 levels fell. When the researchers directly lowered these adhesion proteins, tissue stiffness decreased and Semaphorin3A levels fell as well, even though the cell interiors remained mechanically unchanged. This points to a chain of events in which Piezo1 activity helps maintain strong cell–cell connections, which in turn set the bulk stiffness of the tissue, which then governs how much of certain long-range chemical signals the tissue produces.
Turning Up the Mechanical Dial
To test the flip side of this relationship, the authors increased brain stiffness in several ways. In lab dishes, they embedded small pieces of soft brain tissue in either soft or stiff 3D gels. On stiffer gels, the tissue pulled harder on its surroundings and began to produce much higher levels of Slit1 and Semaphorin3A, even in regions that normally do not make these cues. In intact tadpoles, they either applied a drug that boosts internal contractile forces or gently compressed specific brain regions with a tiny probe. Both approaches stiffened the chosen regions and triggered new Semaphorin3A production in areas that are usually chemically quiet. However, this mechanical “switch” failed in animals where Piezo1 had been reduced, showing that the channel is needed to translate extra stiffness into extra chemical signalling.
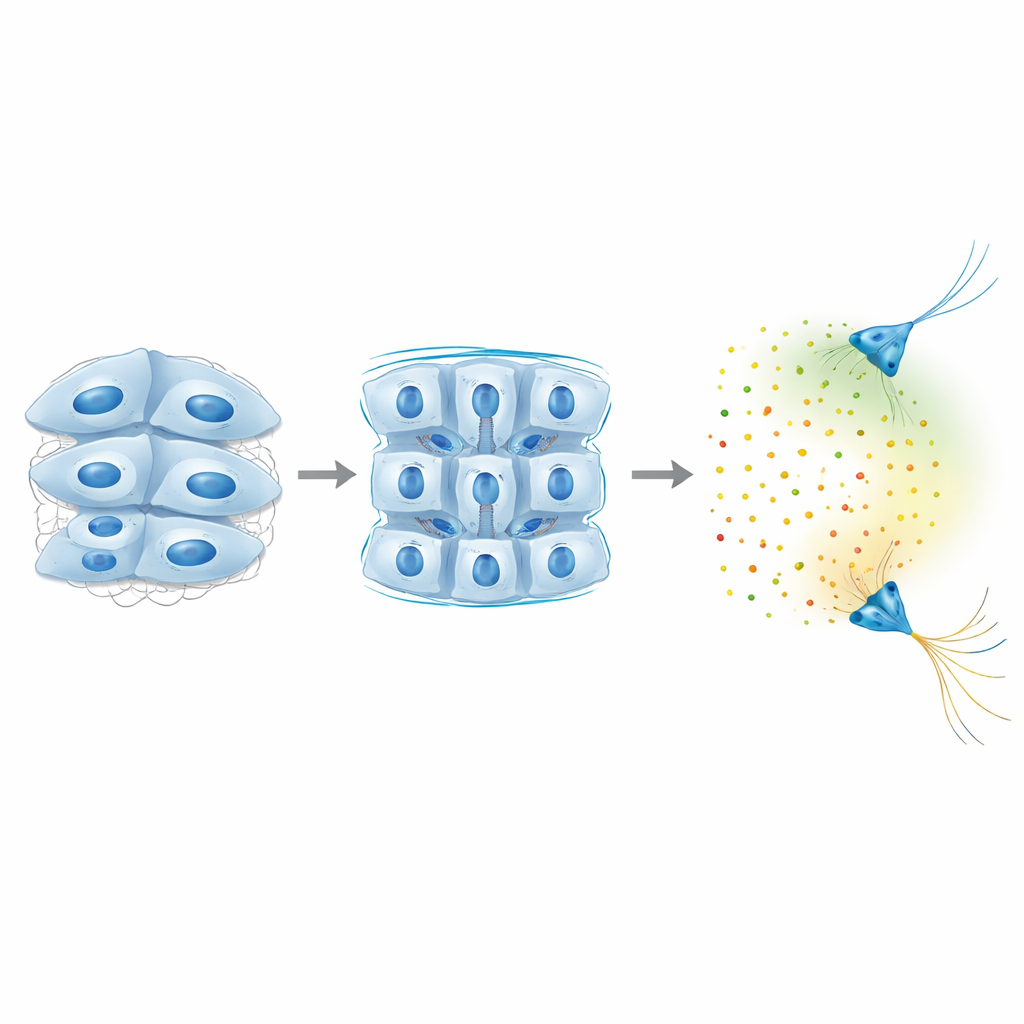
Why This Matters for Growing and Diseased Brains
Altogether, the study reveals a feedback loop in which brain cells pull on and sense their surroundings through Piezo1, adjust their adhesive connections to set how stiff the tissue is, and, only once a threshold stiffness is reached, turn on long-range chemical cues that steer distant nerve fibers. Rather than acting independently, mechanical and chemical signals are tightly intertwined: a local change in tissue firmness can reshape the “chemical weather” far away, influencing where axons grow and how circuits form. This insight suggests that altered tissue stiffness—seen in conditions from brain injury to neurodegeneration—could echo through developing or aging brains by changing the very signals that cells use to talk across long distances.
Citation: Pillai, E.K., Mukherjee, S., Gampl, N. et al. Long-range chemical signalling in vivo is regulated by mechanical signals. Nat. Mater. 25, 687–697 (2026). https://doi.org/10.1038/s41563-025-02463-9
Keywords: mechanotransduction, axon guidance, tissue stiffness, Piezo1, brain development