Clear Sky Science · en
Convergent transcriptomic and connectomic controllers of information integration and its anaesthetic breakdown across mammalian brains
Why this matters for everyday brains
When you drift off under general anaesthesia, your awareness of the world vanishes even though your brain is still physically intact. This study asks a simple but profound question: what actually changes inside the brain at that moment, and is that change shared across very different mammals and drugs? By blending brain scanning, genetics and computer models in humans, monkeys and mice, the authors uncover common control knobs that seem to govern how well the brain knits information together into a coherent conscious experience.
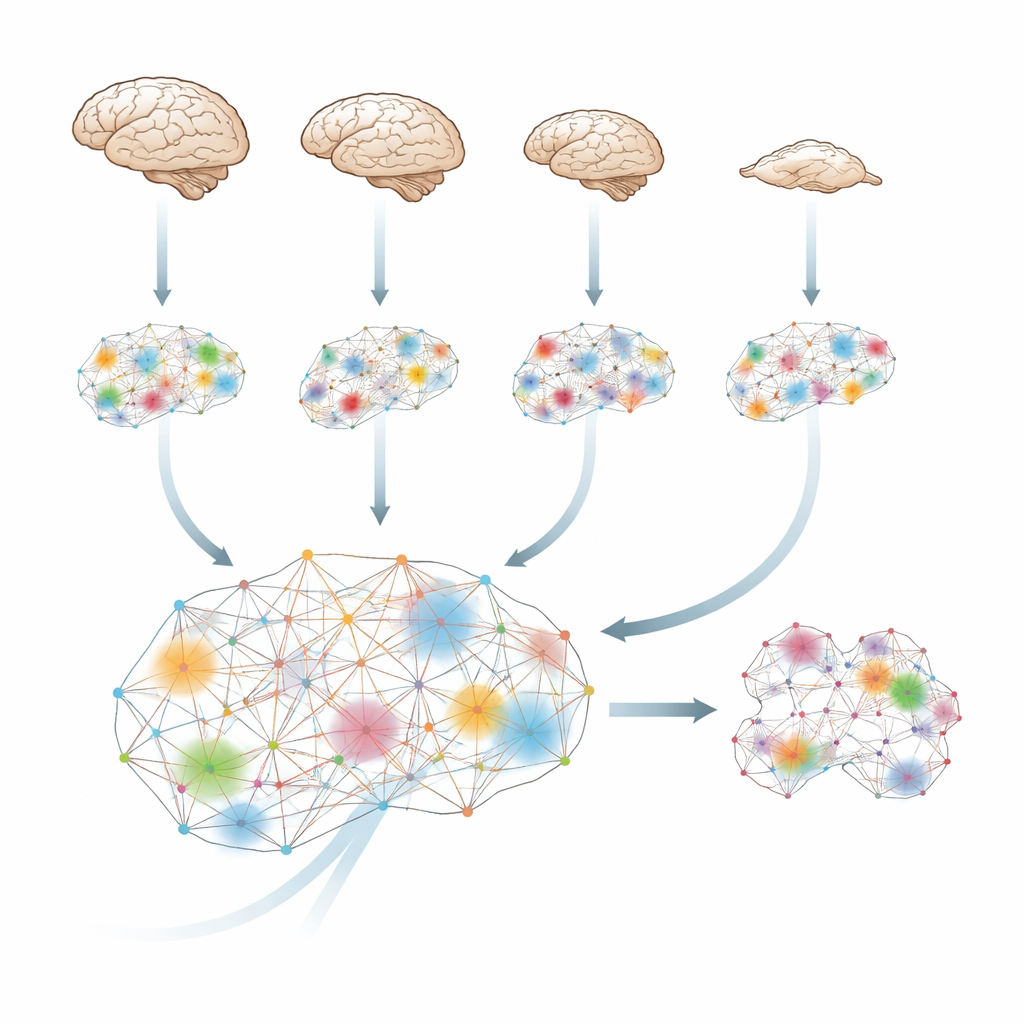
How the brain weaves information into a whole
The brain is constantly predicting and updating, so what happens in the next moment depends partly on what happened just before. Using tools from information theory, the researchers quantify how much "extra" information emerges when brain regions work together, beyond what each region carries alone. They call this integrated information: a measure of how strongly different parts of the brain act as a single, coordinated system rather than as isolated islands or simple echoes of each other.
What anaesthesia does across species
The team analysed functional MRI scans from four species—humans, macaques, marmosets and mice—while they were awake and under a range of anaesthetic drugs. Despite big differences in brains and in drugs, the pattern was strikingly similar. Whenever animals lost behavioural responsiveness, the level of integrated information across the brain dropped. In humans, this measure climbed back up as people spontaneously woke from sevoflurane. In macaques, electrical stimulation of a central thalamic region—deep inside the brain—restored both behaviour and integrated information even while the drug was still being infused. This suggests that anaesthesia’s core effect is not just to silence the brain, but to disrupt its ability to blend information into a unified whole, and that this loss can be reversed by targeted stimulation.
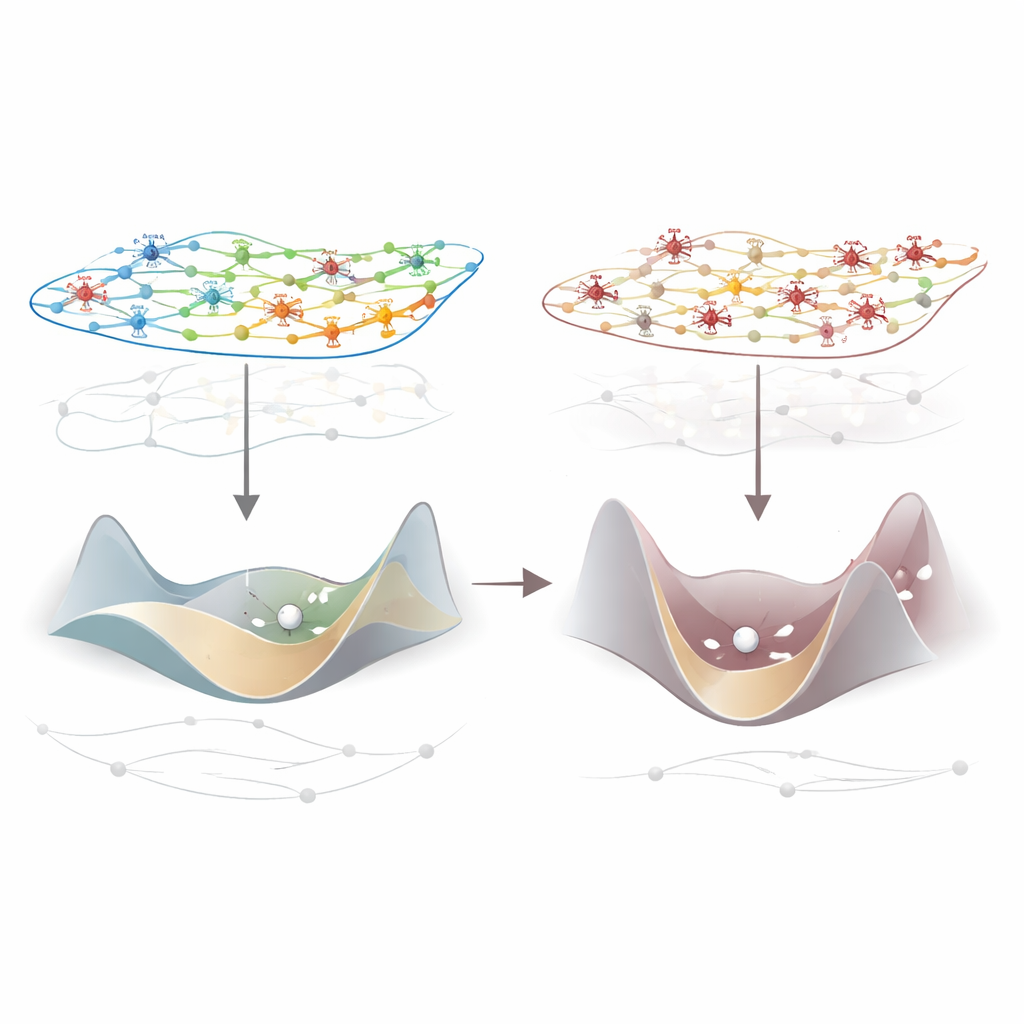
Why brain dynamics become harder to steer
To understand the mechanics behind this breakdown, the authors turned to network control theory, which treats the brain like a complex system that can shift between many activity patterns. Using species-specific wiring maps of how regions are physically connected, they calculated how much "energy" would be needed to move the brain from one momentary activity pattern to the next. Under anaesthesia, this control energy consistently increased: the landscape of possible brain states became steeper and harder to traverse. Importantly, this stiffening of dynamics relaxed again when subjects woke up or when thalamic stimulation restored responsiveness, and it was tightly linked to the drop in integrated information. In simple terms, when the brain is less steerable, it also becomes less able to integrate information.
Genes that tune local brakes on brain activity
Not all regions were affected equally. The biggest losses of integration appeared in primary sensory and motor areas, but changes were widespread. To ask why, the researchers overlaid these patterns with detailed maps of gene activity in human, macaque and mouse cortex. One gene stood out across species: PVALB, which marks a class of fast-acting inhibitory interneurons that put the brakes on local circuits. Regions with stronger PVALB expression tended to show larger anaesthesia-induced drops in integrated information. In mice, these regions also showed the strongest changes—whether increases or decreases—when lighter anaesthesia was used, suggesting that PVALB-rich areas are especially powerful levers for reshaping information flow.
Testing the mechanism in virtual brains
To move beyond correlation, the team built biophysically inspired whole-brain computer models for humans, macaques and mice, wiring them with each species’ actual anatomical connections. They then dialled up inhibition in each model region according to its PVALB level. Across all three species, this manipulation reduced integrated information in the simulated brain activity more than any random pattern of inhibition, and it raised the control energy needed to move between states, just as real anaesthetics do. In a separate macaque model, they added thalamic regions and simulated stimulation: exciting the central thalamus produced a much larger recovery of integrated information than exciting a nearby control nucleus, mirroring the animal experiments.
What this means for consciousness and recovery
Together, these converging lines of evidence point to a common story: in mammalian brains, diverse anaesthetic drugs act, directly or indirectly, on inhibitory circuits marked by the PVALB gene to make large-scale activity harder to control and less integrated. When that integration fails, the brain can no longer knit incoming signals into a unified, responsive state, and consciousness fades. Carefully targeted stimulation of the central thalamus can partially reverse this process. Beyond explaining how anaesthesia works, these findings suggest new strategies for restoring awareness in patients with severe brain injuries, by using brain wiring and gene-expression maps to predict the best regions to stimulate to reawaken the integrated dynamics that support conscious experience.
Citation: Luppi, A.I., Uhrig, L., Tasserie, J. et al. Convergent transcriptomic and connectomic controllers of information integration and its anaesthetic breakdown across mammalian brains. Nat Hum Behav 10, 777–802 (2026). https://doi.org/10.1038/s41562-025-02381-5
Keywords: consciousness, anaesthesia, brain networks, information integration, thalamic stimulation