Clear Sky Science · en
Lung tumouroids as a testing platform for precision CAR T cell therapy
Why Tiny Lung Models Matter for Patients
Lung cancer remains the deadliest cancer worldwide, in large part because every tumour is different and often learns to evade treatment. This study introduces a way to grow miniature, patient‑specific replicas of lung tumours in the lab and use them as a testing ground for advanced immune therapies called CAR T cells. By comparing these tumour “mini-organs” with matching healthy lung organoids from the same patient, the researchers show how doctors might one day predict which therapies will work, which will fail, and how to avoid dangerous side effects before treating the patient.
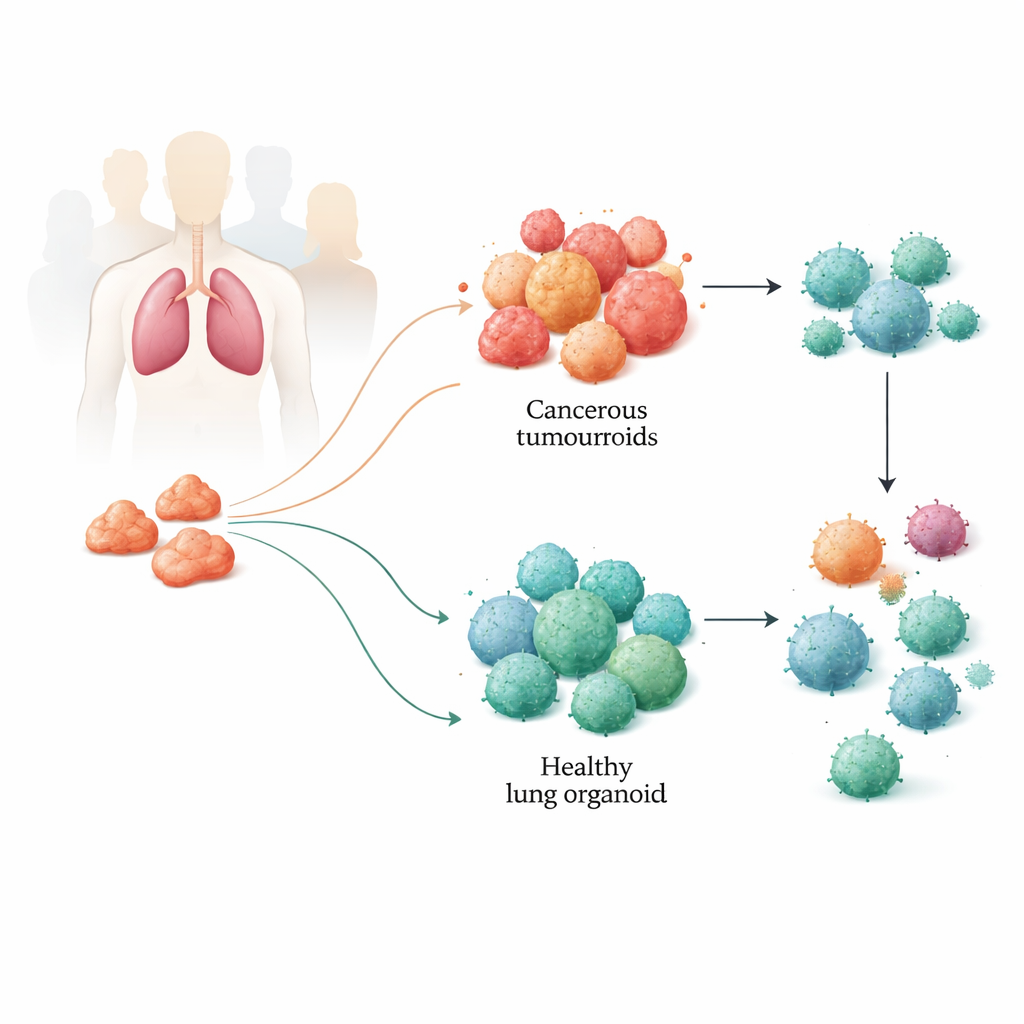
Building Miniature Lungs from Patient Tissue
The team collected tumour samples and nearby healthy lung tissue from patients undergoing lung cancer surgery. From these samples, they grew three-dimensional structures known as organoids: tumouroids from cancer tissue and healthy organoids from normal lung. These tiny spheres self-organize and can be kept alive for many months. Careful genetic, epigenetic and protein-level analyses showed that the tumouroids preserved the key molecular features of the original tumours, including common lung cancer mutations and their complex mix of cell types. The healthy organoids, in turn, retained the hallmarks of non-cancerous lung tissue, giving the researchers a reliable side‑by‑side model of each patient’s disease and normal lung.
Mirroring Real-World Drug Responses
To test whether these mini-organs truly reflect what happens in patients, the researchers exposed the tumouroids to the same chemotherapy and targeted drugs that the patients had already received. Using live-cell imaging and viability assays, they found that tumouroids often behaved like the original tumours did in the clinic: samples from patients whose cancers resisted a drug tended to be drug-resistant in the dish, while tumouroids from partial responders were more sensitive. Proteomic profiling—measuring thousands of proteins at once—revealed patterns linked to drug resistance, such as high levels of molecules previously tied to poor responses to platinum chemotherapy. These findings suggest that tumouroids could be used to anticipate whether a given drug is likely to help an individual patient.
Custom-Testing Next-Generation Immune Cells
The centerpiece of the study is a pipeline for tailoring CAR T cell therapy, a powerful approach in which a patient’s T cells are engineered to recognize specific markers on cancer cells. The scientists first screened tumour and healthy tissues, as well as their matching organoids, for potential target molecules on the cell surface. They then generated panels of CAR T cells—using a virus‑free gene editing method—to recognize promising targets such as HER2 or PDL1, which are present on some lung cancers. By co‑culturing these CAR T cells with tumouroids and healthy organoids, they could simultaneously assess how strongly the immune cells were activated, how much tumour damage they caused and whether they harmed healthy‑like lung tissue.
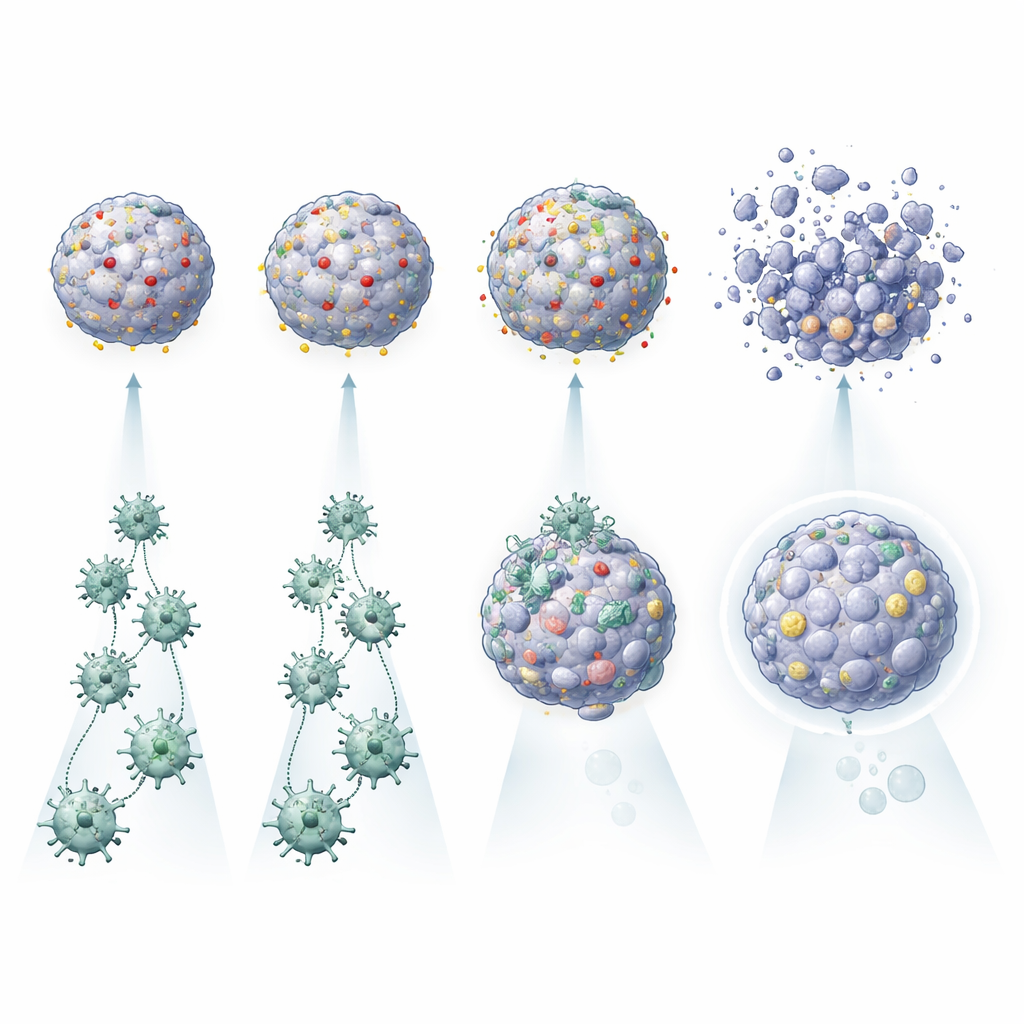
Uncovering Why Some Tumours Resist CAR T Cells
One striking result was that CAR T cell activation alone did not guarantee effective tumour killing. In some patients’ tumouroids, CAR T cells lit up and released inflammatory molecules but only modestly damaged the cancer mini-organs. In others, particularly those with many copies of the target molecule on the tumour surface, the same CAR T cells destroyed tumouroids far more efficiently. The platform also captured subtler influences: tumouroids that secreted certain cytokines, such as IL‑6, appeared to support better CAR T activity, while those rich in autophagy and immune‑evasion proteins, or capable of sharply increasing PDL1 under inflammatory conditions, were harder to eliminate. By comparing each tumour with its own healthy organoids, the team could also identify patients for whom a given CAR design might spare normal lung tissue—or pose unacceptable risks.
What This Could Mean for Future Cancer Care
In plain terms, this work shows that patient‑derived lung tumouroids can act as realistic “crash test dummies” for both standard drugs and sophisticated immune therapies. The model reproduces real patient responses to treatment, reveals why some tumours succumb while others resist, and highlights when a promising CAR T approach might also damage healthy lung cells. Although further refinement is needed, this platform points toward a future in which oncologists use a patient’s own mini‑tumours to choose and fine‑tune therapies—including bespoke CAR T cells—before exposing the patient to their risks.
Citation: Ehlen, L., Farrera-Sal, M., Szyska, M. et al. Lung tumouroids as a testing platform for precision CAR T cell therapy. Nat. Biomed. Eng 10, 815–831 (2026). https://doi.org/10.1038/s41551-025-01594-3
Keywords: lung cancer, organoids, CAR T cell therapy, precision medicine, immunotherapy resistance