Clear Sky Science · en
Generation of T cells with reduced off-target cross-reactivities by engineering co-signalling receptors
Turning Cancer-Fighting Cells into Sharper Weapons
Engineered immune cells are emerging as powerful tools against cancer, but they can sometimes mistake healthy tissues for tumors, with deadly consequences. This study explores a new way to make these cancer-fighting T cells much more selective, so they still attack tumors fiercely while largely ignoring look‑alike targets elsewhere in the body.
Why Off-Target Attacks Are So Dangerous
Many experimental therapies work by giving patients T cells outfitted with custom-made receptors that recognize small protein fragments on tumor cells. The challenge is that each T cell receptor can, in principle, respond to an enormous number of different fragments, not just the intended cancer ones. In past clinical trials, such cross-reactions have led engineered T cells to attack heart or nerve tissues, causing fatal side effects. Current safety strategies mostly try to redesign the receptor itself, a laborious process that must be repeated for every new therapy and can easily introduce new, unforeseen risks.
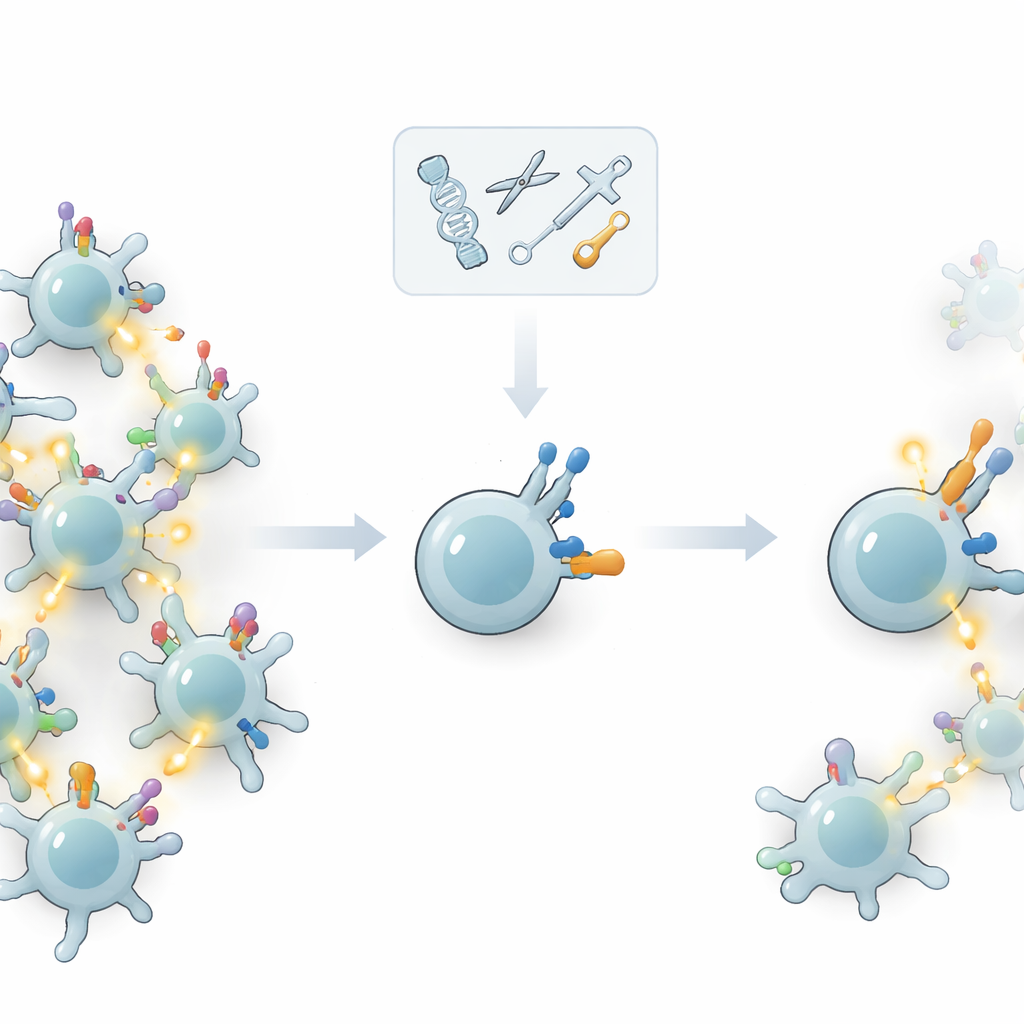
Rewiring the Support Hardware Instead of the Sensor
Instead of tampering with the main receptor, the authors asked whether they could tune the “support hardware” that controls how strongly a T cell reacts once its receptor has bound something. T cells carry additional surface molecules—co-receptors and other co‑signaling proteins—that help set the threshold for activation. By selectively removing or adding these molecules using gene editing, the team tested how well T cells could tell apart strong, likely tumor targets from weaker, potentially self‑derived targets. Their key idea was to increase the cell’s ability to ignore low‑quality signals without dulling its response to high‑quality ones.
Swapping Co-Receptors to Create Super-Selective Cells
The researchers focused on three well-known co-receptors: CD8, CD4, and CD5. They found that deleting CD5 made T cells more easily activated by many different targets, potentially boosting anti‑tumor power but also broadening cross‑reactivity. In contrast, removing CD8, a molecule that normally helps killer T cells recognize targets, had the opposite effect: it sharply reduced responses to weaker binding peptides while preserving robust responses to strongly binding cancer peptides. Going a step further, the team introduced CD4—a co-receptor normally used by a different T cell subtype—into these CD8‑deleted killer cells. This “CD8→CD4 co‑receptor switch” dramatically raised the bar for activation, so that only high‑affinity cancer targets triggered full killing, while a broad variety of weaker, off‑target stimuli were largely ignored.
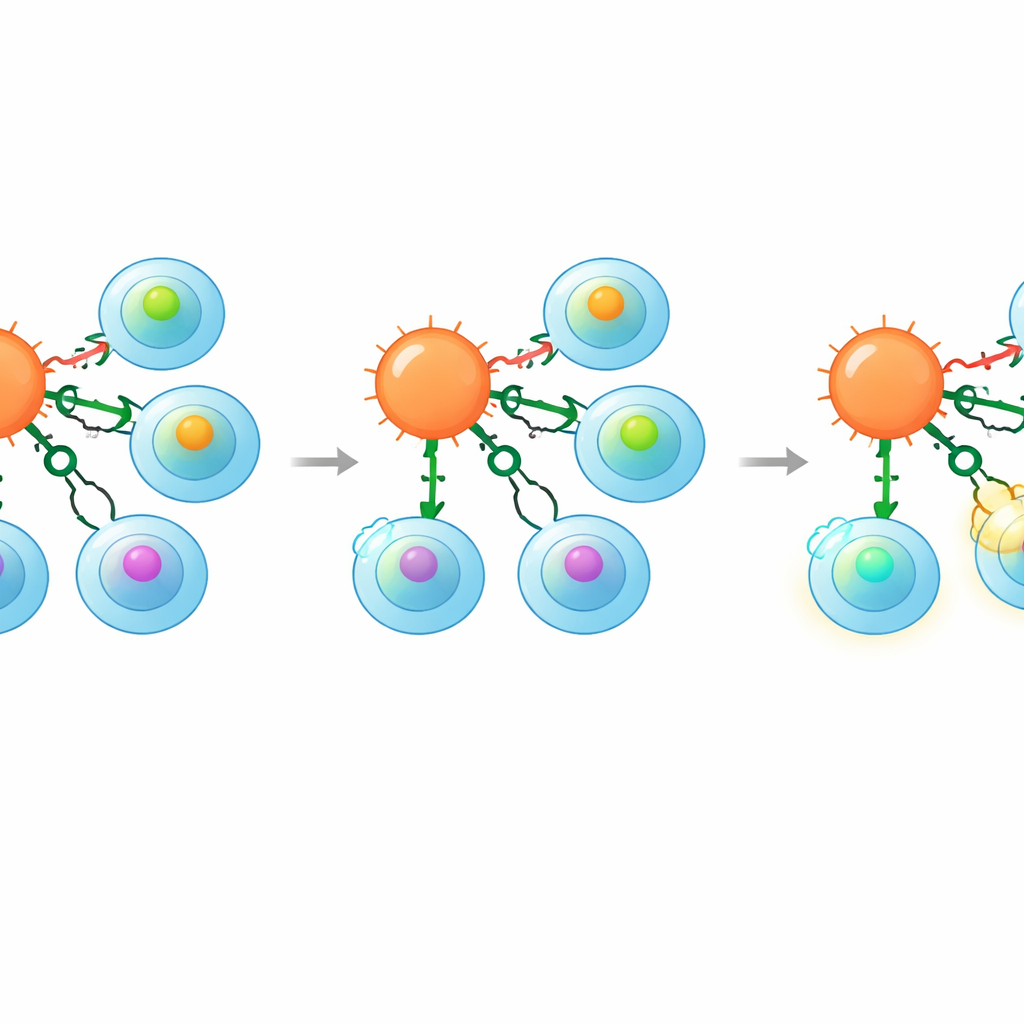
Putting Safety to the Test Across Many Potential Targets
To see whether this tighter selectivity truly translated into safer behavior, the authors challenged their engineered cells with several increasingly realistic tests. They first used libraries containing thousands of random peptides, most of which should bind weakly. Standard engineered T cells killed many cells presenting these random sequences, signaling high cross‑reactivity. In contrast, CD8‑knockout cells, and especially the CD8→CD4 switch cells, showed much less killing under the same conditions. Next, the team examined a focused library of single amino‑acid variants of a known tumor peptide and measured how strongly each variant bound the receptor. Again, cells with the co‑receptor switch ignored many of the lower‑affinity variants that still activated ordinary engineered T cells. Finally, they tested predicted self‑peptides from human proteins that were experimentally confirmed to activate the receptor. Here too, the switched cells were far less reactive, yet they retained strong potency against the original, high‑affinity tumor target.
Learning from Past Tragedies
The researchers also revisited a clinically important cautionary tale. An earlier trial used T cells bearing a receptor for a cancer protein called MAGE‑A3; those cells unexpectedly also recognized a weaker peptide from the heart muscle protein Titin, leading to fatal cardiac damage. In the new work, the authors showed that knocking out CD8 in killer T cells carrying this same receptor abolished their response to the Titin peptide while preserving attack on tumor cells that truly expressed MAGE‑A3. This demonstration suggests that co‑receptor engineering could help rescue otherwise promising receptors that had to be abandoned for safety reasons.
What This Could Mean for Future Cancer Therapies
From a lay perspective, the central message is that the team has found a way to sharpen immune cell “vision” by adjusting internal wiring rather than endlessly redesigning the lens. By switching the combination of helper molecules on T cells—especially by removing CD8 and adding CD4 when the receptor binds its cancer target strongly—the researchers create “super selective” T cells. These cells still attack tumors vigorously but are much less likely to be lured into attacking normal tissues that present similar, weaker signals. Because this approach leaves the main receptor sequence untouched, it could, in principle, be applied broadly to many different T cell therapies, offering a general route to safer, more precise cancer immunotherapy.
Citation: Cabezas-Caballero, J., Huhn, A., Kutuzov, M.A. et al. Generation of T cells with reduced off-target cross-reactivities by engineering co-signalling receptors. Nat. Biomed. Eng 10, 753–764 (2026). https://doi.org/10.1038/s41551-025-01563-w
Keywords: cancer immunotherapy, T cell engineering, T cell receptors, off-target toxicity, co-receptor switching