Clear Sky Science · en
A novel multi-epitope vaccine induces protective and therapeutic immunity against Helicobacter pylori
Why this matters for everyday health
Most people will never hear the name Helicobacter pylori, yet this spiral-shaped bacterium quietly infects about half the world’s population and can lead to stomach ulcers and even gastric cancer. Today the only reliable cure is a cocktail of antibiotics, but growing drug resistance is making these treatments less effective. This study describes a new type of experimental vaccine that not only prevents infection in mice but also clears long‑standing stomach infections, pointing toward a future in which a simple shot could replace complex drug regimens.
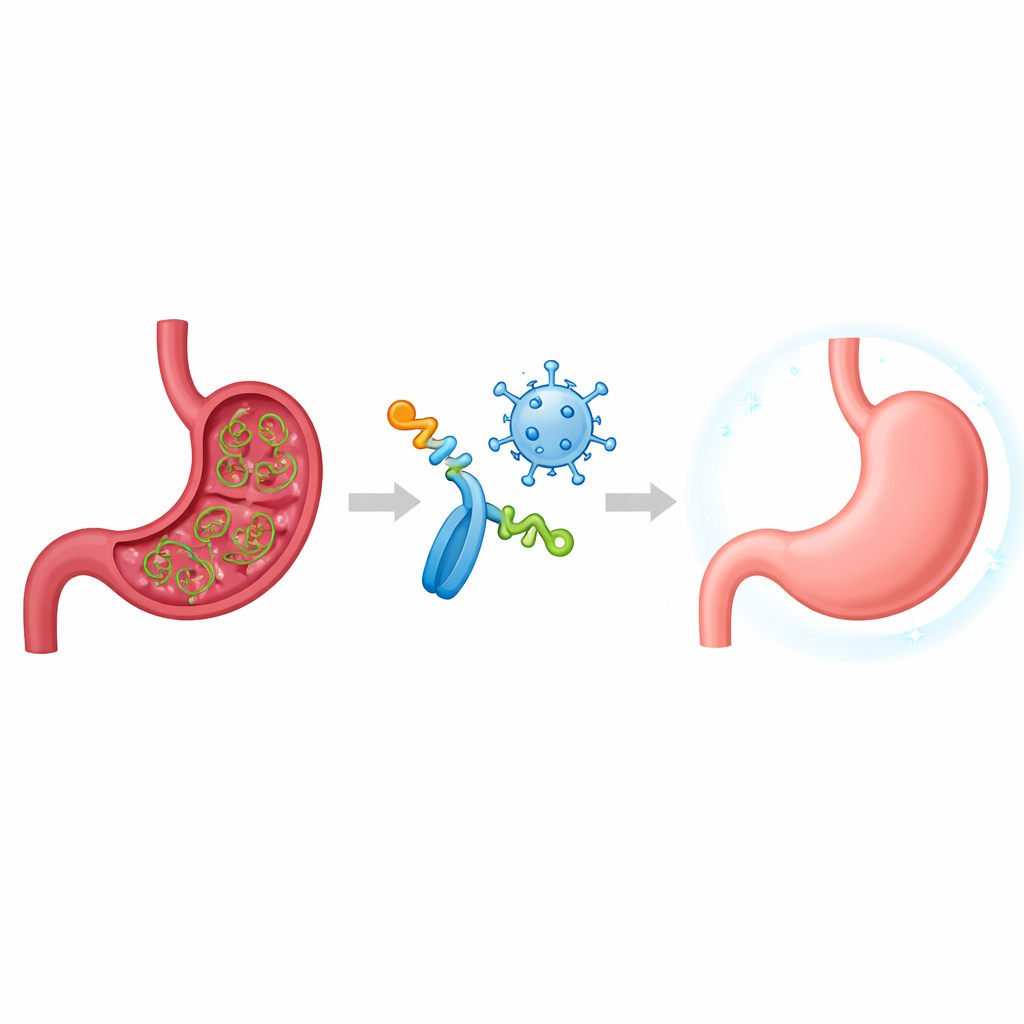
A quiet stomach invader
H. pylori is uniquely adapted to live in the harsh acid of the stomach and can linger there for decades. Many people never develop symptoms, but for others the bacterium triggers chronic inflammation, painful ulcers, and a greatly increased risk of stomach cancer. Standard antibiotic therapies are losing ground as the microbe evolves resistance, and past vaccine attempts have repeatedly fallen short. Earlier vaccines often used single bacterial proteins and could raise strong antibody levels, yet they failed to give lasting protection in clinical trials. Researchers now suspect that successful, long‑term control of H. pylori requires a coordinated response from both antibody‑producing cells and specialized T cells that direct immune attacks.
Building a smarter vaccine
Instead of injecting whole bacterial proteins, the team designed a “multi‑epitope” vaccine. Epitopes are the key snippets of a protein that the immune system actually recognizes. Using computer tools, the researchers sifted through six important H. pylori virulence factors—molecules the bacterium uses to survive in acid, move, cling to stomach cells, and evade defenses. From these they selected sixteen short regions predicted to be especially visible to B cells (which make antibodies) and T cells (which orchestrate cellular immunity). They strung these pieces together into a single artificial antigen called the Multi‑Epitope Unit, or MEU, separated by short linkers to help the immune system see each piece clearly while keeping the overall molecule stable, non‑allergenic, and soluble.
Adding a built‑in immune booster
To make sure the immune system paid attention to this engineered antigen, the researchers fused it to key parts of flagellin, a protein from another bacterium that naturally triggers an alarm receptor known as TLR5. They also built a second version of the vaccine by inserting the MEU instructions into a harmless viral vector called Modified Vaccinia Ankara (MVA), which is known for driving strong T‑cell responses. In mouse studies, the MEU was given either as a flagellin‑fused protein, as the MVA‑based vector, or in a "prime‑boost" schedule that combined both formats. Laboratory tests confirmed that the fusion protein activated the TLR5 pathway as intended, acting as its own adjuvant, and that the MVA vector efficiently produced the MEU antigen inside infected cells.
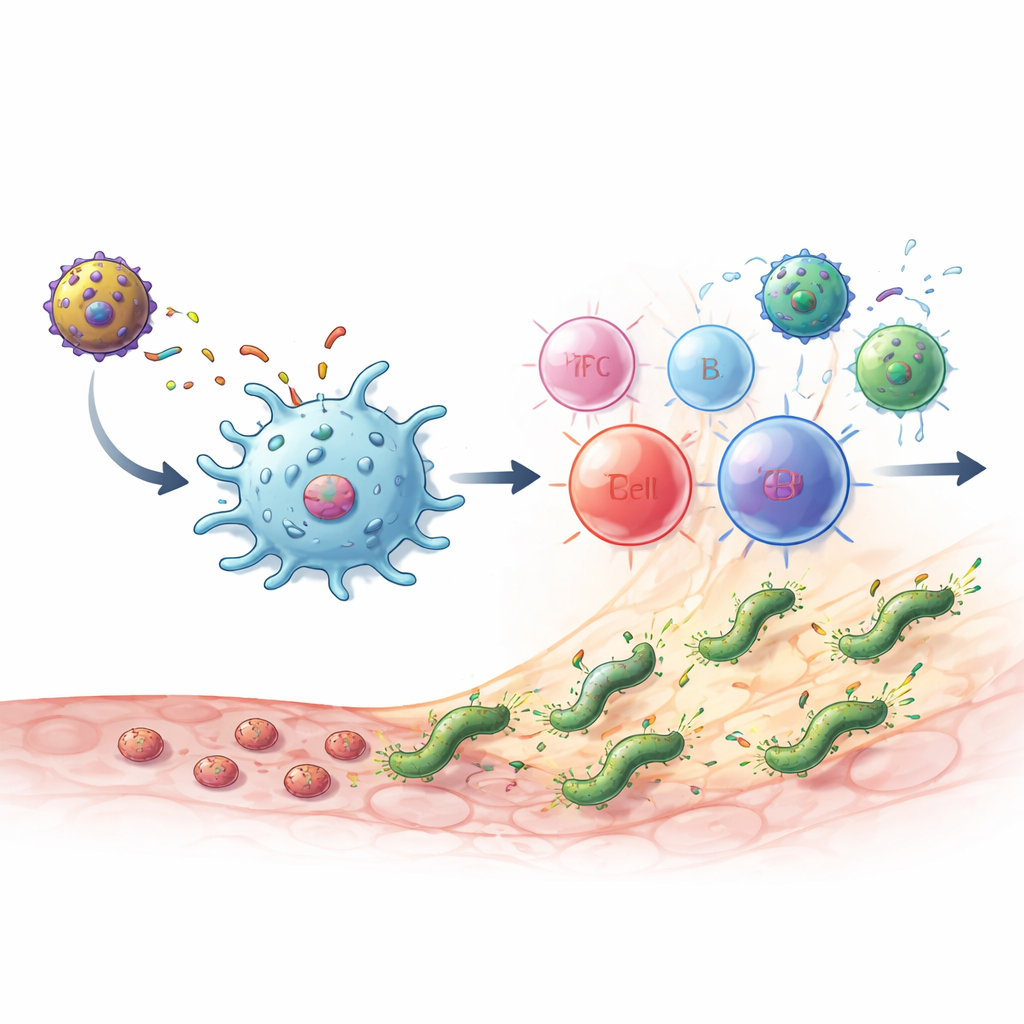
What happened in infected mice
When healthy mice received the vaccines, they generated high levels of MEU‑specific antibodies as well as CD4 T cells that produced key messenger molecules such as IL‑2 and interferon‑gamma. The response showed a healthy balance of two major helper T‑cell types: one linked to direct microbe killing and another that supports antibody production and helps limit tissue damage. Important “bridge” cells that connect innate and adaptive immunity, such as NKT‑like cells, were also expanded. The most powerful schedule was a heterologous prime‑boost: first the MEU‑flagellin protein, then the MVA‑MEU vector. In both preventive tests (vaccinating before exposure) and therapeutic tests (vaccinating after chronic infection was established), either this combination or two doses of MVA‑MEU completely cleared detectable H. pylori from the stomach, as measured by culture and genetic tests.
What this could mean for future care
To a non‑specialist, the bottom line is striking: in a demanding mouse model, this carefully engineered multi‑epitope vaccine not only blocked H. pylori from taking hold but also eliminated entrenched infections that mimic long‑term human disease. By targeting many conserved bacterial parts at once and pairing them with a strong, built‑in immune booster and a viral delivery system, the approach overcomes limitations that sank earlier, simpler vaccines. The work is now moving into safety studies in larger animals. If similar protection can be achieved in people, this strategy could one day offer a one‑two punch against ulcers, stomach cancer, and antibiotic resistance driven by this remarkably persistent germ.
Citation: Moeini, H., Mostafazadeh, A., Schoenemann, L. et al. A novel multi-epitope vaccine induces protective and therapeutic immunity against Helicobacter pylori. npj Vaccines 11, 69 (2026). https://doi.org/10.1038/s41541-026-01409-9
Keywords: Helicobacter pylori, stomach infection, multi-epitope vaccine, antibiotic resistance, gastric cancer prevention