Clear Sky Science · en
A rule-based simulation model illuminates the role of asymmetric mitochondrial fission on beta-cell health
Why tiny powerhouses matter for blood sugar
Inside each insulin-producing cell of the pancreas sit dozens of mitochondria, the cell’s miniature power plants. Their job is to turn sugar into the energy signal that tells these cells to release insulin and keep blood sugar in check. In type 2 diabetes, this system falters, and the mitochondria themselves often look broken and fragmented under the microscope. This study uses advanced computer simulations to ask a simple but important question: how does the way mitochondria split and renew themselves affect the long‑term health of these cells and their ability to release insulin?
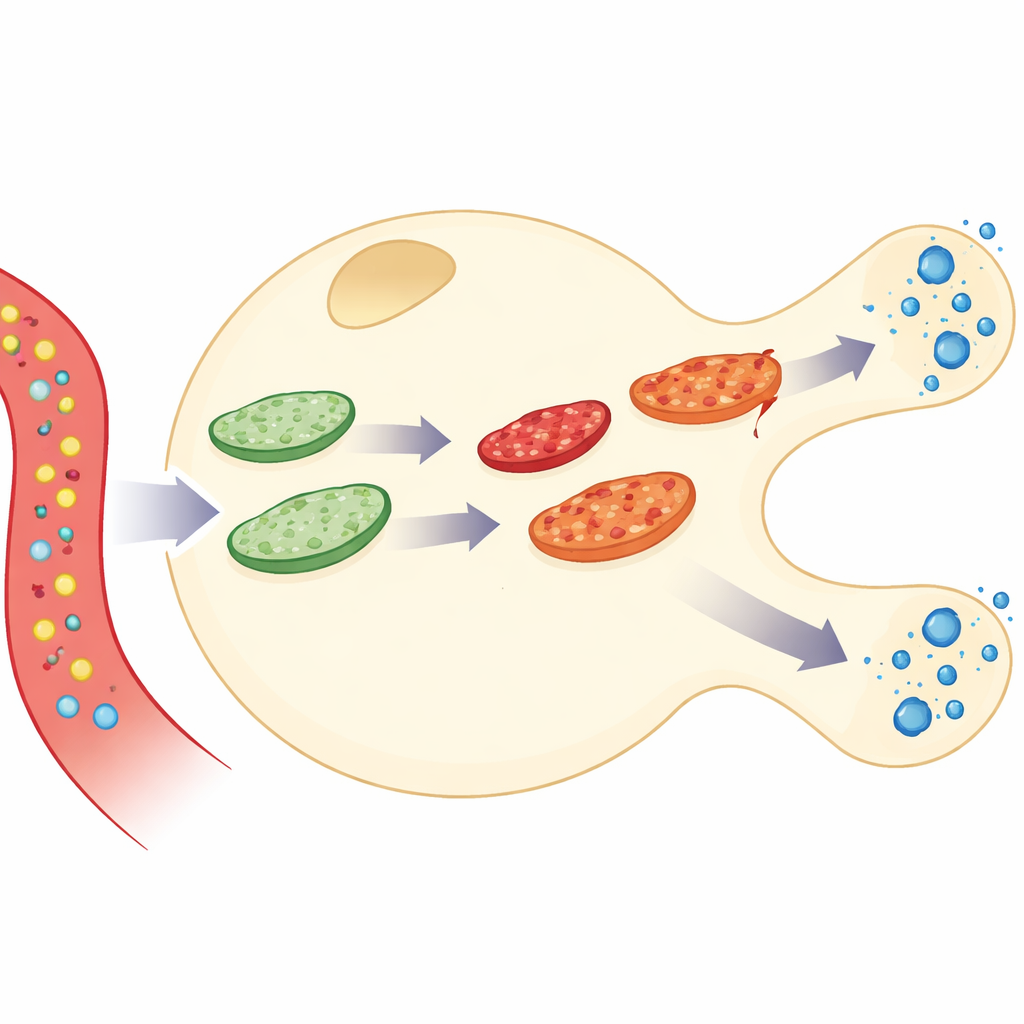
Connecting energy use to insulin release
The authors built a detailed simulation of a single pancreatic beta-cell, the cell type that makes and secretes insulin. Their model links three layers of activity: how glucose is burned to make the energy molecule ATP, how calcium inside the cell responds, and how insulin packets are released. On top of this, they added a dynamic mitochondrial “network” that constantly fuses together and splits apart. By combining these parts, the model can follow what happens as glucose enters the cell, ATP rises, calcium pulses, and insulin is secreted—while the mitochondria themselves age, accumulate damage, repair, and are removed.
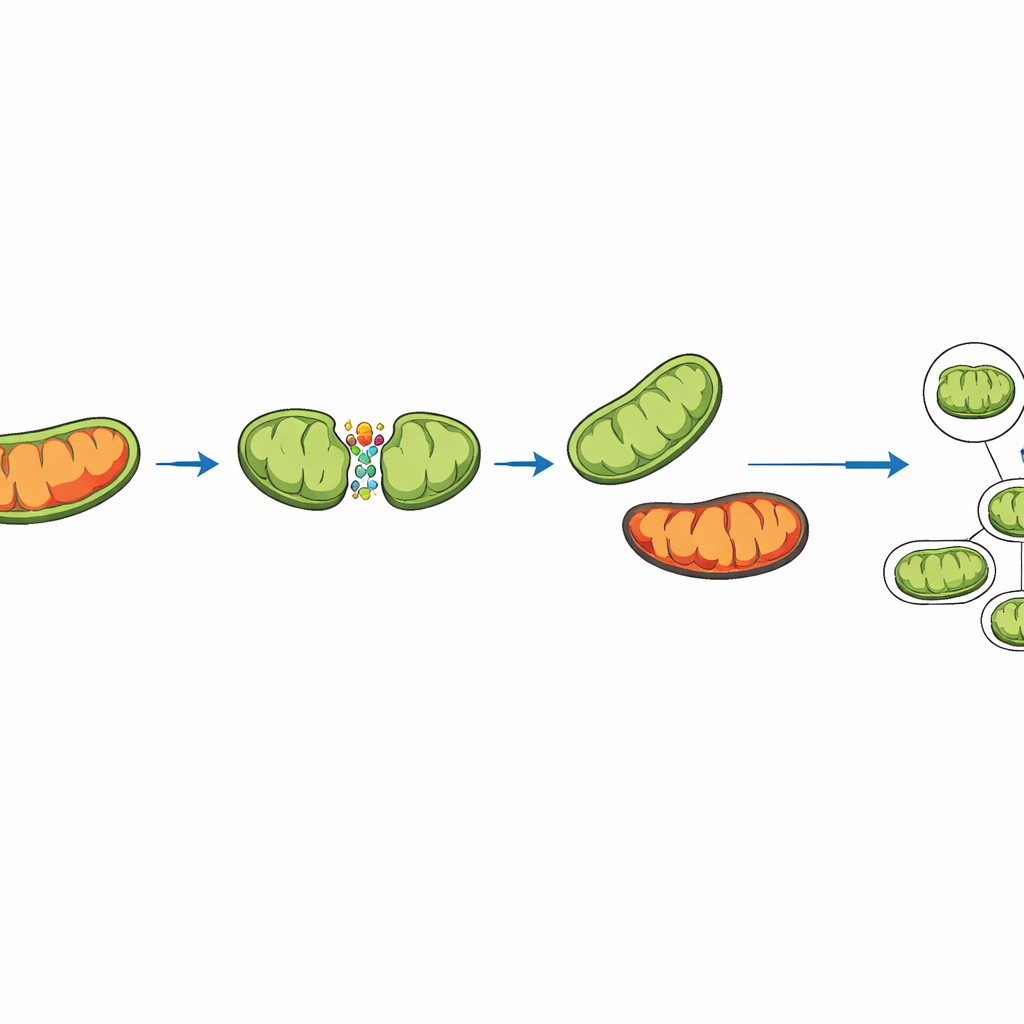
Two ways mitochondria split—and why one saves the day
Recent experiments show that mitochondria do not all divide in the same way. Sometimes they split near the middle, making two similar daughter mitochondria. Other times they divide off a small, highly damaged tip, leaving behind a larger, healthier piece. The simulation captures both types of splitting by representing key proteins that anchor the fission machinery at different spots on the mitochondrial surface. In the model, a “damage score” is tracked for each mitochondrion. When a peripheral split occurs, extra damage is deliberately shunted into the small fragment. This fragment is more likely to be swallowed and recycled by the cell, a process called mitophagy, while the larger, cleaner fragment stays in the network.
How damage sorting keeps the network young
By systematically varying how strongly damage is sorted into the small fragment and how damaged a mitochondrion can be before it is removed, the researchers explored thousands of virtual scenarios. When splitting did not sort damage, nearly all mitochondria drifted toward the same, rather unhealthy level of wear and tear. In contrast, even a modest bias that pushed just a bit more damage into the small fragment greatly lowered the average damage across the network. Repeated rounds of such asymmetric splitting and selective removal created a self-cleaning system, where healthy mitochondria remained capable of producing ATP efficiently.
When key fission proteins go missing
The team then mimicked common experimental manipulations. One anchor protein, Fis1, mainly promotes the peripheral fission events that concentrate damage into small fragments. Reducing Fis1 in the model led to fewer peripheral splits, more uniformly damaged mitochondria, and a breakdown of the network’s health. Another anchor, MFF, favors midzone splits that produce similar daughters. Lowering MFF caused only mild changes in damage levels but produced larger, more fused mitochondria, matching lab observations that this pathway is more about preparing cells to divide than clearing damage. When the core fission protein Drp1 was reduced, splitting nearly stopped altogether. Damage then rose close to the maximum possible, leaving the network severely compromised.
From tired mitochondria to poor insulin release
Because the model links mitochondrial health to ATP output, it can predict how these structural changes impact insulin secretion. With moderate loss of Fis1, the cell still released insulin at low glucose, but its response to high glucose was blunted—similar to beta-cells from people with early metabolic disease. With strong Drp1 loss, the cell became almost unresponsive to glucose: insulin release stayed near a low basal level even when sugar was high. These patterns resemble reported experimental results and suggest that balanced expression of the fission proteins is essential for keeping beta-cells both energized and responsive.
What this means for understanding diabetes
To a layperson, the study’s message is that how mitochondria break apart is just as important as how they make energy. A carefully tuned “good break” trims away damaged pieces and keeps the network young, supporting strong insulin release. When that trimming mechanism is weakened or shut down, damaged mitochondria pile up, ATP production falters, and beta-cells struggle to respond to rising blood sugar. By capturing these links in a flexible computer model, the work offers a test bed for future ideas about how to protect or restore mitochondrial quality in diabetes, potentially guiding new treatments that help beta-cells stay healthy longer.
Citation: Henning, P., Schultz, J., Baltrusch, S. et al. A rule-based simulation model illuminates the role of asymmetric mitochondrial fission on beta-cell health. npj Syst Biol Appl 12, 64 (2026). https://doi.org/10.1038/s41540-026-00732-0
Keywords: mitochondria, insulin secretion, beta-cells, type 2 diabetes, computer modeling