Clear Sky Science · en
Cortical and basal ganglia beta oscillations and frequency-dependent DBS effects in the A53T Parkinson’s disease rat model
Why this brain rhythm study matters
Parkinson’s disease is best known for tremors and slowed movement, but deep inside the brain it is also marked by abnormal “beta” rhythms, a kind of electrical background beat. Surgeons can ease symptoms with deep brain stimulation, a pacemaker-like therapy that delivers pulses to specific brain regions. Yet it has remained unclear how much of this benefit comes from quieting those beta rhythms and which stimulation patterns work best. This study uses a rat model that closely mimics human Parkinson’s disease to watch those rhythms emerge and test how different kinds of stimulation shape them, offering clues that may help refine future brain therapies.
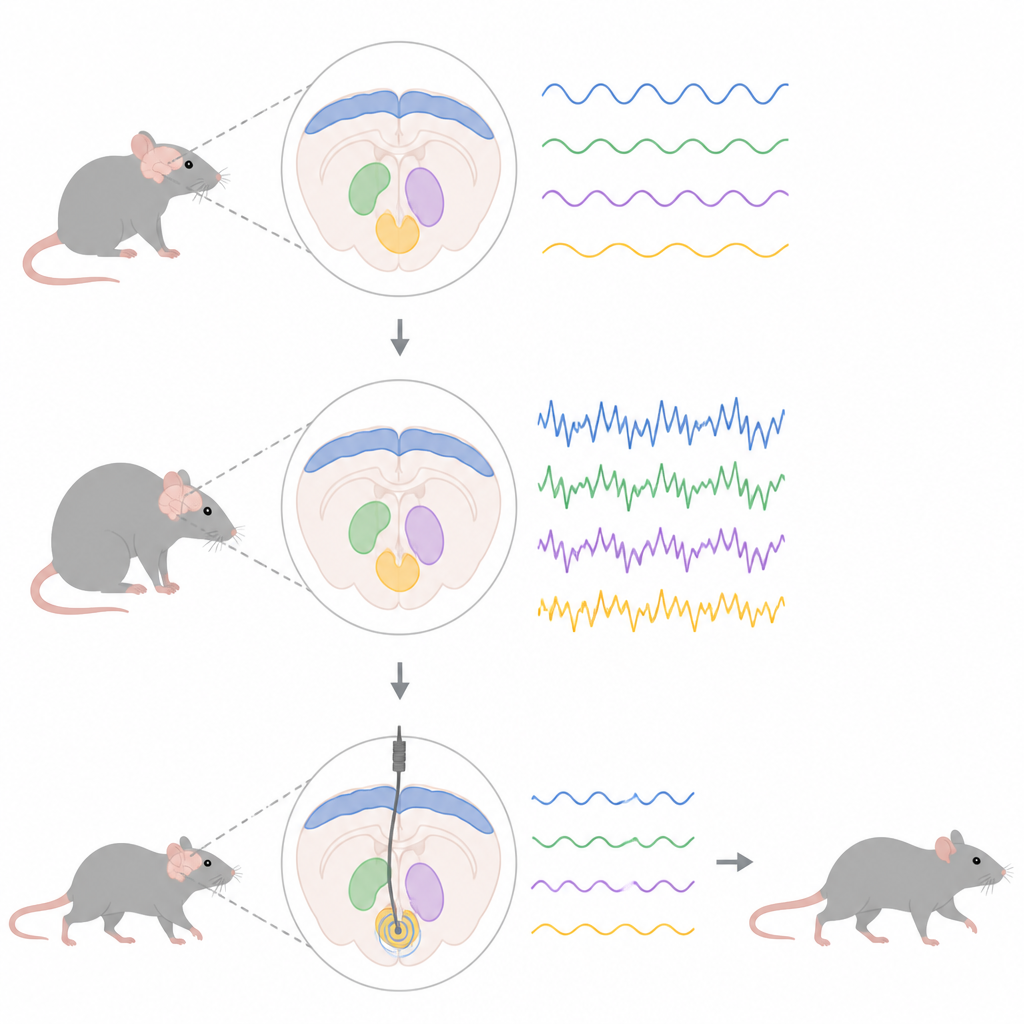
Following the brain’s hidden beat
The researchers used a genetic trick to make rats overproduce a mutant form of alpha-synuclein, the same protein that builds up in many people with Parkinson’s disease. This gradually killed off dopamine-producing cells in the midbrain, setting off changes in a larger movement network that links the outer cortex to deep structures called the subthalamic nucleus and entopeduncular nucleus, the rat equivalent of a key human output hub. By placing fine electrodes in these areas, the team tracked local field potentials, the combined electrical activity of many nerve cells, focusing on activity in the beta range between 13 and 30 cycles per second.
Stepwise spread of unhealthy rhythms
Recordings under light anesthesia revealed that abnormal beta activity did not appear everywhere at once. Early in the disease process, when dopamine loss was still modest, extra beta power was already visible in the motor cortex, the outer layer that helps plan and start movements. Only later, as more dopamine cells died, did strong beta rhythms emerge in the subthalamic and entopeduncular nuclei. In rats with the disease protein, nerve cells in these deep regions also changed how they fired: cells in the entopeduncular nucleus began firing faster and in bursts, while subthalamic cells developed more irregular, bursty patterns at later stages, even without a big change in their average firing rate.
Bursts that block smooth movement
To see how these signals behave during natural behavior, the team repeated the recordings in awake rats moving freely. Instead of a steady hum, beta activity appeared as brief bursts. Rats with the Parkinson-like changes showed bursts that were longer, happened more often, and together occupied more time than in control animals. In the subthalamic nucleus, stronger beta activity went hand-in-hand with less movement, echoing observations in people with Parkinson’s disease where beta bursts are linked to difficulty starting and controlling actions. These patterns suggest that it is the timing and shape of the bursts, not just overall beta strength, that matters for how the disease disrupts movement.
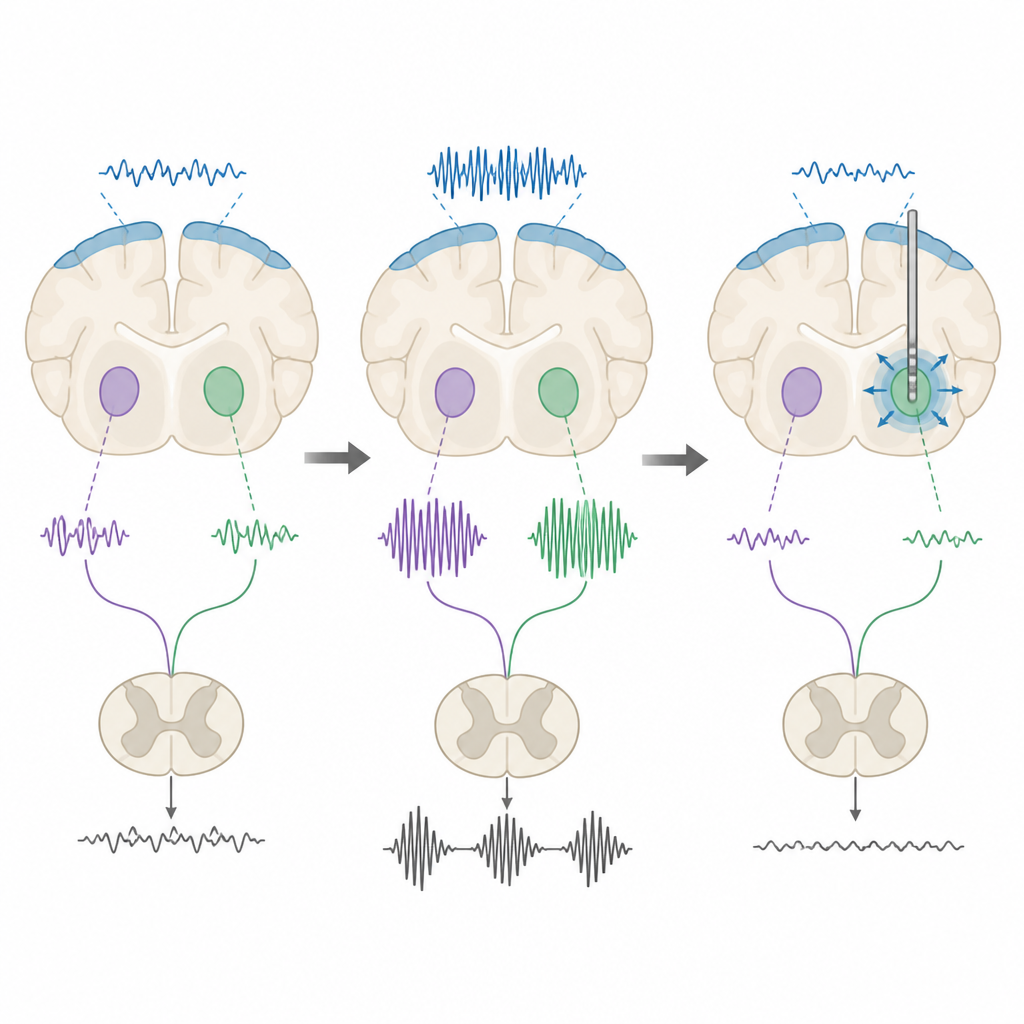
Testing brain stimulation patterns
The researchers then tested whether deep brain stimulation could tune these rhythms. They applied both low-frequency and high-frequency electrical pulses to the subthalamic and entopeduncular nuclei. Under anesthesia, both kinds of stimulation reduced overall beta power in rats with the disease changes, but not in healthy controls, showing that the abnormal rhythms are especially sensitive to stimulation. In awake rats, a more nuanced picture emerged. High-frequency stimulation tended to shorten beta bursts or reduce the time the signal spent in a high-beta state, while low-frequency stimulation often had the opposite effect, lengthening bursts or increasing their total duration. When the team measured movement on a grid-walking test, only high-frequency stimulation of the entopeduncular nucleus improved the rats’ stepping accuracy.
What this means for future treatment
Taken together, the findings show that this rat model reliably develops the same kind of beta bursts and firing irregularities seen in people with Parkinson’s disease and responds to deep brain stimulation in a frequency-dependent way. High-frequency pulses appear better suited than low-frequency ones to breaking up long, disruptive beta bursts and supporting smoother movement. By charting how abnormal rhythms arise across the movement network and how different stimulation settings reshape them, this work provides a powerful testing ground for future strategies that aim not just to silence brain regions, but to retune their electrical timing for better control of Parkinson’s symptoms.
Citation: Kondrataviciute, L., Kapadia, M., Skelin, I. et al. Cortical and basal ganglia beta oscillations and frequency-dependent DBS effects in the A53T Parkinson’s disease rat model. npj Parkinsons Dis. 12, 113 (2026). https://doi.org/10.1038/s41531-026-01304-z
Keywords: Parkinson’s disease, beta oscillations, deep brain stimulation, basal ganglia, rat model