Clear Sky Science · en
Functional signatures of de novo GABBR1 and GABBR2 variants associated with neurodevelopmental disorders
Why tiny changes in brain chemistry matter
Many children with autism, learning difficulties, or movement and attention problems never receive a clear biological explanation for their symptoms. This study looks at one possible cause: rare genetic changes that subtly distort how brain cells respond to a calming chemical called GABA. By tracing these changes from DNA to individual receptors on neurons, the researchers show how small molecular glitches can tip the balance of brain activity and point the way toward more precise, personalized treatments.
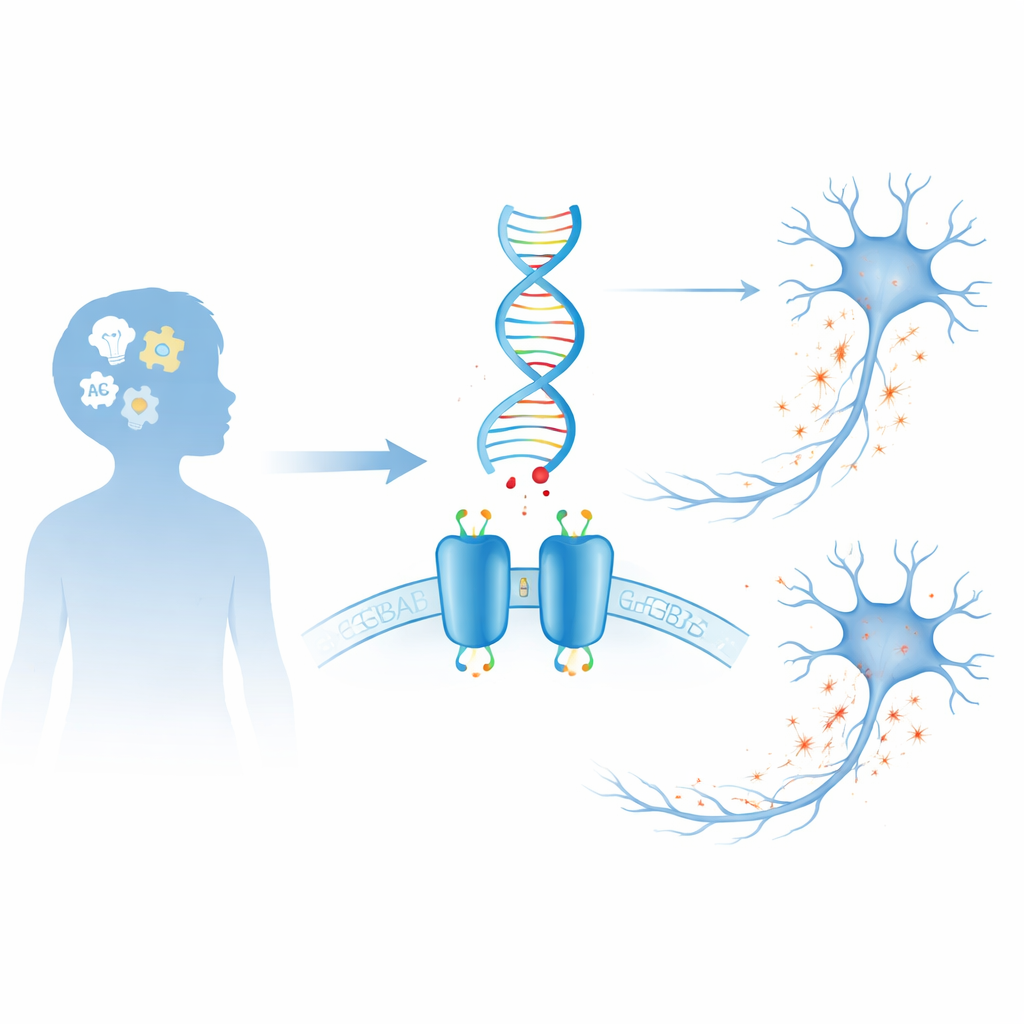
The brain’s braking system
Our brains rely on a constant tug-of-war between signals that excite neurons and signals that quiet them down. GABA is the brain’s main “brake” chemical. One of the key devices it uses is the GABAB receptor, a two-part protein that sits on the surface of neurons and some support cells. When GABA binds, the receptor triggers internal changes that lower cellular activity, helping to prevent overload, shape brain rhythms, and fine-tune communication between cells. Because this receptor is so central to controlling excitability, even slight changes in its structure can have wide-ranging effects on brain function and behavior.
Seven rare genetic changes under the microscope
The researchers focused on seven rare, spontaneous DNA changes—called de novo variants—in the genes that encode the two halves of the GABAB receptor, GABBR1 and GABBR2. Each variant was found in a child with neurodevelopmental problems such as delayed speech and motor skills, intellectual disability, autism spectrum features, or attention difficulties; some also had seizures or movement abnormalities. Computer algorithms all suggested that these variants were likely harmful, but such predictions cannot tell whether a receptor becomes overactive, underactive, or mislocalized. To answer that, the team recreated each variant in human cells in the lab and measured how the altered receptors behaved.
When the brain’s brake sticks or slips
Experiments revealed that the seven variants did not all act in the same way. Some weakened the receptor’s response to GABA: much more of the chemical was needed to switch the receptor fully on, or fewer receptors reached the cell surface at all. These changes effectively reduced the available braking power in the brain. Other variants had almost the opposite effect, leaving the receptor partially switched on even when no GABA was present. This constant low-level activity boosted baseline signaling but paradoxically left less room for further response when GABA levels rose, blunting the impact of real inhibitory signals during intense brain activity.
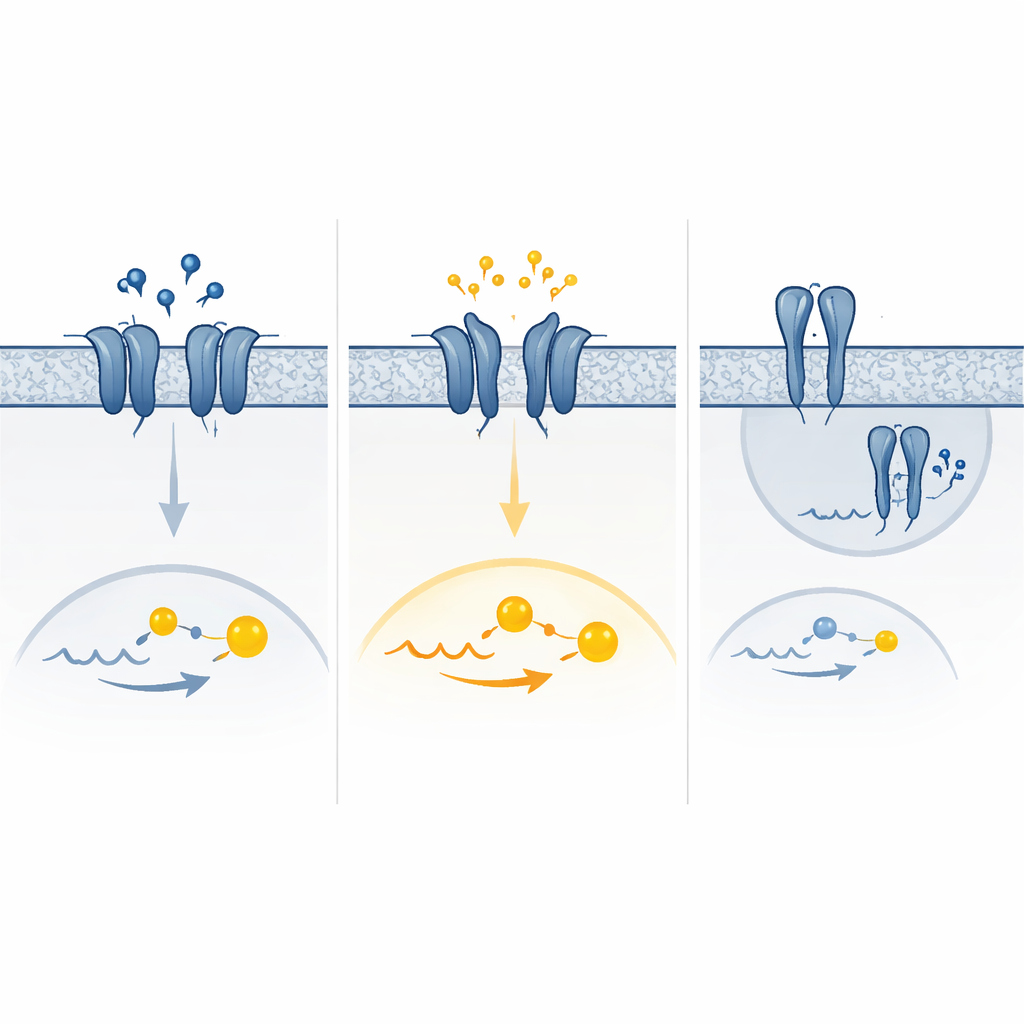
A mix of too much and too little signal
Computer simulations of receptor motion helped explain one striking variant that showed strong baseline activity and virtually no response to GABA. In this case, part of the receptor’s outer “clamp” domain tended to close on its own and stay in an active-like shape, mimicking what normally happens only when GABA is bound. Other variants appeared to favor active arrangements of the receptor’s membrane-spanning segments or altered how receptor pairs assembled and reached the cell surface. The end result was a spectrum of effects—pure loss of function, pure gain of function, or a combination of both depending on GABA levels and how cells adapt over time by removing or downregulating overactive receptors.
Implications for personalized treatment
Although the children shared overlapping symptoms, the underlying receptor problems were quite different. This finding matters for therapy. Variants that reduce receptor number or sensitivity might be helped by drugs that boost the effect of GABA on the remaining normal receptors. In contrast, variants that make receptors too active could respond very differently, and simply blocking the receptor might worsen certain aspects of signaling. The study shows that relying on computer predictions alone is not enough: direct functional tests are crucial to distinguish between these scenarios. By mapping exactly how each GABAB receptor variant changes signaling, this work lays groundwork for more tailored treatment strategies in neurodevelopmental disorders where the brain’s braking system goes awry.
Citation: Stawarski, M., Bielopolski, N., Roitman, I. et al. Functional signatures of de novo GABBR1 and GABBR2 variants associated with neurodevelopmental disorders. npj Genom. Med. 11, 23 (2026). https://doi.org/10.1038/s41525-026-00558-z
Keywords: GABAB receptor, neurodevelopmental disorders, genetic variants, autism spectrum, precision medicine