Clear Sky Science · en
In vivo single-cell RNA metabolic labeling resolves early transcriptional responders in the regenerating zebrafish heart
Why fixing a broken heart is so hard
When a person has a heart attack, the damaged muscle is usually replaced by scar tissue that cannot beat, leaving the heart permanently weakened. Yet some animals, like zebrafish, can regrow healthy heart muscle after injury. This article explores how scientists are beginning to watch, in real time and cell by cell, the very first genetic switches that flip on in a zebrafish heart after damage—and how tweaking an early immune reaction can change the course of healing.
Watching fresh genetic messages as they appear
Every cell in the heart constantly turns genes on and off, producing short-lived RNA messages that tell the cell what to do. Traditional methods read out all RNA in bulk, mixing old and new signals together. The researchers adapted a chemical “timestamp” method, called RNA metabolic labeling, for use in live adult zebrafish. They injected a harmless building block that gets incorporated only into newly made RNA. Later, a specific chemical treatment turns those new pieces into a form that shows up as a distinct signature when the RNA is sequenced. By combining this with single-cell analysis, they could separate, in each individual heart cell, which gene messages were present before injury and which were freshly made afterwards.
Zooming in on the first hours after damage
The team focused on the crucial first six hours after freezing and damaging a portion of the zebrafish heart, a procedure that mimics a heart attack. Because many long‑lived RNA molecules were already present before the injury, simply measuring total RNA would blur early changes. With their labeling approach, the scientists compared newly made RNA in injured hearts to that in sham-operated controls, cell type by cell type. This allowed them to detect even subtle and short-lived bursts of gene activity that would otherwise be drowned out by older messages or by differences between individual animals.
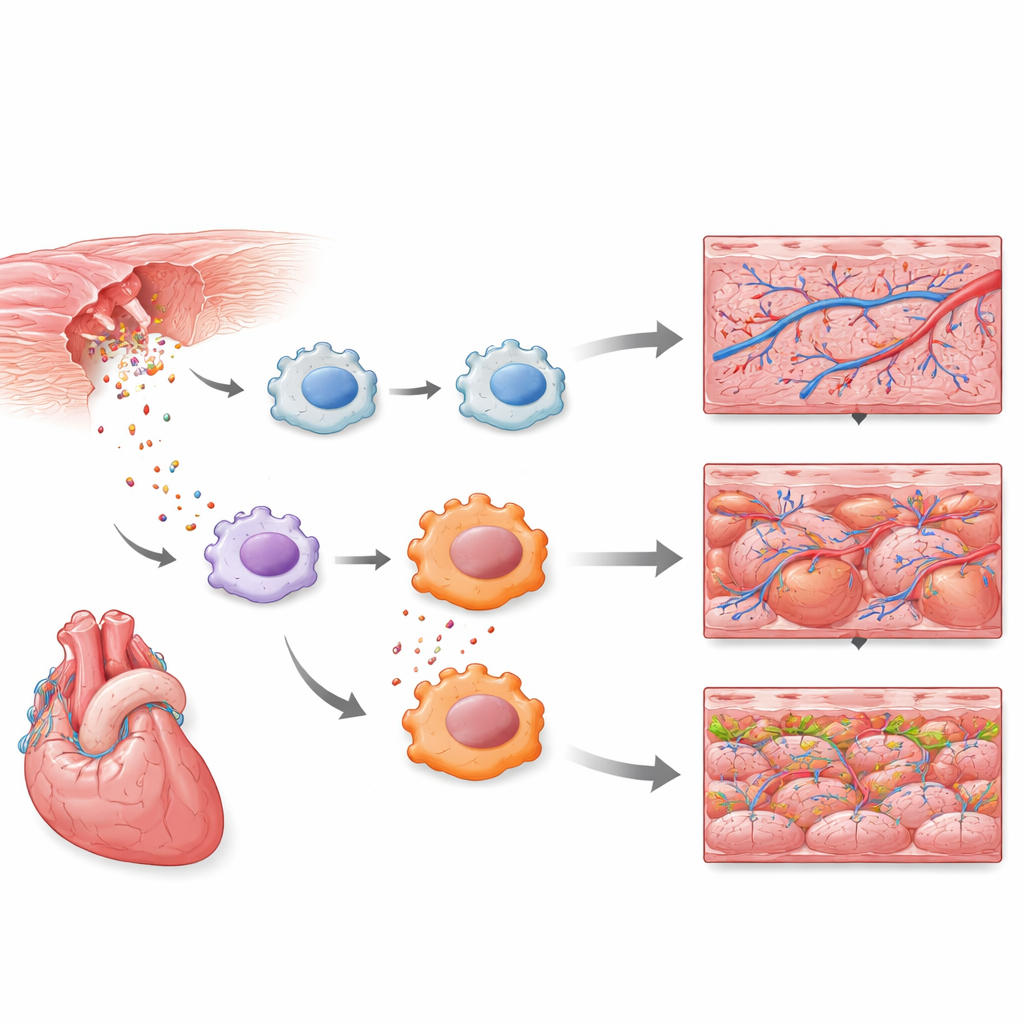
Early immune cells step into the spotlight
The single-cell measurements revealed that a subset of immune cells resembling macrophages were among the very earliest responders. Within hours, these cells switched on innate defense programs, including Toll-like receptor signaling, a well-known alarm pathway that senses danger and infection. A larger reference map of heart cells over many days of regeneration confirmed that neutrophils, another kind of white blood cell, also mount a strong early response. Together, these results show that the first wave of heart repair is driven not just by structural cells like muscle and blood vessels, but by a precisely timed burst of inflammatory activity in specific immune cell types.
Fine-tuning inflammation to aid repair
Inflammation is a double-edged sword: too little can delay clean-up of dead tissue, while too much or too long can cause extra damage and scarring. Guided by their single-cell data, the researchers tested what happens if they selectively dial down one key immune signal in macrophages. They created zebrafish in which a dominant-negative version of MyD88—an essential relay in Toll-like receptor pathways—could be switched on only in macrophages after treatment. When this brake was engaged, fewer strongly inflammatory macrophages accumulated at the injury site. Strikingly, damaged hearts in these fish showed faster early hallmarks of regeneration: more growth of coronary blood vessels around the wound and increased division of heart muscle cells, without obvious harm to other steps of healing.
What this means for future heart repair
This work shows that it is now possible to watch, within hours and at single-cell resolution, which genes switch on in response to heart damage in a living animal. The findings highlight that early activation of innate immune cells is not simply an on-or-off phenomenon, but a tunable process whose strength and timing can speed up or slow down regeneration. While zebrafish hearts are far more regenerative than human hearts, the principle may carry over: carefully adjusting specific immune signals—rather than blocking inflammation outright—could one day help coax injured mammalian hearts toward better recovery.
Citation: Mintcheva, J., Tseng, TL., Goumenaki, P. et al. In vivo single-cell RNA metabolic labeling resolves early transcriptional responders in the regenerating zebrafish heart. Nat Commun 17, 4073 (2026). https://doi.org/10.1038/s41467-026-72781-2
Keywords: heart regeneration, zebrafish, single-cell RNA, immune response, Toll-like receptor signaling