Clear Sky Science · en
A tissue-intrinsic mechanism sensitizes HIV-1 particles for TLR-triggered innate immune responses
How Our Tissues Quietly Fight HIV
HIV usually makes headlines for how it disables the immune system, but this study flips the story: it asks what our tissues themselves do to push back. Deep inside lymph nodes and other organs, immune cells and viruses move through a three‑dimensional mesh of collagen and other proteins. This work shows that this mesh is not just passive scaffolding. Instead, it actively weakens HIV’s ability to infect cells while, at the same time, making the virus more visible to our innate immune defenses.
The Hidden World Between Cells
Inside our bodies, cells are surrounded by an intricate web called the extracellular matrix, built largely from collagen fibers. The researchers created simplified “tissue‑like” gels made of different types of collagen to mimic this environment. They then let HIV particles move through these three‑dimensional gels and compared them with virus kept in ordinary liquid cultures. They found that brief, physical contact with collagen fibers was enough to sharply reduce the virus’s ability to infect new cells, a phenomenon they call extracellular restriction of virion infectivity. This effect appeared quickly, depended on direct contact with the matrix, and was seen across different HIV strains and several collagen‑rich materials, but not in non‑adhesive gels like agarose.
How Collagen Tames the Virus
One obvious worry was that collagen might simply destroy the virus particles. Using high‑resolution electron microscopy, the team showed that this was not the case: HIV particles emerging from collagen gels looked structurally normal, with intact membranes and cores. Nor did collagen strip off the virus’s surface proteins. Instead, the key change was functional. Collagen‑exposed viruses still bound to target cells, but they fused with them much less efficiently, meaning that entry into the cell was blocked at a very early step. Artificial “infectivity‑boosting” nanofibers that help viruses fuse with cells were able to restore the infectiousness of collagen‑treated HIV, reinforcing the idea that the matrix mainly interferes with the fusion process rather than damaging the virus as a whole.
From Silent Particles to Immune Alarms
The most surprising finding was that collagen contact did not just blunt infection; it also turned HIV into a stronger alarm signal for innate immune cells, especially monocyte‑derived macrophages. When these cells were exposed to collagen‑experienced HIV, they produced far more inflammatory molecules such as IL‑6, IL‑8, and TNF than when exposed to ordinary virus, even though actual infection levels were only modestly reduced. Detailed gene‑expression profiling showed that macrophages triggered by collagen‑primed virus switched on a wide antiviral program, including many known immune restriction factors and warning signals that help coordinate broader immune responses.
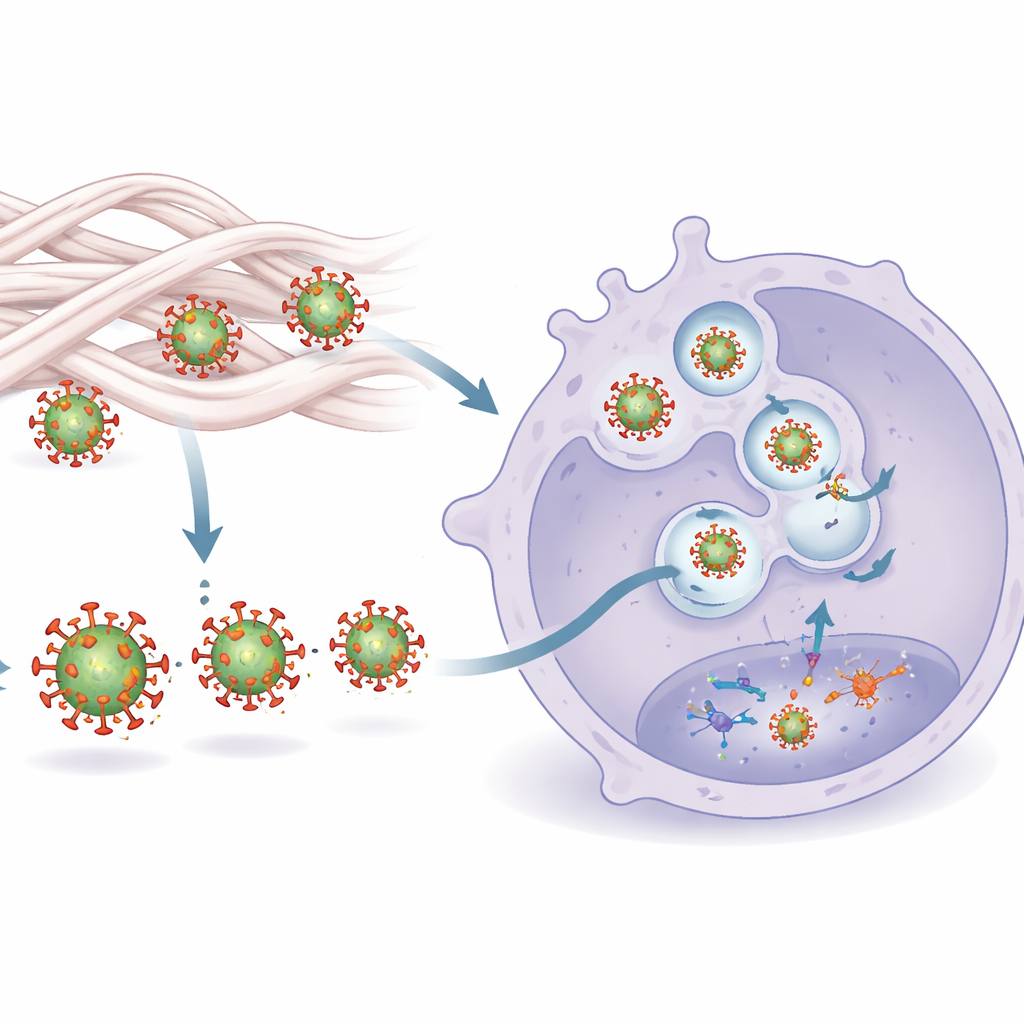
How Sensing Is Switched On
The study traced this heightened sensing to a two‑part mechanism. First, contact with collagen subtly reshapes HIV’s outer coat protein, Env. These shape shifts make Env easier for a sensor on macrophages, Toll‑like receptor 2 (TLR2), to grab. Second, once recognized at the surface, the virus is shuttled more efficiently into internal compartments that contain another sensor, TLR8, which detects the virus’s RNA genome. Blocking either TLR2 or TLR8 shut down the collagen‑induced cytokine surge, showing that both signals are needed. Microscopy confirmed that collagen‑treated viruses bound more strongly to a soluble version of TLR2 and were more likely to end up in TLR8‑rich endosomes, where they could trigger a robust antiviral cascade.
Why This Matters for People Living With HIV
For a layperson, the core message is that our tissues are not neutral ground during HIV infection. The very fibers that give organs their structure can directly handicap HIV’s ability to spread and, at the same time, help immune cells recognize the virus more clearly. This “tissue‑intrinsic” defense acts outside cells, complementing the better‑known antiviral proteins inside them. The work also hints at a double‑edged sword: the same inflammatory signals that help control virus may, over time, contribute to tissue scarring and chronic inflammation seen in people living with HIV. Understanding how the extracellular matrix tunes this balance could open new ways to boost protective immunity while limiting long‑term damage.
Citation: Sid Ahmed, S., Zimmermann, L., Imle, A. et al. A tissue-intrinsic mechanism sensitizes HIV-1 particles for TLR-triggered innate immune responses. Nat Commun 17, 4209 (2026). https://doi.org/10.1038/s41467-026-72586-3
Keywords: HIV innate immunity, extracellular matrix, collagen and virus, toll-like receptors, tissue antiviral defense