Clear Sky Science · en
Mechanism and mitigation of stainless steel dissolution in LiFSI-based lithium-ion battery electrolytes
Why this research matters for future batteries
Lithium-ion batteries power everything from laptops to electric cars, and manufacturers are pushing them to store more energy and charge faster. To reach those goals, new electrolyte recipes and high-energy electrodes are being adopted. One promising electrolyte ingredient, called LiFSI, improves stability and performance, but it comes with a hidden drawback: it can slowly corrode stainless steel parts inside the cell. This paper uncovers exactly how that damage happens and presents a practical way to stop it, paving the way for safer, longer-lasting high-energy batteries.
Hidden damage inside the battery shell
Many commercial battery formats, including cylindrical and coin cells, use stainless steel for housings and internal components. Under normal conditions, stainless steel protects itself with a thin, stable oxide film. The authors show that in LiFSI-based electrolytes this protection can fail, especially at the higher voltages used with modern nickel-rich cathodes. Tiny amounts of chloride, often left over from salt production, and the FSI anion from LiFSI work together to attack the steel. Chloride starts the process by creating microscopic pits in the surface. Once these pits form, iron from the stainless steel dissolves, and FSI-based species help keep that iron in solution instead of letting it re-form a protective oxide, so the metal continues to dissolve and the pits grow deeper.
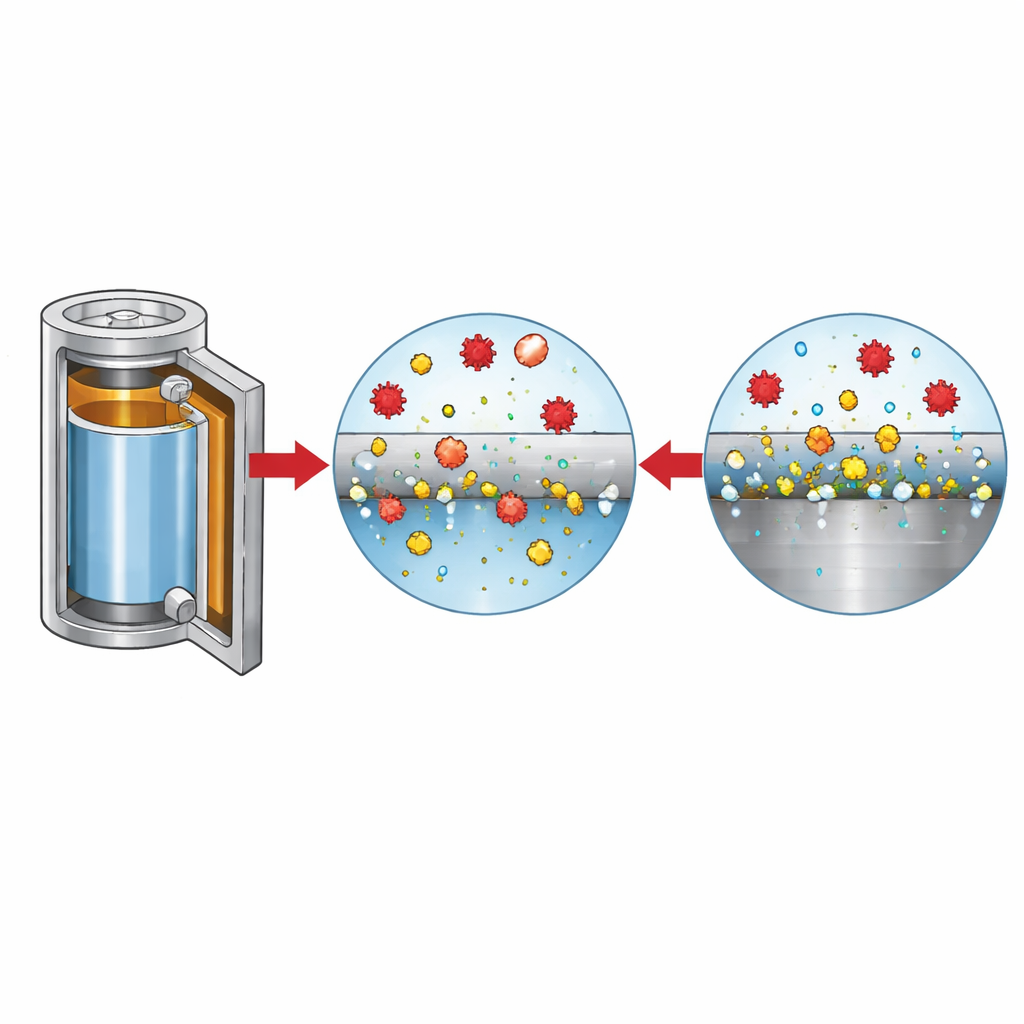
How aggressive ions eat away the metal
To disentangle the roles of the different ions, the researchers systematically varied the chloride level and salt composition, while monitoring current, surface morphology, and gas evolution. At low chloride levels, the steel surface passivates again after an initial disturbance, but once chloride exceeds about a few tens of parts per million, the pits no longer heal and dissolution continues. Imaging and chemical analysis reveal that in chloride-rich solutions the surface becomes dotted with iron oxides and pits, whereas in pure LiFSI electrolytes the pits are fewer but much deeper, with dissolved iron forming soluble complexes instead of precipitating. These dissolved metal ions migrate through the cell and deposit on the negative electrode, roughening the lithium surface and contributing to gas formation such as hydrogen and carbon dioxide—both undesirable for battery safety and lifetime.
A protective helper salt that reshapes the interface
The team then explored whether adding a second salt, LiDFOB, could protect the steel without sacrificing the benefits of LiFSI. Electrochemical tests showed that even modest amounts of LiDFOB pushed the onset of stainless steel dissolution to higher voltages and drastically reduced the corrosion current. Microscopy confirmed that pits nearly disappeared when LiDFOB was present. Surface-sensitive spectroscopy revealed that, in LiFSI-only electrolytes, oxidized iron and chromium species combine with FSI-related fragments, consistent with ongoing dissolution. In contrast, LiDFOB-containing electrolytes left more metallic iron and chromium at the surface and introduced boron- and fluorine-rich compounds linked to LiDFOB breakdown.
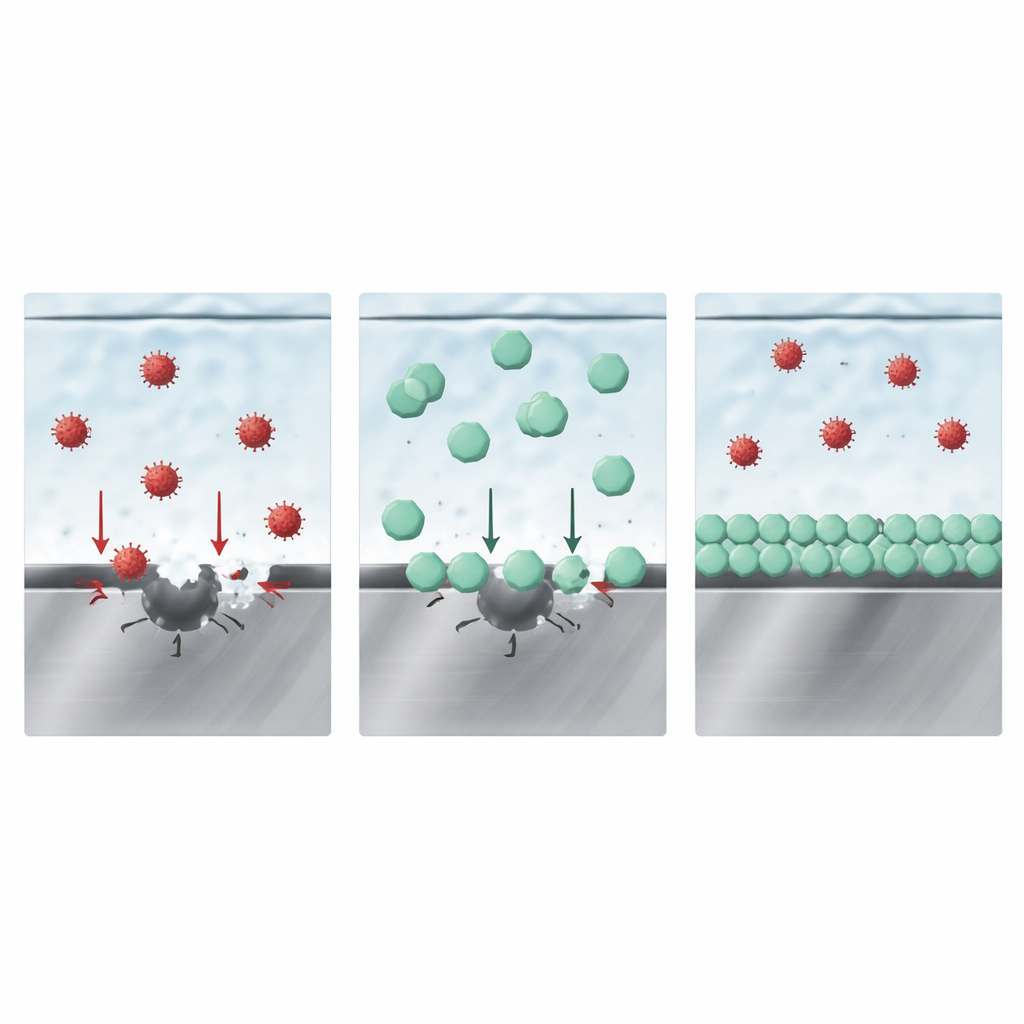
How the protective layer wins the competition
At first glance, it might seem that a solid boron-containing film alone shields the steel. However, additional immersion experiments and modeling suggested a subtler picture. The authors propose that the key is competition between different anions for space directly at the metal surface. Measurements of the surface charge behavior, along with computer simulations, show that the anion from LiDFOB binds more strongly to the iron oxide surface than either chloride or FSI. In practical terms, LiDFOB’s anions crowd into the innermost layer of the liquid at the steel interface, occupying the very spots where the aggressive ions would otherwise initiate attack. By blocking access, they suppress both the initial chloride-driven pitting and the FSI-driven deep dissolution that follows, which in turn reduces gas evolution and metal deposition elsewhere in the cell.
From mechanism to better-performing batteries
To test whether this chemical protection translates into real-world benefits, the researchers cycled graphite||NMC811 and silicon-graphite||NMC811 cells using different electrolytes and stainless steel grades. Cells with LiFSI alone failed early due to metal dissolution, while those using mixed LiFSI–LiDFOB electrolytes showed greatly extended lifetimes. When more corrosion-resistant stainless steel (SUS316L) was combined with the optimized dual-salt electrolyte, silicon-graphite cells reached around 300 cycles to 80% remaining capacity. Pouch cells without stainless steel hardware but using the same electrolyte also lasted roughly twice as long as with a conventional LiPF6-based formulation, highlighting the broader stabilizing effect of LiDFOB on battery interfaces.
What this means for everyday technology
In accessible terms, the study shows that a promising high-performance salt, LiFSI, can quietly corrode the steel parts that hold a battery together—especially when trace chloride is present. The authors reveal that chloride opens tiny holes in the protective film, and LiFSI then prevents those holes from healing by keeping dissolved iron in the liquid, so the damage spreads. By adding LiDFOB, whose ions preferentially line up on the steel surface and keep the harmful ions at bay, the corrosion is largely stopped, gas generation is reduced, and both coin and pouch cells last far longer. This combination of a powerful but corrosive salt with an intelligently chosen “bodyguard” salt offers a practical recipe for safer, longer-lived, high-energy lithium-ion batteries that could find their way into future electric vehicles and large-format cells.
Citation: Yan, P., Stan, M.C., Zhour, K. et al. Mechanism and mitigation of stainless steel dissolution in LiFSI-based lithium-ion battery electrolytes. Nat Commun 17, 3866 (2026). https://doi.org/10.1038/s41467-026-72530-5
Keywords: lithium-ion batteries, electrolyte additives, stainless steel corrosion, LiFSI, LiDFOB