Clear Sky Science · en
Unveiling a flip-over retention mechanism in the gas-phase Cl− + (CH3)3CI SN2 reaction
Why tiny collisions matter
Every second, countless chemical reactions shape the world around us, from how medicines work to how fuels burn. Many of these reactions follow a handful of textbook patterns that students learn in introductory chemistry. This paper takes a very familiar type of reaction—where one small charged particle swaps places with another in an organic molecule—and shows that reality is more surprising than the standard picture. By watching single molecules collide in the gas phase and simulating their motions in detail, the authors discover a new way that atoms can rearrange, one that had not been seen before.
Two rival reaction paths
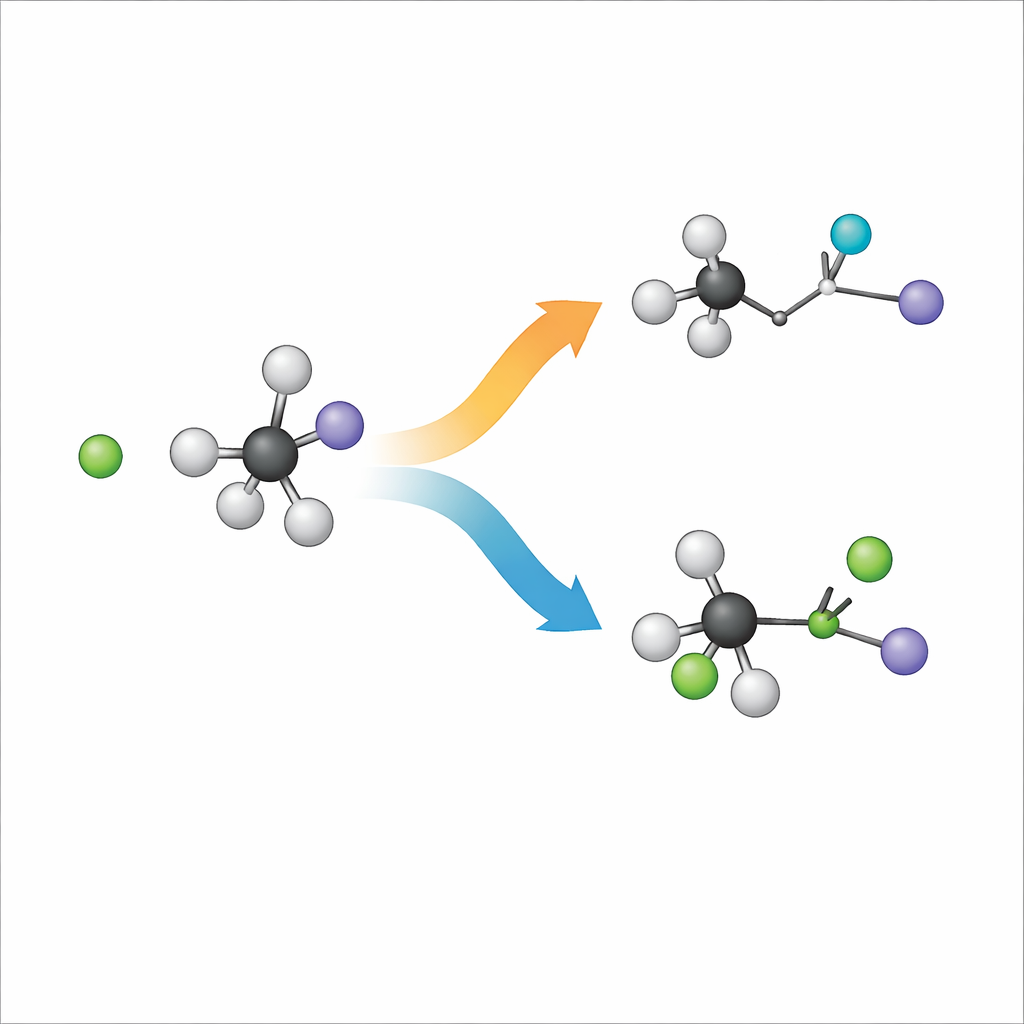
When a negatively charged chloride ion meets a bulky molecule called tert-butyl iodide, two main outcomes are possible. In one path, known as substitution, chloride takes the place of iodine on the carbon framework. In the other, called elimination, chloride grabs a hydrogen atom instead, and the carbon skeleton reshapes to form a double bond while iodine leaves separately. Chemists label these routes SN2 and E2, but at heart they are two different ways the same ingredients can rearrange. Understanding which path wins, and why, is crucial for designing reactions that give the desired product in synthetic chemistry.
Filming reactions in midair
To uncover what really happens, the researchers strip away the clutter of solvents and study the reaction in the gas phase, where single ions and molecules collide in a near-vacuum. They cross a beam of chloride ions with a beam of tert-butyl iodide and use a specialized imaging setup to record the three-dimensional speeds and directions of the iodine-containing fragments that fly out. At the same time, they build an extremely detailed "map" of the forces between all 15 atoms using a machine-learning approach trained on high-level quantum calculations. They then launch millions of simulated trajectories on this 39-dimensional landscape to follow how the atoms move during each collision.
Elimination takes the lead
The experimental images and computer simulations line up remarkably well, giving confidence that the reaction is being captured accurately. Both show that the elimination path dominates: most collisions send the system down the E2 route, especially as the collision energy increases. In these events, the products carry a lot of internal vibrational energy—most of the extra energy ends up shaking and twisting the new molecules rather than simply propelling them apart. This behavior matches earlier work on related systems and highlights how the bulky carbon framework of tert-butyl iodide steers the reaction away from simple substitution.
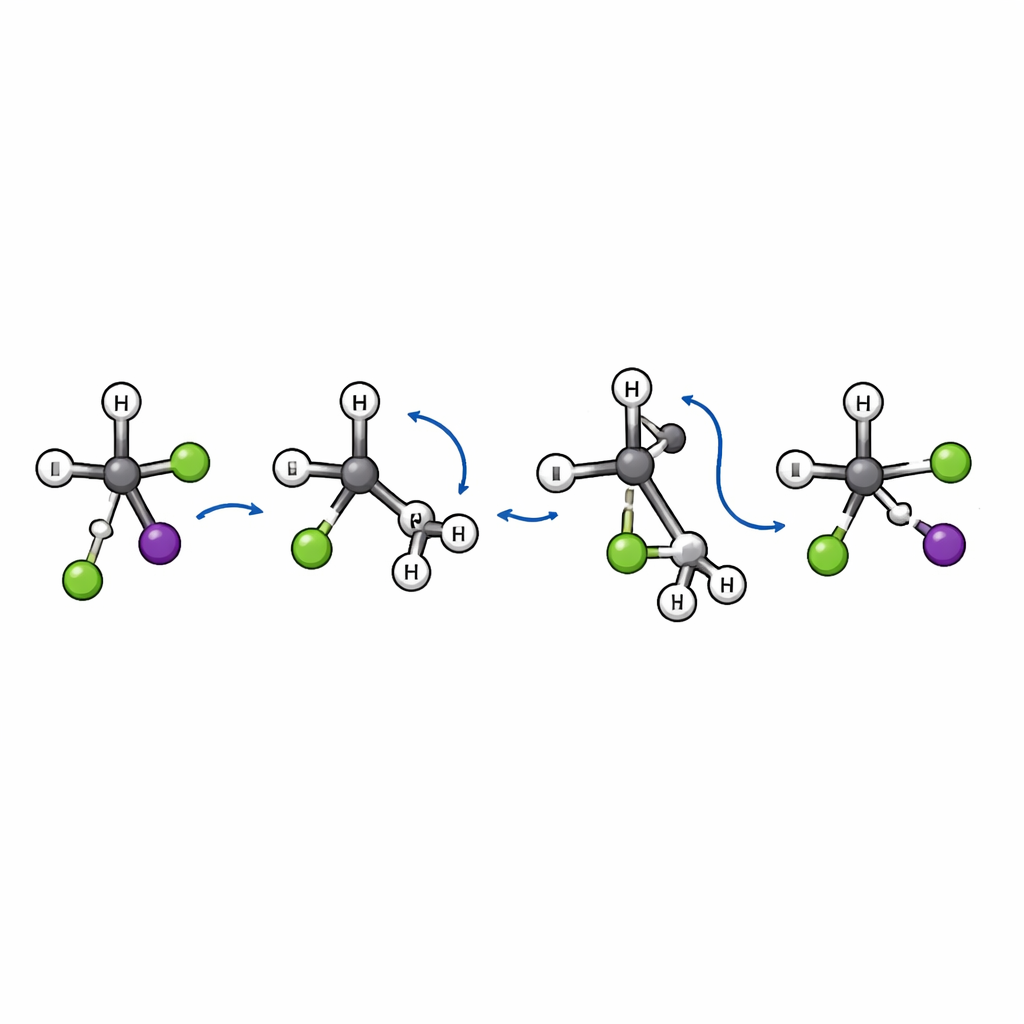
A new kind of molecular flip
Within the smaller fraction of substitution events, the team finds something unexpected. The classic SN2 picture involves a "back-side attack": the incoming ion approaches behind the leaving group and the central carbon inverts its geometry, like an umbrella turning inside out. That inversion changes the three-dimensional handedness of the molecule. Here, the simulations reveal an additional, distinct route. In this newly discovered "flip-over" mechanism, the chloride first approaches and stretches the bond between carbon and iodine. The bulky tert-butyl group then flips as a unit, like a page being turned, while chloride finally bonds to the central carbon and iodine departs. Because the carbon framework flips instead of inverting, the overall spatial arrangement around the key carbon atom is retained rather than reversed.
Signatures of the flip-over motion
This flip-over pathway leaves clear fingerprints in the scattering patterns. Substitution events that proceed through the usual inversion tend to send the iodine fragment backward relative to the incoming chloride. By contrast, trajectories that follow the flip-over route mostly scatter iodine forward, a sign that the collision is very direct and that the bulky group’s rotational motion plays a key role. The authors also identify a specific transition state—a kind of energetic gateway—that connects reactants and products along this new path, confirming that it is a genuine and distinct mechanism rather than a minor variation of known ones.
What this means for chemistry
By combining cutting-edge experiments with an accurate, machine-learned energy landscape, this study shows that even one of the most studied reaction types still hides surprises. The discovery of a flip-over route that preserves the three-dimensional arrangement of atoms broadens the known ways that substitution reactions can control molecular shape. In practical terms, such insights can help chemists better predict when certain products will form, especially in crowded molecules where subtle motions of the carbon framework matter. As similar techniques are applied to more complex systems, other hidden pathways may emerge, refining how we think about the microscopic choreography of chemical change.
Citation: Lu, X., Meyer, J., Li, L. et al. Unveiling a flip-over retention mechanism in the gas-phase Cl− + (CH3)3CI SN2 reaction. Nat Commun 17, 3947 (2026). https://doi.org/10.1038/s41467-026-72121-4
Keywords: SN2 reactions, reaction dynamics, stereochemistry, elimination vs substitution, molecular collision imaging