Clear Sky Science · en
Genome-wide screening reveals producer-cell modifications that improve virus-like particle production and delivery potency
Why Better Delivery Vehicles Matter
Modern gene-editing tools like CRISPR can, in principle, fix disease-causing mutations at their source, but a central challenge remains: how do we safely and efficiently get these molecular tools into the right cells, in the right amount, and only for as long as needed? This study focuses on an emerging delivery vehicle called engineered virus-like particles, or eVLPs—tiny, non-infectious shells that can carry gene-editing machinery. While past work has mainly tuned the particles themselves, this paper asks a different question: what if we re-engineer the living cells that manufacture the particles so that every batch is more potent from the start?
Reprogramming the Factory, Not Just the Product
eVLPs are produced by human “producer” cells in the lab, which assemble viral shell proteins and load them with gene-editing cargo before releasing them into the surrounding fluid. Traditionally, scientists have optimized the particle’s components and architecture—changing shell proteins, cargo fusions, or surface molecules—while assuming the producer cells are just passive factories. The authors challenged this view by systematically testing how thousands of different genetic changes inside producer cells alter the number and quality of eVLPs they release. Their aim was simple but powerful: identify which genes, when turned off, make the cells better at packaging gene-editing tools into particles.
A Genome-Wide Search for Helpful Tweaks
To run this massive search, the team used a genome-wide CRISPR “knockout” library that disrupts almost every human gene. Each producer cell received a mutation guided by a unique RNA barcode, and because eVLPs naturally package these guide RNAs along with the gene-editing protein, each particle carried a molecular tag indicating which genetic change existed in the cell that made it. By comparing how often each barcode appeared in the particles versus in the cells themselves, the researchers could see which gene disruptions boosted or harmed particle production and cargo loading. Most mutations had little effect, many made things worse, and a small but important set significantly improved or impaired the eVLPs’ performance.
Unexpected Roles for Gene Silencing and RNA Production
Among the strongest “negative” hits were three genes (MPP8, TASOR, MORC2) that form part of a gene-silencing system known as the HUSH complex. When these genes were knocked out, the resulting particles carried fewer guide RNAs and showed weaker delivery in cells, largely because excess Cas9 protein in the modified cells grabbed the guides before they could be packaged. Yet the story was more nuanced: HUSH loss also increased production of the cargo protein itself. In situations where the cargo was supplied from DNA stably integrated into the cell’s genome (rather than from transiently added plasmids), this extra protein actually improved the balance of particle components and led to markedly stronger gene-editing activity. In other words, the same genetic tweak could either hurt or help, depending on how the production system was set up.
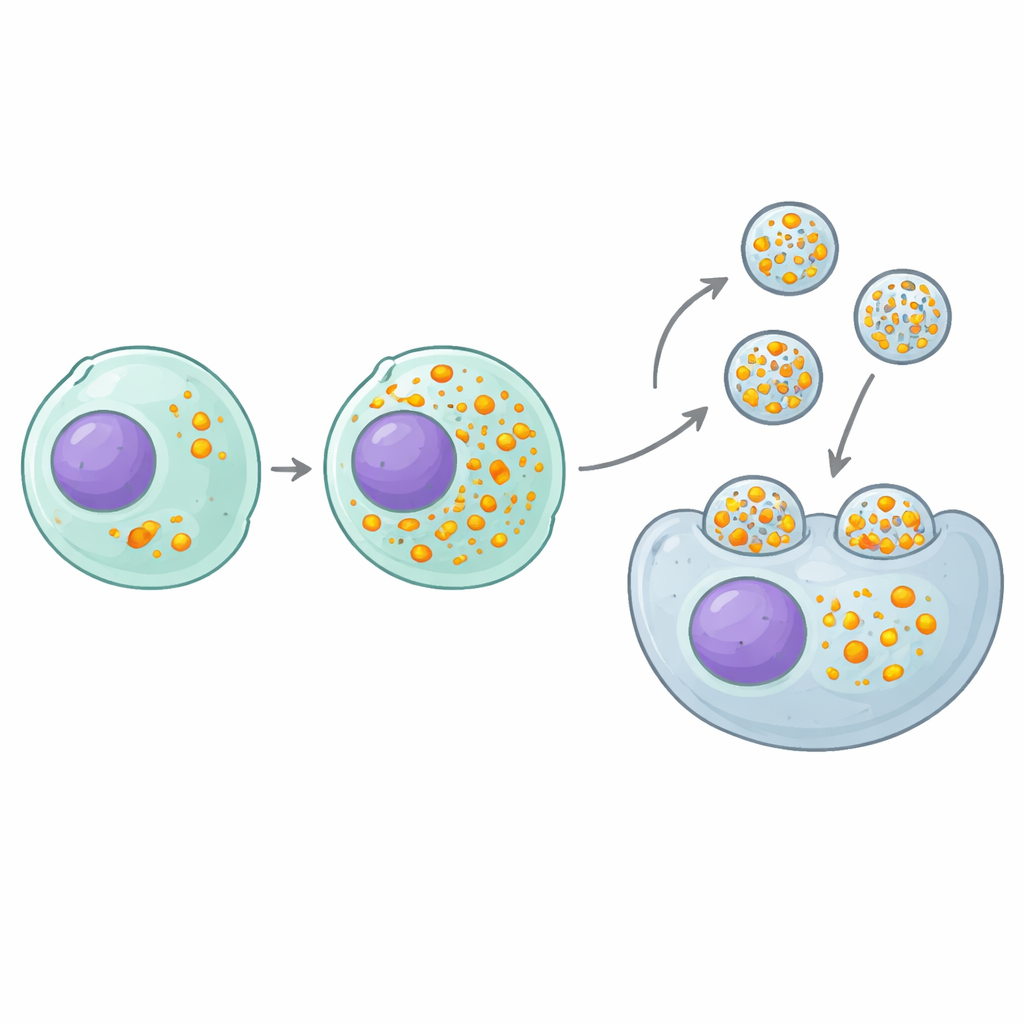
A Key Switch That Packs More Cargo Per Particle
The most promising “positive” hit was a gene called MAF1, a natural brake on a basic cellular machine that makes small RNAs. The guide RNAs used for CRISPR are produced by this machine, so removing MAF1 effectively turns up the faucet on guide RNA production. Producer cells lacking MAF1 loaded roughly twice as many guides into each eVLP without changing how many particles they made or how much protein each particle carried. This translated into a two- to nine-fold boost in editing potency across many different guide sequences, gene targets, and cell types, both in cell culture and in mice. Crucially, the benefit extended beyond this particular eVLP design: several other RNA–protein–carrying particle systems, built on different scaffolds, all became more effective when produced in MAF1-deficient cells.
Combining Strategies for Maximum Impact
The authors next asked whether MAF1 knockout could work together with other tricks to raise guide RNA levels even further. By embedding extra guide RNA cassettes into the standard plasmids used during eVLP production, they increased the amount of RNA available without upsetting the overall DNA balance in the cell. This “++sgRNA” approach and the MAF1 knockout each boosted RNA packaging on their own, and together they were additive: the fraction of Cas9 molecules inside particles that were correctly paired with guides rose from about one in five to about one in two. Particles made this way achieved the highest gene-editing efficiencies seen in the study, all while using the same or even lower doses.
What This Means for Future Gene Therapies
For non-specialists, the key message is that improving gene delivery is not just about building better particles; it is also about upgrading the cellular factories that make them. By mapping how each gene in producer cells influences the quantity and quality of eVLPs, this work identifies practical genetic “dials” that manufacturers can turn to get more potent batches. MAF1-deficient cells, in particular, reliably pack more of the active gene-editing cargo into each particle, allowing the same therapeutic effect at much lower doses. As gene-editing therapies move toward the clinic, such producer-cell engineering offers a powerful and flexible way to make treatments safer, more efficient, and easier to scale.
Citation: Ly, D., Jang, H., Goel, A. et al. Genome-wide screening reveals producer-cell modifications that improve virus-like particle production and delivery potency. Nat Commun 17, 3695 (2026). https://doi.org/10.1038/s41467-026-71925-8
Keywords: virus-like particles, gene editing delivery, producer cell engineering, CRISPR therapeutics, genome-wide screening