Clear Sky Science · en
Signaling cascades shape functional subpopulations of cortical astrocytes in male wild-type mice and APP/PS1dE9 Alzheimer’s disease model
Hidden Helpers in the Brain
Alzheimer’s disease is often described as a story about sick neurons and sticky protein clumps called plaques. But neurons do not work alone. They are surrounded by star-shaped support cells, astrocytes, that quietly keep brain circuits running. This study shows that even among these “helper” cells there are distinct teams with different jobs, and that their internal communication systems can influence both the build-up of plaques and behavior in a mouse model of Alzheimer’s disease.
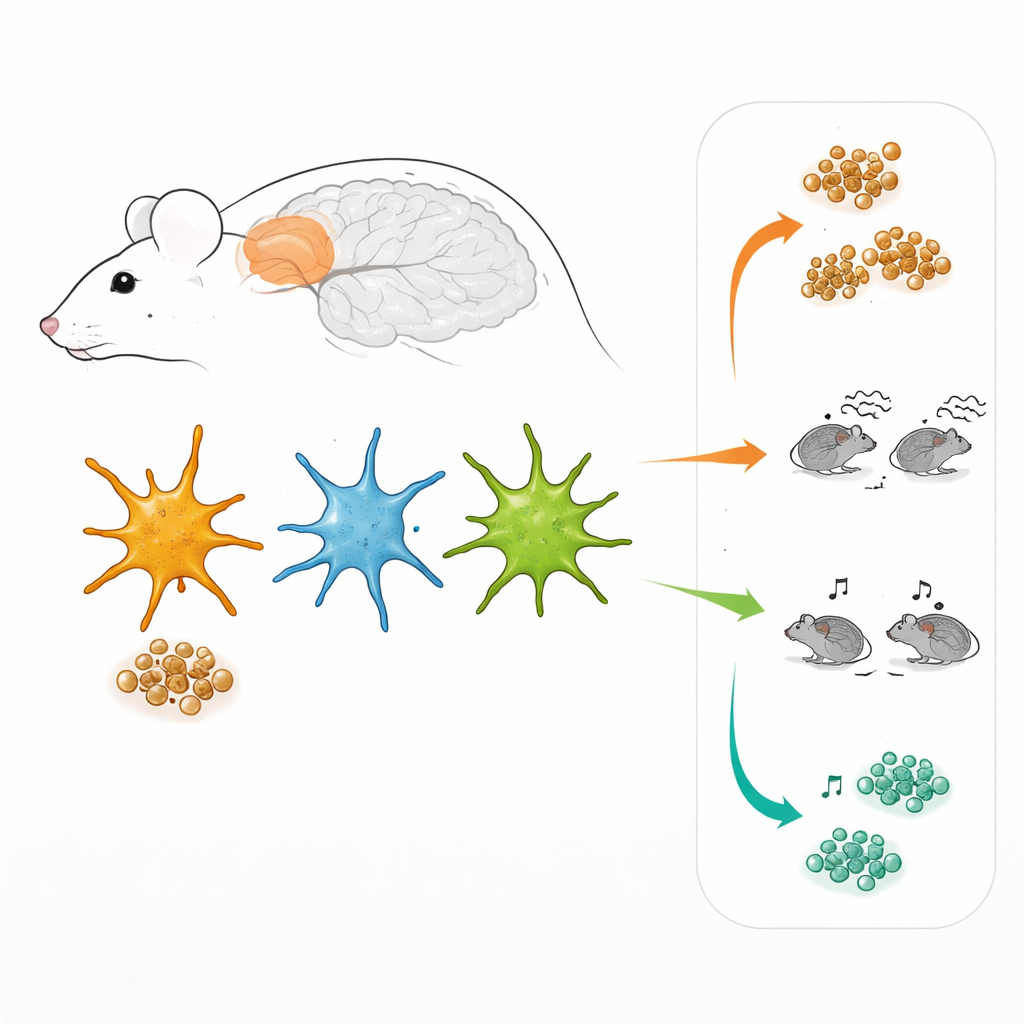
Different Teams of Support Cells
Astrocytes are not a uniform crowd. Working in the prefrontal cortex, a brain region involved in decision-making and social behavior, the researchers used specially designed viruses to make astrocytes light up when two major internal signaling routes were active. One route is controlled by a factor called STAT3, the other by NF-kB. In living brain tissue, this allowed them to “tag” three subgroups of astrocytes: those mainly using the STAT3 route, those mainly using the NF-kB route, and those using both at once. Surprisingly, these three groups were present not only in Alzheimer-like mice but also in healthy mice, and they were scattered throughout the cortex rather than clustered in a particular layer or around plaques.
Not Just a Reaction to Plaques
Because protein plaques in Alzheimer’s disease strongly affect nearby brain cells, one might expect these astrocyte subgroups to appear chiefly around those deposits. Instead, the levels of STAT3 and NF-kB activity in astrocytes did not depend on how close the cells were to plaques or how large the plaques were. The same three astrocyte types were already present in younger mice before many plaques had formed, and in normal mice that never develop amyloid build-up. This suggests that the different astrocyte teams arise from built-in, “hard-wired” properties of the cells, with disease then modifying what each team does rather than creating entirely new types.
Distinct Shapes and Housekeeping Jobs
The three astrocyte teams differed not only in their signaling, but also in their shape, molecular makeup, and everyday chores. Cells dominated by the NF-kB route tended to cover a larger territory in the tissue and showed stronger activity of internal recycling compartments that break down proteins. STAT3-dominated astrocytes, in contrast, were smaller but had more active membrane “gates” called hemichannels, which control the passage of small molecules between the cell and its surroundings. Gene activity measurements from sorted cells confirmed that each group turned on a different set of genes, especially those involved in inflammation and in protein handling and clearance. In the Alzheimer-like mice, these molecular differences were amplified, with some pathways in STAT3-rich cells showing clear signs of stress.
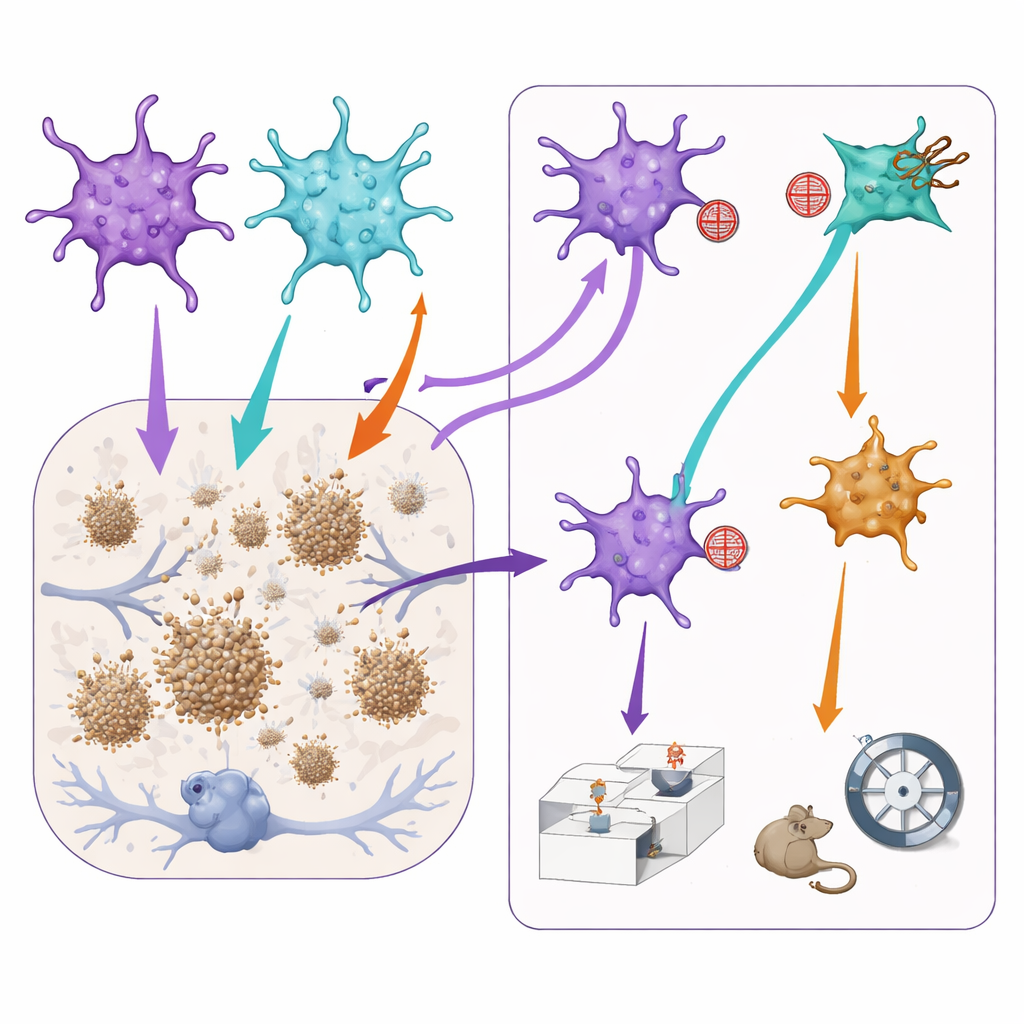
Tuning Astrocyte Signals Changes Plaques and Behavior
To test how these subgroups affect disease features, the team then used additional viral tools to gently dampen either the STAT3 or NF-kB signaling only in astrocytes where those routes were already active. In Alzheimer-like mice, blocking the STAT3 route led to smaller amyloid plaques in the prefrontal cortex, hinting that STAT3-dominated astrocytes may, directly or indirectly, help plaques grow. However, this same manipulation worsened aspects of social memory: mice had more trouble distinguishing a familiar mouse from a new one. Conversely, reducing NF-kB signaling seemed to slightly relieve anxiety-like behavior but also reduced normal social interest and memory. Importantly, performing the same manipulations in healthy mice had little effect, indicating that these astrocyte teams become especially influential in a diseased brain.
What This Means for Alzheimer’s Disease
This work paints a more nuanced picture of the brain’s support cells in Alzheimer’s disease. Rather than responding in a uniform way, astrocytes fall into distinct functional groups defined by their internal signaling cascades. These groups differ in size, protein-cleaning abilities, and how they communicate with neighboring cells, and they contribute differently to plaque growth and to behaviors related to anxiety and social interaction. For future therapies, this suggests that broadly turning astrocytes “up” or “down” may be too crude. Instead, carefully targeting specific signaling routes in selected astrocyte subpopulations could one day help fine-tune the balance between protecting brain circuits, limiting harmful protein build-up, and preserving normal behavior.
Citation: Poulot-Becq-Giraudon, Y., Guillemaud, O., Degl’Innocenti, E. et al. Signaling cascades shape functional subpopulations of cortical astrocytes in male wild-type mice and APP/PS1dE9 Alzheimer’s disease model. Nat Commun 17, 4194 (2026). https://doi.org/10.1038/s41467-026-71826-w
Keywords: astrocytes, Alzheimer’s disease, brain signaling, neuroinflammation, protein clearance