Clear Sky Science · en
Cryo-EM structures of higher order Gephyrin oligomers reveal principles of inhibitory postsynaptic scaffold organization
How Brain Brakes Stay in Tune
Our brains rely on a delicate balance between “go” and “stop” signals. The “stop” signals, carried by inhibitory synapses, prevent runaway activity linked to seizures, anxiety, and other disorders. This study looks at gephyrin, a key protein that builds the microscopic scaffolds holding inhibitory receptors in place at synapses. By visualizing gephyrin’s three‑dimensional shapes with cryo‑electron microscopy, the authors reveal how this protein self‑assembles into larger structures that organize receptors with surprising precision.
The Silent Architect of Inhibitory Synapses
At many inhibitory synapses, gephyrin is the main organizer that anchors two types of receptors: glycine receptors and GABAA receptors. Each gephyrin molecule has rigid end domains connected by a floppy middle region, and different splice variants add further complexity. For years, scientists thought full‑length gephyrin mainly formed three‑part assemblies, and these trimers were used to sketch lattice‑like designs of the postsynaptic scaffold. However, newer imaging of intact brain tissue suggested a more flexible meshwork of receptors and scaffolds, hinting that the old trimer‑centric view was incomplete.
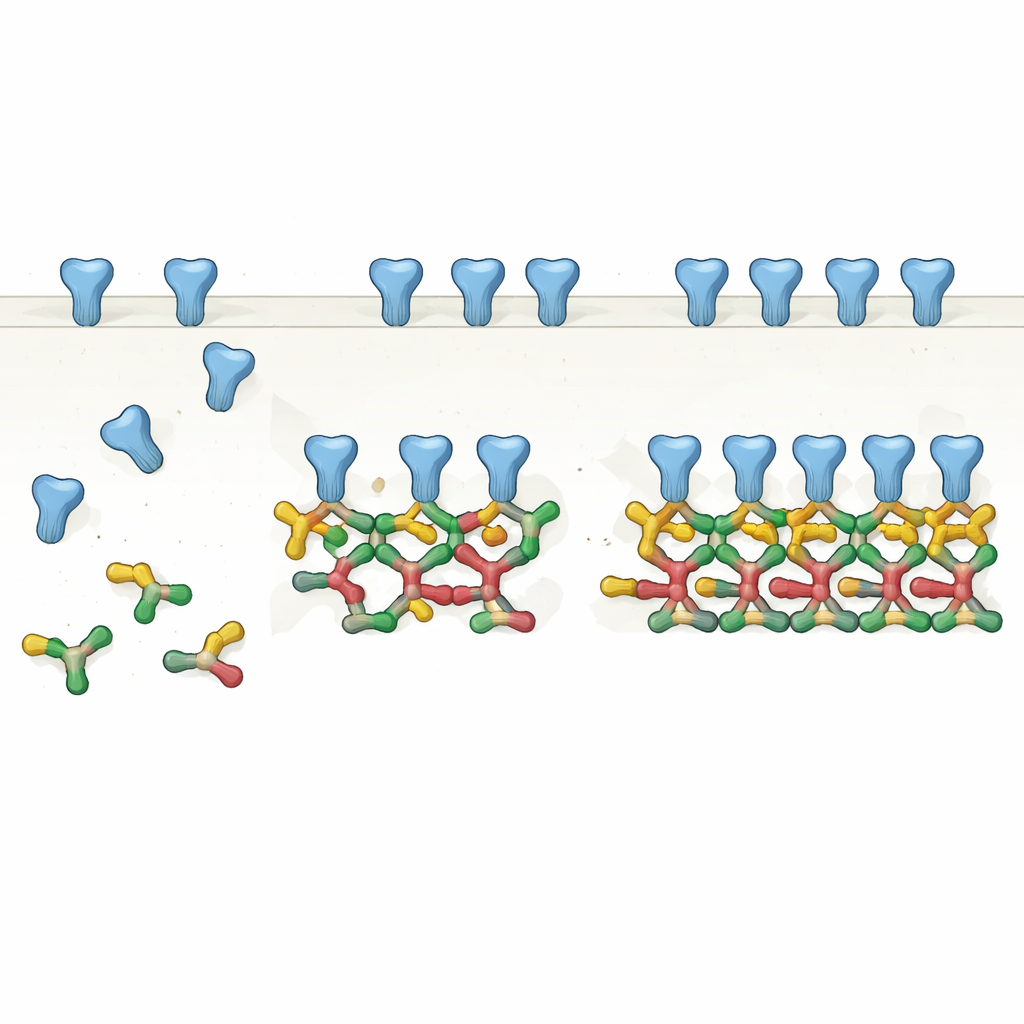
From Pairs to Chains: A New View of the Scaffold
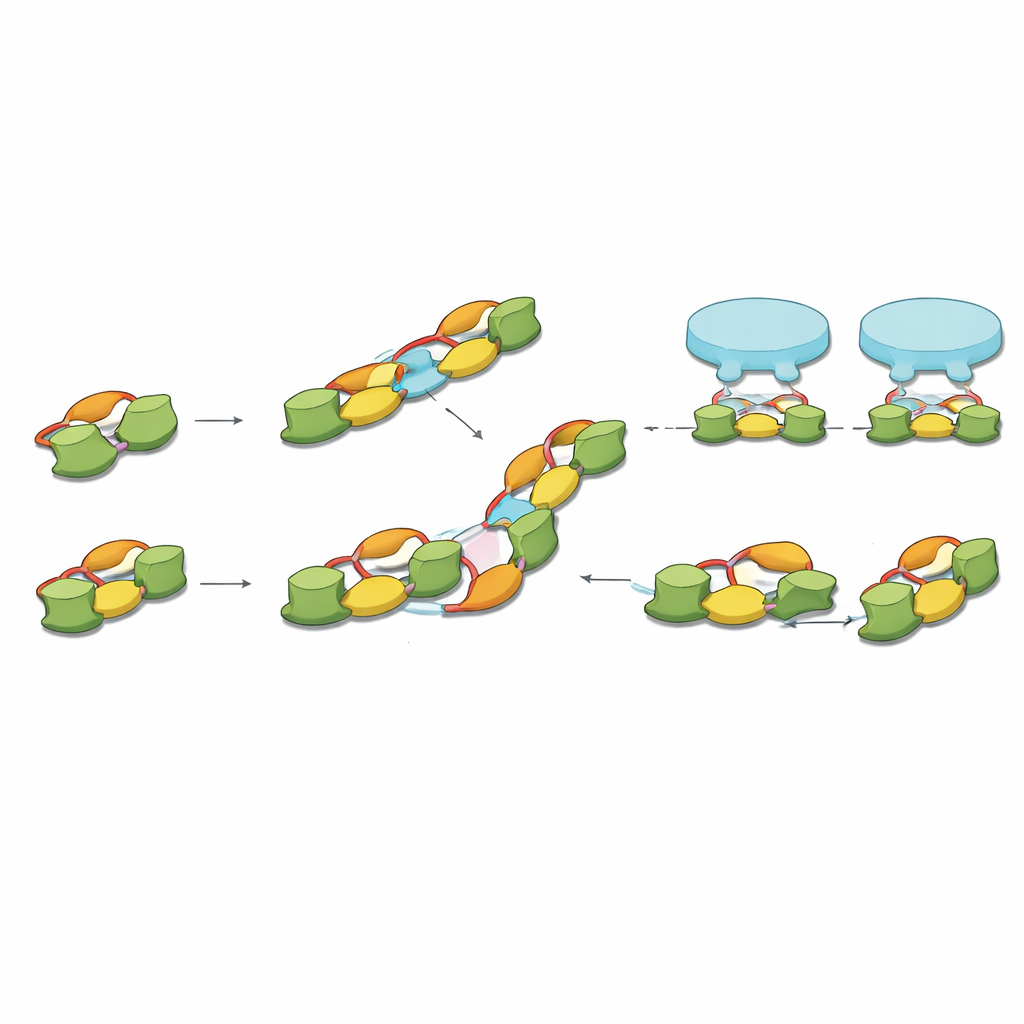
By carefully purifying one common splice form of full‑length gephyrin and analyzing it with several biochemical methods, the authors show that gephyrin most naturally forms pairs, or dimers. These dimers then act as basic building blocks for larger structures: straight chains of four molecules (dimers of dimers) and even six‑membered chains. Using cryo‑electron microscopy, they solved high‑resolution structures of these assemblies and found that specific surfaces on one part of gephyrin repeatedly contact matching surfaces on neighboring dimers. Rather than a static trimeric hub, gephyrin emerges as a modular system that can extend into linear and angled chains, matching the spacing and arrangements of receptors seen in previous in‑situ brain imaging.
A Flexible Linker with a Hidden Switch
One of the most intriguing discoveries lies in the protein’s flexible middle segment, or linker, which had long escaped structural analysis. The team captured this linker wrapping back onto one end domain of gephyrin, where it both stabilizes the protein and reaches into the same surface used to bind receptors. In one conformation, part of the linker sits inside the receptor‑binding pocket, effectively blocking it; in another, it swings out, leaving the pocket open for receptors. Several of the amino acids that change position between these “in” and “out” states are known targets of phosphorylation, a common chemical modification that cells use as an on–off switch. This suggests that brain cells may tune inhibitory strength by chemically nudging gephyrin’s linker between a receptor‑blocking and a receptor‑welcoming shape.
Electrostatic Glue and Liquid Droplets
The study also pinpoints charged residues that act as electrostatic “Velcro,” helping gephyrin dimers link into higher‑order chains and promoting the formation of liquid‑like protein droplets, or condensates, inside cells. When the researchers mutated key positive or negative charges, gephyrin in cultured cells lost its ability to form large droplets and assembled into far fewer or smaller clusters. In neurons, the same mutations weakened the buildup of gephyrin at inhibitory synapses, even though the altered proteins could still reach those sites. Together, these experiments show that the very same charged regions and linker segment that stabilize gephyrin’s structure are also essential for creating dense receptor clusters at real synapses.
Connecting Molecules to Synaptic Patterns
Finally, the authors connect their molecular snapshots to earlier cryo‑electron tomography of intact brain tissue, which had measured a characteristic spacing between neighboring inhibitory receptors. The length of the gephyrin dimer‑of‑dimers closely matches this spacing, and combinations of straight and angled chains can recreate the most common receptor patterns observed in neurons. In this picture, gephyrin dimers provide the basic clamp for receptor pairs, and higher‑order chains emerge as these clamps connect side‑by‑side through charged interfaces.
Why This Matters for Brain Health
Overall, the work replaces a simple trimer‑based cartoon of gephyrin with a dynamic, chain‑forming scaffold whose shape and receptor‑binding capacity can be tuned by subtle chemical marks on a flexible linker. This helps explain how inhibitory synapses can be both structurally ordered and rapidly adjustable, and it offers a molecular framework for understanding disease‑linked mutations that disrupt gephyrin’s assembly. By clarifying how the brain’s braking system is physically built and modulated, the study opens avenues for targeting inhibitory balance in conditions such as epilepsy, autism spectrum disorders, and anxiety.
Citation: Ortiz-López, D., Hove, T.T., Huhn, C. et al. Cryo-EM structures of higher order Gephyrin oligomers reveal principles of inhibitory postsynaptic scaffold organization. Nat Commun 17, 3541 (2026). https://doi.org/10.1038/s41467-026-71771-8
Keywords: gephyrin, inhibitory synapse, GABAA receptor, cryo-electron microscopy, synaptic scaffold