Clear Sky Science · en
Trajectories of plasma and CSF MTBR-tau243 and phosphorylated-tau species across the Alzheimer’s disease continuum
Why timing matters in Alzheimer’s disease
Alzheimer’s disease quietly unfolds over many years before memory problems become obvious. Doctors and researchers are racing to catch it earlier, both to test new drugs and to guide treatment for patients. Brain scans can reveal the build-up of the proteins amyloid and tau, which define Alzheimer’s disease, but these scans are expensive and not widely available. This study explores whether simple blood tests and spinal fluid tests can reliably track how Alzheimer’s disease progresses over time, potentially offering a more accessible way to stage the illness and forecast its course.
Following the disease from silent beginnings
The researchers drew on a large Swedish study of people ranging from cognitively healthy to those with mild memory problems and dementia. All had advanced brain imaging to measure amyloid and tau deposits and gave blood and, in a subset, cerebrospinal fluid taken by lumbar puncture. Using a mathematical method called SILA, the team built a "disease clock" for each person, estimating how many years had passed since amyloid or tau first became detectable on scans. They then mapped how different forms of tau floating in the blood and spinal fluid rose and fell along this timeline.
Early chemical warning signs in body fluids
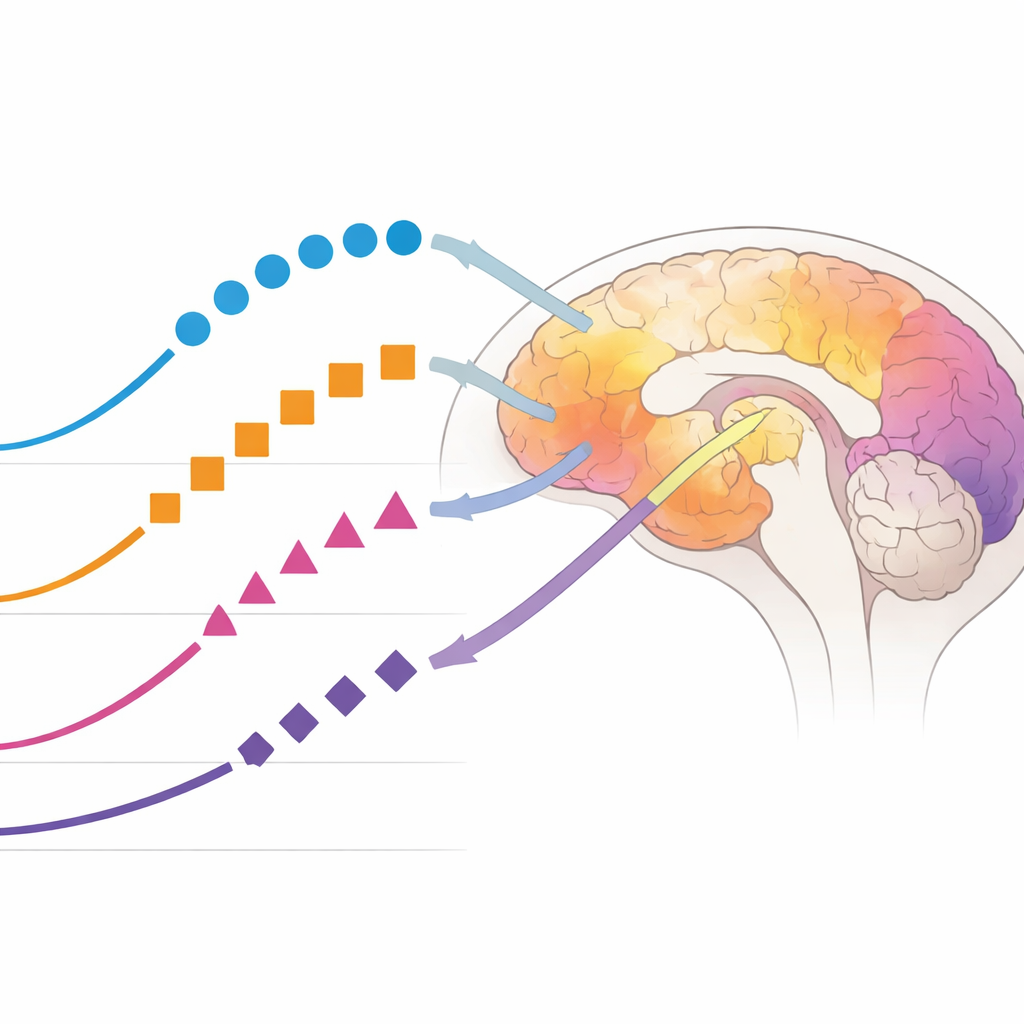
One particular chemical modification of tau, known as phosphorylation at position 217, emerged as the most sensitive early signal. The fraction of tau carrying this change (%p-tau217) in both spinal fluid and blood began to shift roughly one to two years before amyloid scans turned positive, well before clear memory decline. Other phosphorylated forms of tau changed later, rose less dramatically, and tended to plateau sooner. This pattern suggests that %p-tau217 could flag the earliest stages of disease, when amyloid is starting to accumulate and tau is beginning to react, but before large tangles of tau have formed throughout the brain.
Markers that mirror tangle build-up
As the disease clock ticked forward and tau tangles spread through the cortex, a different set of markers became most informative. A fragment from the microtubule-binding region of tau, called MTBR-tau243 in spinal fluid and eMTBR-tau243 in blood, closely rose in step with tau seen on brain scans, especially in regions affected in later stages. Another phosphorylated form, %p-tau205, began to change around the time tau scans first became positive. Unlike most other tau measures, MTBR-tau243 in spinal fluid kept rising sharply even in advanced disease, and its blood counterpart tracked the amount of cortical tau particularly well. These findings support the idea that MTBR-tau243 reflects the presence of actual tau tangles in brain tissue rather than just early biochemical disturbances.
Late-stage signs and individual differences
Not all tau signals behave the same way. Forms of tau that lack added phosphate groups in the mid-section of the molecule remained mostly stable until very late in the disease, when memory and thinking had clearly worsened. At that point they rose, marking advanced damage and suggesting their use as late-stage indicators. The team also examined whether sex or carrying the APOE-ε4 risk gene changed the overall ordering of these markers. While people with the risk gene tended to show abnormal blood and spinal fluid readings somewhat earlier, the sequence of events—early %p-tau217 changes, followed by %p-tau205 and MTBR-tau243 surges, and finally late non-phosphorylated tau—remained largely the same across groups.
What this means for patients and future treatments
Taken together, the study shows that no single blood or spinal fluid test can capture the entire story of Alzheimer’s disease, but a small panel can. An early marker like %p-tau217 signals when amyloid-driven changes begin; mid-stage markers such as %p-tau205 and MTBR-tau243 mirror the spread of tau tangles; and non-phosphorylated tau highlights late damage. Because these markers can be measured in blood or spinal fluid, they could one day reduce the need for expensive brain scans, help doctors pinpoint where a person lies on the disease continuum, and match them to the right kind of treatment or clinical trial at the right moment.
Citation: Collij, L.E., Salvadó, G., Horie, K. et al. Trajectories of plasma and CSF MTBR-tau243 and phosphorylated-tau species across the Alzheimer’s disease continuum. Nat Commun 17, 3400 (2026). https://doi.org/10.1038/s41467-026-71732-1
Keywords: Alzheimer’s biomarkers, plasma tau, cerebrospinal fluid, amyloid and tau PET, disease progression