Clear Sky Science · en
p38 MAPK orchestrates cross-tissue potassium homeostasis for survival
Why This Matters for Everyday Health
Potassium is a simple mineral you see listed on food labels, yet too little of it in the body can trigger muscle spasms, heart problems, and even be life‑threatening. This study reveals that animals don’t handle potassium shortages one cell at a time. Instead, they use a body‑wide alarm-and-response system, coordinated by a stress-sensing pathway called p38, to keep potassium levels in balance and preserve survival. Because this system is partly conserved from tiny worms to mammals, it hints at new ways to understand human disorders linked to low potassium and aging.
A Tiny Worm as a Window into Potassium Balance
The researchers used the microscopic worm Caenorhabditis elegans as a living testbed to explore what happens when dietary potassium runs low. They placed adult worms in carefully controlled liquid solutions that either lacked potassium or contained different amounts of it, while keeping other salts and conditions constant. Worms with a working p38 signaling pathway survived potassium-poor conditions far longer than mutant worms missing key p38 components. Adding potassium chloride rescued the fragile mutants, whereas sodium or calcium salts could not. This showed that the problem was not generic “salt stress” but a specific inability to cope with potassium loss.
A Stress Pathway That Turns on Survival Genes
To understand how p38 helps worms withstand potassium scarcity, the team dissected the full chain of proteins in this pathway, from an upstream sensor (TIR-1) through a kinase cascade (NSY-1, SEK-1, PMK-1) down to a transcription factor called ATF-7, which controls gene activity. When any of these pieces were disabled, worms died rapidly under potassium deficiency, even though many other well-known longevity and stress genes were far less critical. Genetic screens pinpointed ATF-7 as a central player: when ATF-7 was locked in its “repressor” form, survival was even worse, indicating that p38 must modify ATF-7 to flip specific protective genes on and turn harmful repression off during potassium stress.
A Potassium Pump in the Skin-Like Layer
By comparing gene activity in worms under normal, high-potassium, and low-potassium conditions, the authors found hundreds of genes that switched on only when potassium was scarce. Many of these were typical immune and stress-response genes, but one stood out: a P-type ATPase pump called CATP-3, specialized for moving potassium across membranes. Worms lacking CATP-3 looked much like p38 mutants under deficiency: they developed muscle spasms, body shrinkage, and ruptured internal tissues, and died quickly when external potassium dropped below a few millimoles. Using fluorescent tags, the team showed that CATP-3 is mainly produced in the hypodermis—the worm’s skin-like outer sheet of cells that wraps around muscles and internal organs. Restoring CATP-3 only in this layer was enough to rescue survival, whereas expressing it in muscle or certain neurons was not, implying that the hypodermis acts as a key gateway for potassium uptake into the whole body.
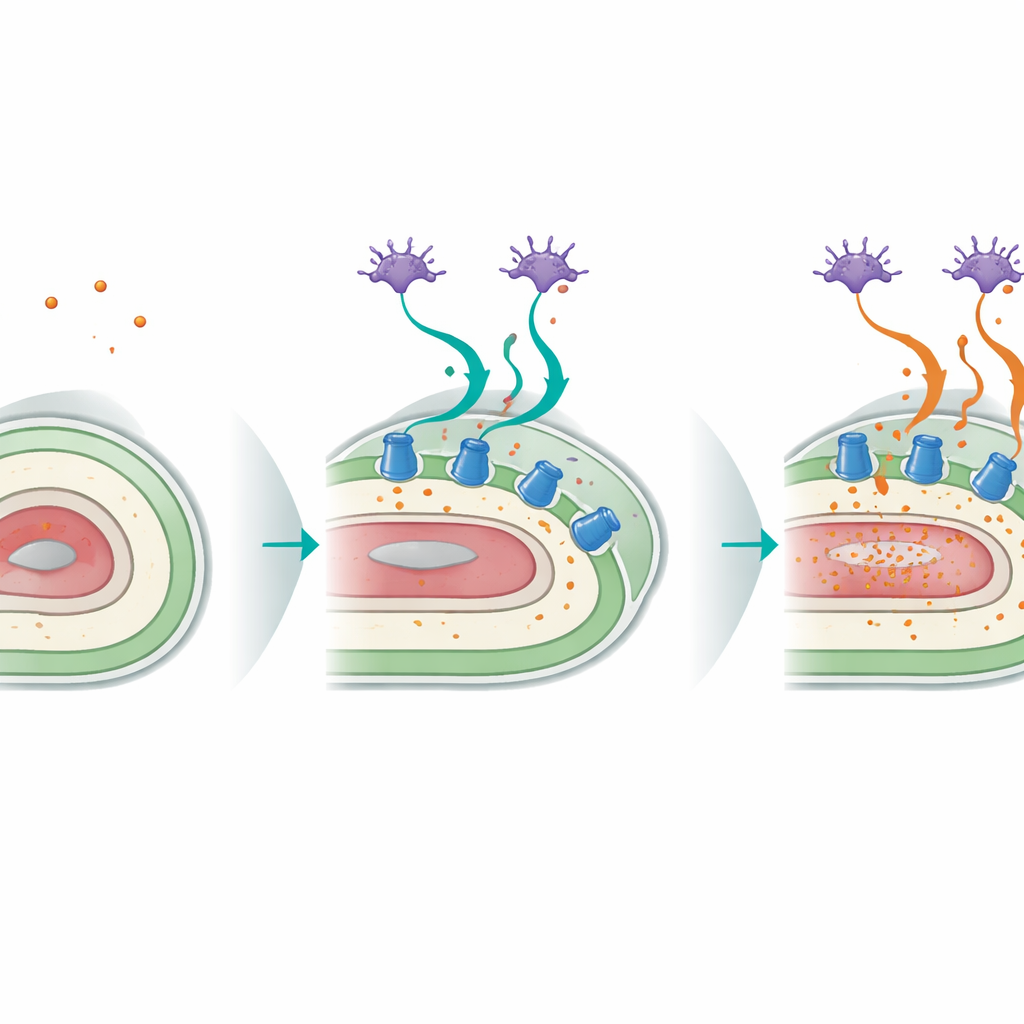
Cross-Talk Between Sensing Neurons and the Body Surface
The study also uncovered a striking dialogue between the worm’s sensory neurons and its hypodermis. Special head neurons known as ASI, which normally detect food and other environmental cues, rely on p38 signaling to help orchestrate the response to potassium loss. When p38 was restored only in ASI neurons of otherwise mutant worms, survival under low potassium improved markedly, and the crucial CATP-3 pump in the hypodermis was re-induced. Tissue-specific gene-silencing experiments showed that p38 function in neurons and in the hypodermis is both necessary and intertwined: neurons sense the nutrient threat, while the skin-like tissue executes the protective program by ramping up potassium pumps. This cross-tissue circuit becomes less effective with age, as expression of CATP-3 and its induction by potassium deficiency both decline.
A Conserved Strategy from Worms to Muscles and Yeast
To test whether this logic extends beyond worms, the authors turned to mouse muscle cells and budding yeast. In cultured mouse myoblasts, p38 activity and potassium were both required for the cells to mature into contracting muscle fibers. During differentiation, p38-dependent genes included specific Na⁺/K⁺ pump subunits, such as Atp1a2, which help maintain ion gradients in excitable tissues. In yeast, the p38-like kinase Hog1 and the transcription factor Sko1 regulated P-type ATPase genes (ENA family) that manage sodium and potassium export, and influenced cell size under varying salt conditions. Across these systems, a conserved theme emerged: a p38-type stress pathway adjusts the production of energy-hungry ion pumps to stabilize potassium balance when conditions change.
What This Means for Health and Aging
In accessible terms, this work shows that potassium balance is guarded by a body-wide early warning network, rather than by isolated cells passively leaking ions. A stress sensor (p38) listens to signals from environmental-sensing neurons and then orders the skin-like barrier to install more molecular “pumps” that pull potassium back into the body, keeping muscles and nerves from failing during shortages. Similar control systems appear to operate in mammalian muscle and in yeast, and they weaken with age. Understanding how this circuitry fails or can be re-energized could eventually inform strategies to manage hypokalemia, muscle wasting, and age-related decline in humans.
Citation: Huang, R., Hu, F., Li, Y. et al. p38 MAPK orchestrates cross-tissue potassium homeostasis for survival. Nat Commun 17, 3663 (2026). https://doi.org/10.1038/s41467-026-70641-7
Keywords: potassium homeostasis, p38 MAPK, Caenorhabditis elegans, ion pumps, aging