Clear Sky Science · en
Magnetic-driven multifunctional optoelectronic catheter for in vivo chemical mapping and precisely guided-tumor therapy
Smarter Tools for Finding and Treating Hidden Tumors
Cancer doctors increasingly rely on minimally invasive procedures, threading thin tubes through blood vessels or body cavities to reach hard-to-access tumors. Yet today’s tools often struggle to steer precisely, see tumor edges clearly, and deliver treatment without harming healthy tissue. This research introduces a new type of tiny, magnetically guided catheter designed to navigate complex anatomy, sense the tumor’s chemical footprint in real time, and deliver focused therapy right where it is needed—aiming to make cancer treatment both more accurate and gentler on the rest of the body.
Why Tumor Edges Are So Hard to See
Standard cancer care still leans heavily on biopsies, scans, surgery, chemotherapy, and radiation. While lifesaving, these approaches have blind spots. A single biopsy sample may miss parts of a patchy, irregular tumor, and the long wait between sampling and treatment can delay crucial decisions. Surgeons often cannot see the true boundary between cancer and normal tissue, which raises the risk of leaving behind stray cancer cells. Systemic chemotherapy and radiation, meanwhile, bathe the whole body in treatment, causing side effects because they cannot easily distinguish sick cells from healthy ones. The authors argue that what is needed is a tool that can move nimbly inside the body, map tumor chemistry on the spot, and focus therapy only where it is truly required.
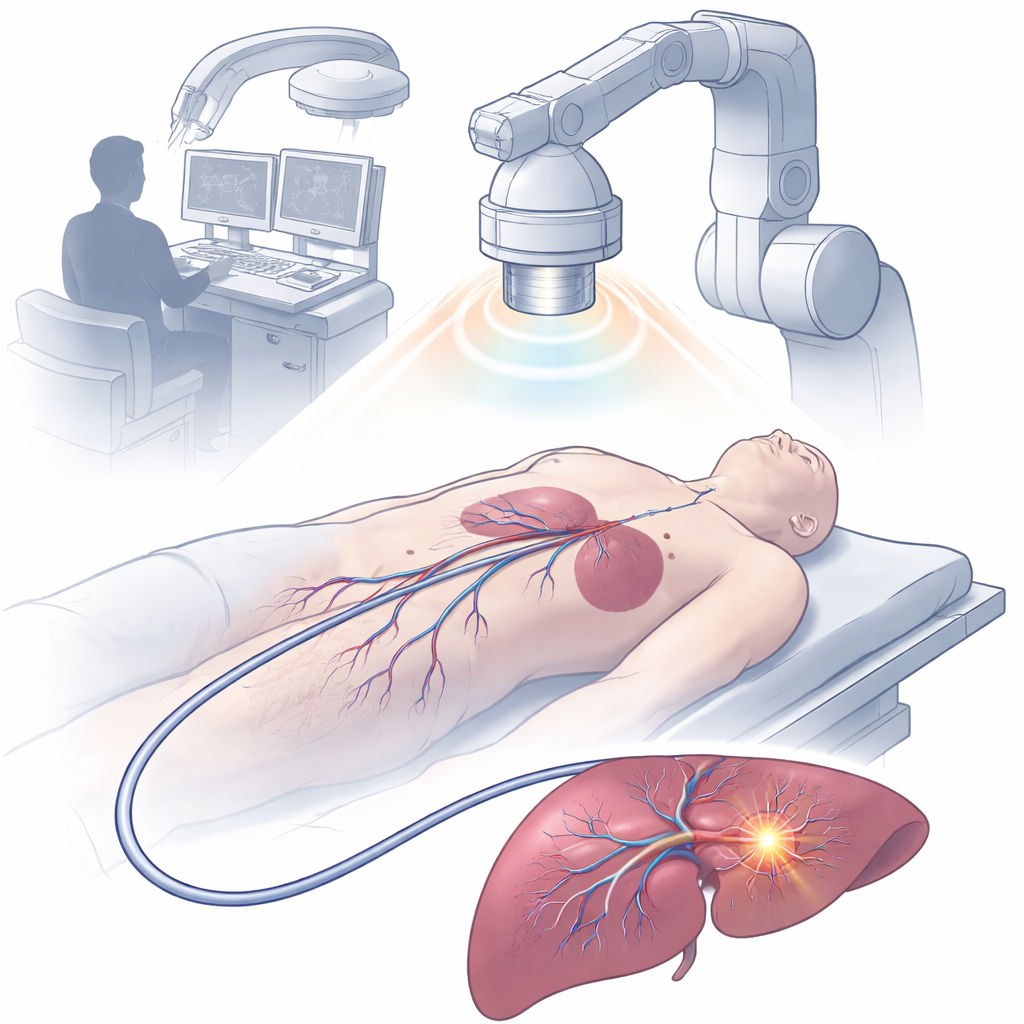
A Tiny Magnet-Guided Catheter Packed With Functions
The team built a 2.5-millimeter-wide device called a magnetic-driven multifunctional optoelectronic catheter, or MDMOC. Using advanced 3D multi-axis printing, they assembled a flexible shaft that contains liquid-metal channels for electrical conduction, an optical fiber for light delivery, hollow paths for drugs, and ring-shaped magnets for steering and X-ray visibility. The tip of the catheter is capped with a small polymer head pierced by diamond-shaped pores that let local fluids flow past protected sensing electrodes. A thin hydrogel coating on the outside makes the catheter surface slippery, cutting friction with tissues and helping it move safely through blood vessels and organs. Despite its small size, the MDMOC can be remotely bent and aimed by an external magnetic field, allowing it to snake through sharp turns and complex vascular branches.
Reading the Tumor’s Chemical Signature in Real Time
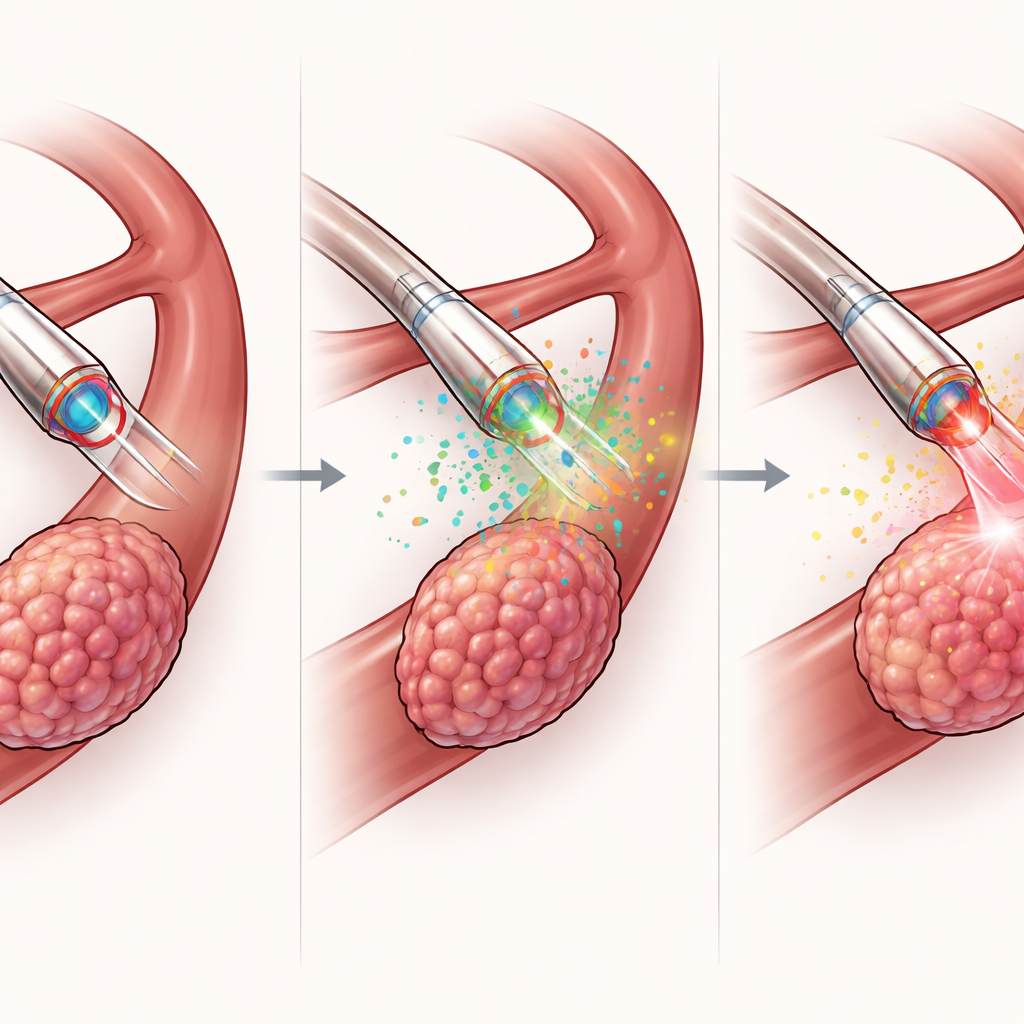
What sets this catheter apart is its built-in “chemical eyes.” Four miniature electrochemical sensors at the tip continuously measure key markers that differ between tumors and healthy tissues: acidity (pH), hydrogen peroxide, potassium ions, and glutathione, a molecule tied to cellular stress and defense. In laboratory tests, each sensor showed high sensitivity, selectivity, and stability, even in the presence of other common body chemicals. When used in rabbits with liver tumors, the catheter was magnetically guided through a small abdominal opening to suspected lesion sites. By sampling multiple points, the team produced detailed chemical maps that revealed sharp gradients where tumor met normal tissue. Combining data from all four sensors into a single “fusion” map improved boundary detection down to roughly sub-millimeter scales, and matched well with ultrasound scans and gold-standard tissue pathology.
From Mapping to Targeted Treatment
Beyond diagnosis, the MDMOC is designed to act. Through its internal channel, it delivers a concentrated dose of a light-activated drug directly into the tumor, using far less drug than systemic routes. The optical fiber then shines red light to trigger photodynamic therapy, causing the drug to generate reactive oxygen species that kill nearby cancer cells. In mouse models of liver cancer, this localized approach led to stronger tumor shrinkage, smaller final tumor volumes, and more cancer cell death than a comparable treatment delivered through the skin or drug-free light alone, all without noticeable weight loss or damage to major organs. Importantly, the same catheter that guides therapy also measures the tumor’s chemistry before and during treatment, enabling a rapid “sense–decide–treat” loop within minutes instead of days.
Testing the Device in Larger, More Realistic Bodies
To explore how well this technology might translate to clinical practice, the researchers tested a longer version of the catheter in pigs, whose blood vessels and organs more closely resemble those of humans. Under X-ray imaging and magnetic control, the MDMOC navigated from major veins into narrow branches of the hepatic and renal veins, taking sharp turns that would challenge conventional catheters. It could deliver contrast agents precisely into these branches without leakage, and under laparoscopic guidance it punctured specific spots on the liver surface and bladder wall to perform in situ sensing and simulated treatment. Even with organ motion from the beating heart, the magnetically guided catheter remained stable at target sites, highlighting its promise for real-world, image-guided procedures.
What This Could Mean for Future Cancer Care
In essence, this work points toward a new class of smart, magnetically controlled catheters that combine navigation, chemical mapping, and focused therapy in a single, slender tool. By reading the tumor’s chemical landscape on the fly and using that information to steer drugs and light exactly where they are most effective, the MDMOC could help surgeons more accurately define tumor margins, spare healthy tissue, and reduce whole-body side effects. While more studies are needed before human use, especially to broaden the range of detectable biomarkers and tailor the system to specific cancers, the concept offers a glimpse of minimally invasive cancer treatments that are not just guided by images—but by the tumor’s own chemistry.
Citation: Chen, F., Liu, X., Zhang, Y. et al. Magnetic-driven multifunctional optoelectronic catheter for in vivo chemical mapping and precisely guided-tumor therapy. Nat Commun 17, 3725 (2026). https://doi.org/10.1038/s41467-026-70529-6
Keywords: magnetically guided catheter, tumor microenvironment, minimally invasive cancer therapy, real-time biosensing, photodynamic therapy