Clear Sky Science · en
A microbiota-IPA axis facilitates intestinal stem cell-mediated regeneration in colitis through a Hopx-associated program
How Friendly Gut Germs Help a Hurting Intestine Heal
Millions of people with inflammatory bowel disease live with a chronically irritated and damaged intestine. When the inner lining of the gut breaks down, it becomes painful and leaky, allowing microbes and food particles to irritate the body even more. This study reveals how certain helpful gut bacteria and the chemicals they make can switch on a special backup pool of intestinal stem cells, speeding up repair of this delicate barrier. Understanding this partnership between microbes, diet, and stem cells points toward gentler treatments that help the gut rebuild itself instead of relying only on strong immune-suppressing drugs.
A Missing Helper in the Inflamed Gut
The researchers began by comparing the gut microbes of people with Crohn’s disease, a form of inflammatory bowel disease, to those of healthy volunteers. They found that a normally common bacterium, Blautia coccoides, was strikingly reduced in patients and in several mouse models of colitis. When they purposely added this bacterium to the intestines of mice, the animals were much better protected from chemically induced gut injury: their colons stayed longer and less shrunken, tissue damage under the microscope was milder, and inflammatory signals were reduced. The protective effect held up across different kinds of gut injury, including chemical irritation and radiation, suggesting that this microbe broadly supports the intestine’s ability to withstand and recover from harm. 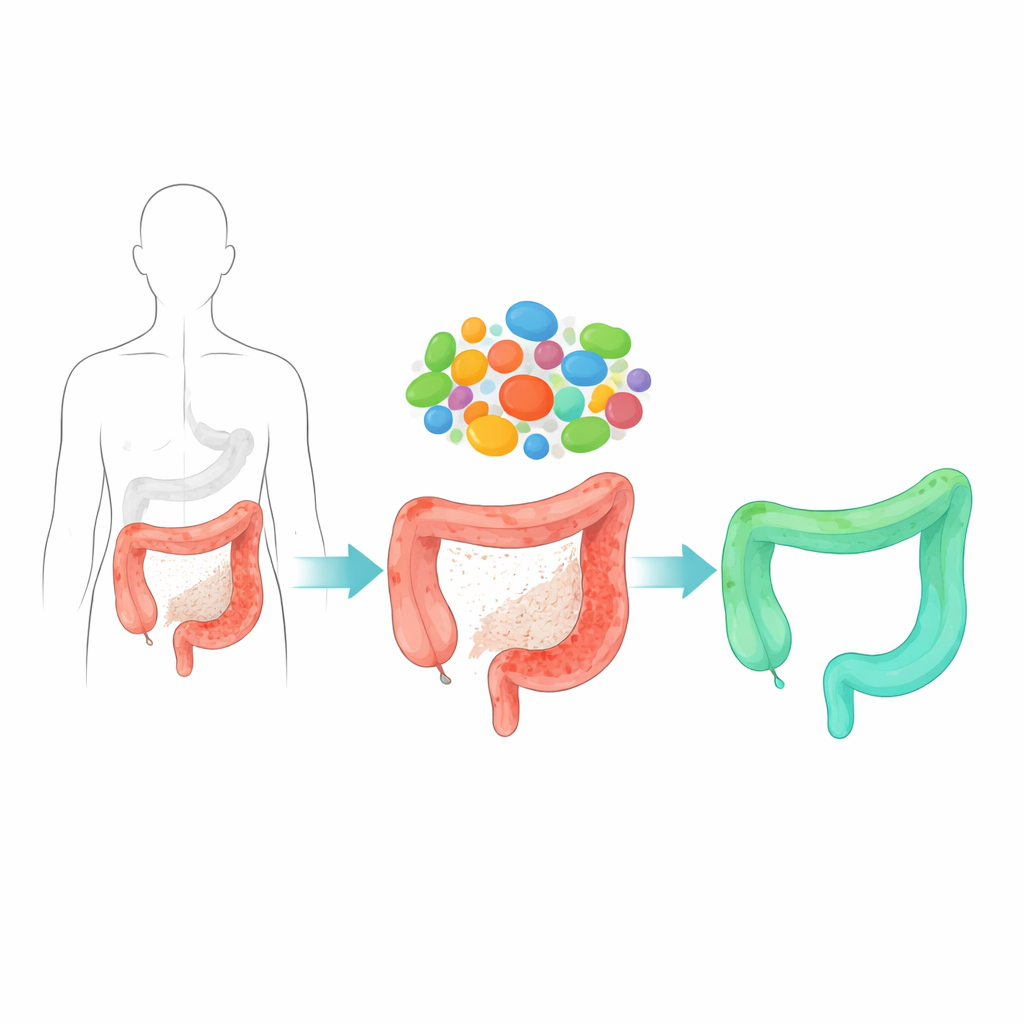
Backup Stem Cells: The Gut’s Emergency Crew
The lining of the intestine is renewed constantly by stem cells that live in tiny pits called crypts. The best-known stem cells are highly active but fragile; they are often wiped out during severe inflammation. The team showed that Blautia coccoides helps another, more resilient group of “reserve” stem cells step in. In mice given this bacterium, genes linked to reserve stem cells were switched on early after injury, and both reserve and active stem cell compartments were preserved days later, when control animals had largely lost theirs. Experiments tracing the fate of reserve stem cells showed that, under the bacterium’s influence, these quiet cells woke up, multiplied, and gave rise to new active stem cells that rebuilt the damaged crypts. Human and mouse mini-intestines grown in the lab confirmed that this regenerative program can also operate in human tissue.
A Chemical Chain from Diet to Microbe to Stem Cell
Next, the scientists asked how a gut bacterium could send such a precise repair signal. They discovered that Blautia coccoides reshapes the chemistry inside intestinal cells to boost production of a small energy-related molecule called beta-hydroxybutyrate. Rather than being made by the bacterium itself, this compound is produced by the host cells when they sense certain microbial products. Beta-hydroxybutyrate turned out to be crucial: giving it directly to mice or human organoids mimicked the bacterium’s ability to activate reserve stem cells after injury and then shift the system back toward a replenished pool of active stem cells as healing progressed. When the researchers deleted a key gene, Hopx, specifically in reserve stem cells, beta-hydroxybutyrate no longer improved regeneration, proving that this chemical acts through a Hopx-linked program in the backup stem cell pool.
Teamwork Between Gut Bacteria and Diet
Digging deeper, the team mapped a longer chain connecting diet, different bacterial partners, and stem cell behavior. Blautia coccoides converts the dietary amino acid tryptophan into an intermediate compound called indole-3-lactic acid, but cannot complete the pathway alone. Other common gut microbes, such as Peptostreptococcus russellii and Clostridium sporogenes, take over from there, transforming this intermediate into indole-3-propionic acid. This final product stimulates intestinal cells to make more beta-hydroxybutyrate, which then activates Hopx-positive reserve stem cells. Co-culturing these microbes in dishes, or co-colonizing germ-free mice, increased levels of indole-3-propionic acid and boosted stem cell repair signals. An engineered strain of E. coli carrying a single enzyme from Blautia reproduced this pathway in mice, further confirming the key steps. 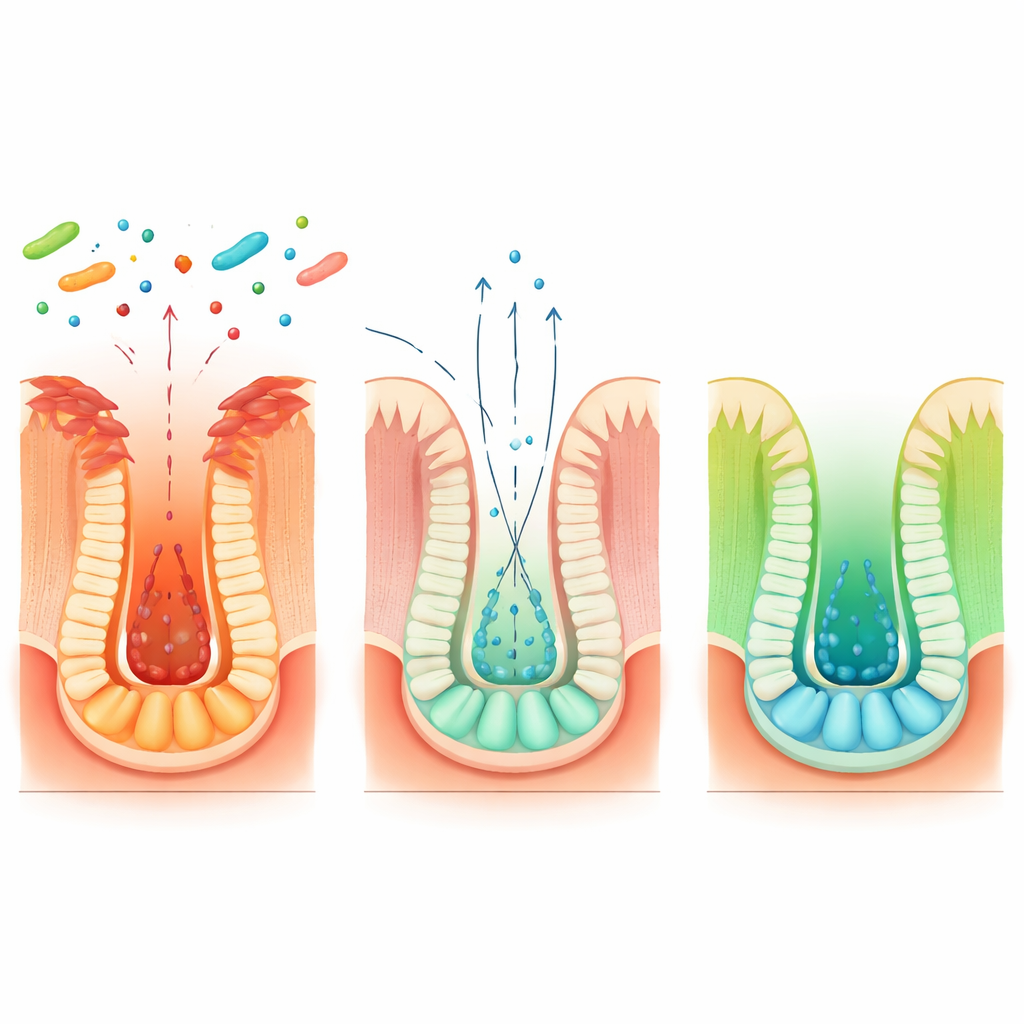
What This Means for Future Gut Therapies
To test the importance of diet, the researchers placed mice on a tryptophan-free regimen that sharply reduced production of indole compounds. These animals suffered worse colitis and weaker stem cell responses. Remarkably, supplying indole-3-propionic acid directly, or reintroducing tryptophan together with the engineered bacteria, restored healing and stem cell activation. Taken together, the work outlines a “microbiota–metabolite–stem cell” axis: dietary tryptophan is processed by a team of gut microbes into a signal that prompts intestinal cells to generate beta-hydroxybutyrate, which then switches on reserve stem cells via Hopx to rebuild the damaged lining. For patients, this suggests a new class of therapies—using carefully chosen microbes, enzymes, or metabolites, along with diet—to gently encourage the intestine’s own repair crews rather than simply turning down the immune response.
Citation: Zhang, Y., Meng, J., Tu, S. et al. A microbiota-IPA axis facilitates intestinal stem cell-mediated regeneration in colitis through a Hopx-associated program. Nat Commun 17, 3196 (2026). https://doi.org/10.1038/s41467-026-70062-6
Keywords: gut microbiome, inflammatory bowel disease, intestinal stem cells, microbial metabolites, mucosal healing