Clear Sky Science · en
VRAC coordinates the trade-off between nutrient absorption and antimicrobial defense in enterocytes against inflammation
Why the Gut’s Balancing Act Matters
Our intestines face a constant dilemma: they must soak up nutrients from food while also fending off harmful microbes. This study explores how a little-known gate in intestinal cells, called VRAC, helps manage that trade-off. When this gate malfunctions, the gut’s delicate balance tips toward overzealous defense, poor nutrient handling, disturbed microbes, and chronic inflammation similar to inflammatory bowel disease.
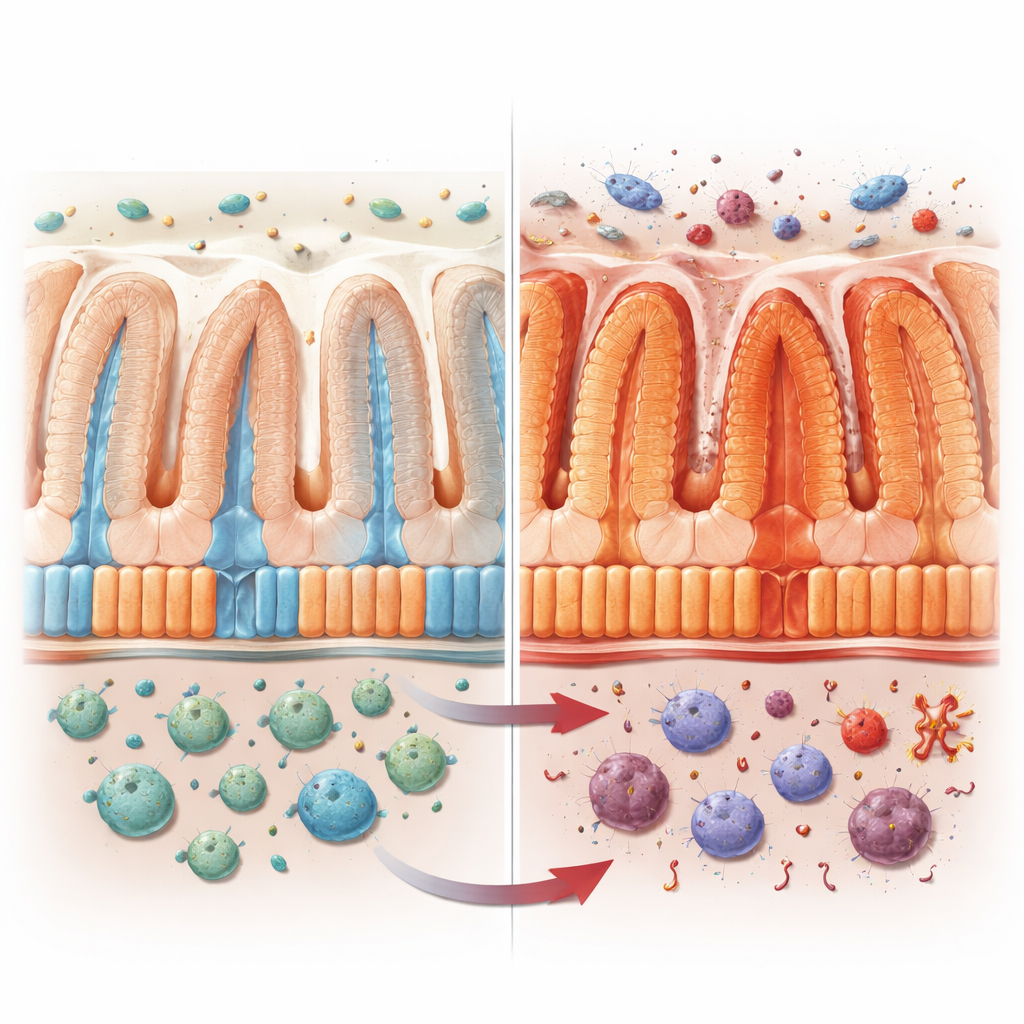
The Everyday Work of Gut Lining Cells
The inner surface of the intestine is lined with enterocytes, cells that make up most of the gut’s barrier. These cells are not all alike. Those deep in the folds focus on defense, making antimicrobial substances, while those nearer the surface specialize in absorbing sugars, fats, vitamins, and other nutrients. Together they create a “zoned” assembly line that keeps the body both nourished and protected. The authors set out to understand how VRAC, a volume-sensing ion channel in these cells, contributes to this division of labor, especially during inflammation.
What Happens When the Gate Fails
To probe VRAC’s role, the researchers engineered mice that lacked its key component, LRRC8A, only in intestinal lining cells. Under normal conditions these animals looked healthy. But when exposed to chemicals that mimic colitis or drive colon cancer, they lost more weight, had shorter, more damaged colons, and developed more tumors than their normal littermates. Their gut barriers leaked, tight junctions between cells were disrupted, and genes linked to inflammation and antimicrobial defenses surged, while genes for nutrient and fat transport dropped. Removing VRAC from immune cells did not produce the same effects, pinpointing the intestinal lining as the critical site.
Shifting from Feeding to Fighting
Single-cell analyses and miniature “gut-in-a-dish” organoids revealed what was happening inside the tissue. Without VRAC, enterocytes failed to mature properly along the crypt-to-surface axis. Cells associated with nutrient uptake, especially at the surface, were depleted, while defense-oriented cells at the base expanded. This rewiring hurt nutrient handling: organoids from VRAC-deficient mice absorbed less lipid and glucose. A central casualty of this shift was vitamin A processing. Enzymes that convert dietary vitamin A into retinoic acid—a signaling molecule that supports enterocyte identity, tissue repair, and immune balance—were sharply reduced, and overall retinoic dehydrogenase activity fell.
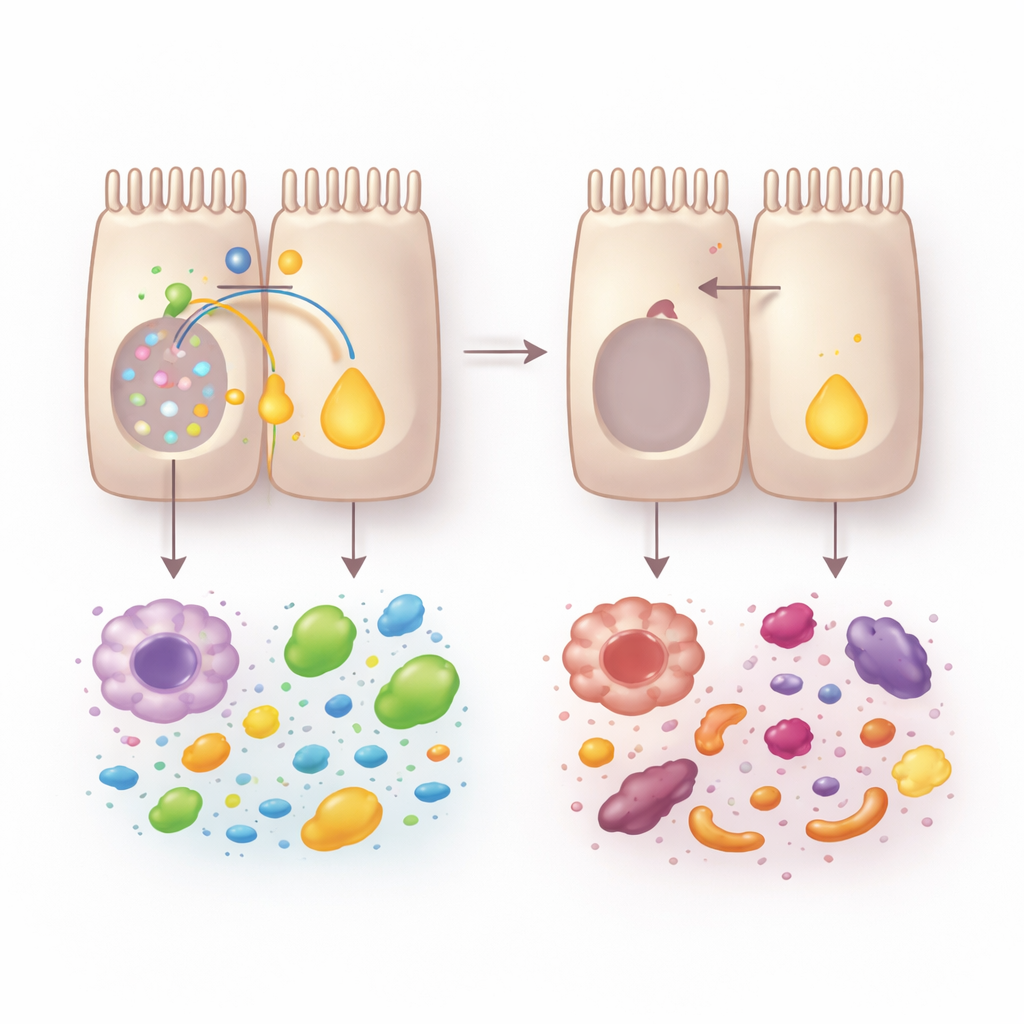
Vitamin A Signals and Friendly Microbes to the Rescue
Because retinoic acid emerged as a key pathway, the team tested whether restoring it could counteract the damage. Giving mice all-trans retinoic acid before colitis reduced weight loss and colon injury, revived vitamin A–processing enzymes, and improved the presence of mature nutrient-absorbing enterocytes. It also restored helpful immune cells that produce IL-17 and IL-22, molecules crucial for repairing the gut lining. At the same time, loss of VRAC altered the microbiota: diversity fell, and beneficial Lactobacillus species declined. These microbial changes proved important; equalizing microbes by co-housing mice or wiping them out with antibiotics erased much of the difference between normal and VRAC-deficient animals. Transplanting stool from VRAC-deficient mice into healthy recipients made colitis worse, while supplementing specific Lactobacillus strains boosted vitamin A metabolism, normalized antimicrobial peptide levels, and revived protective immune cells.
What This Means for Gut Health
Overall, the study shows that VRAC in intestinal lining cells acts like a rheostat, tuning how much effort the gut invests in absorbing nutrients versus fighting microbes. When VRAC is missing, the system locks into high-defense mode: antimicrobial activity and inflammation rise, nutrient absorption and vitamin A signaling fall, and beneficial bacteria are pushed out. By restoring retinoic acid or reintroducing friendly Lactobacillus, the researchers could rebalance this system and ease disease in mice. For people with conditions like inflammatory bowel disease, these findings highlight the gut lining’s own ion channels—and their links to diet-derived vitamins and probiotics—as promising levers for future therapies.
Citation: Yi, X., Zhang, S., Gu, X. et al. VRAC coordinates the trade-off between nutrient absorption and antimicrobial defense in enterocytes against inflammation. Nat Commun 17, 3146 (2026). https://doi.org/10.1038/s41467-026-69963-3
Keywords: inflammatory bowel disease, intestinal epithelium, retinoic acid, gut microbiome, probiotics