Clear Sky Science · en
Role of CTGF-LRP1 in impaired healing of cesarean section incisions
Why C-Section Scars Sometimes Don’t Heal Right
As cesarean births become more common worldwide, a quiet complication is affecting many women long after delivery. Some cesarean scars in the womb heal poorly, leaving a small pouch-like defect in the uterine wall called a “niche.” This defect can cause prolonged bleeding, pain, and fertility problems, and may raise risks in future pregnancies. Yet doctors still do not fully understand why some scars heal well while others do not. This study uses cutting-edge single-cell analysis and animal experiments to uncover what goes wrong in the microscopic healing environment of these uterine scars and points to new ways to predict and possibly improve recovery.
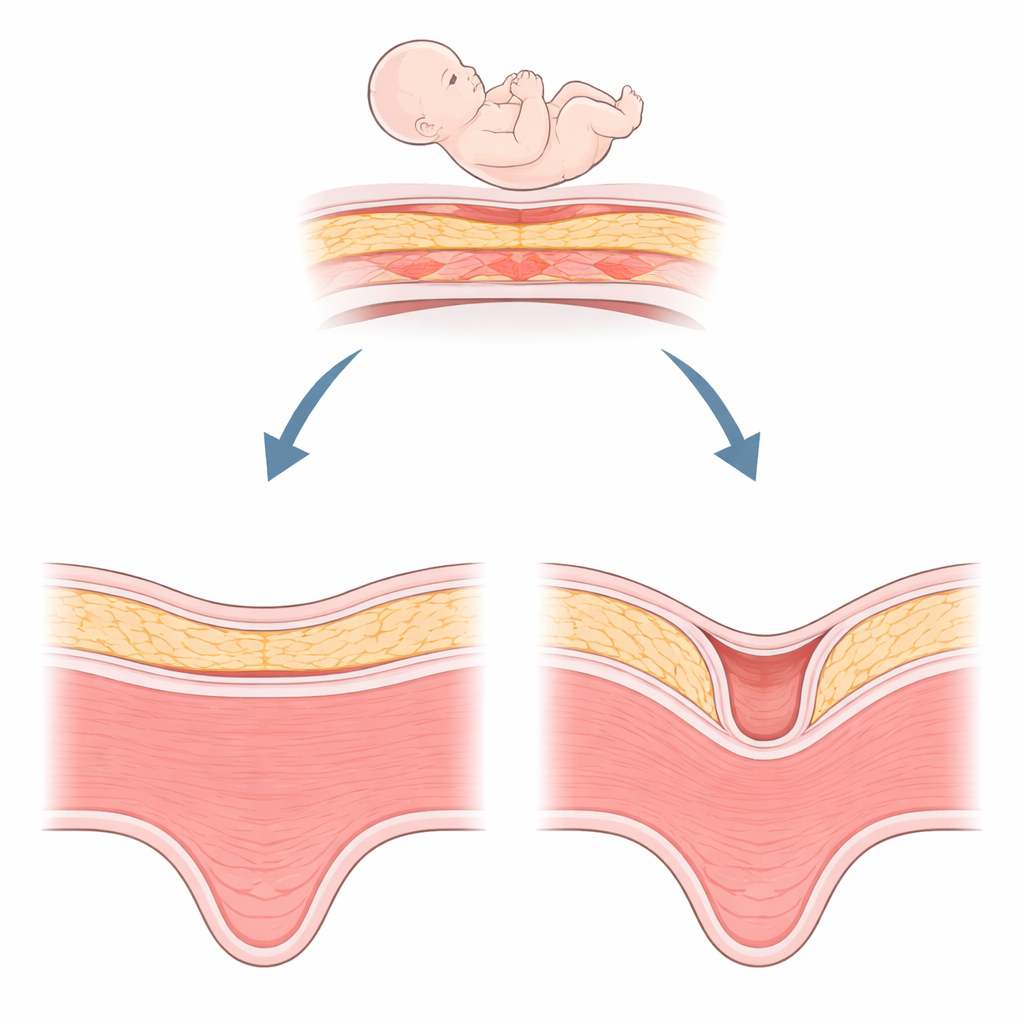
A Hidden Problem After Cesarean Birth
A niche is essentially a dent in the muscle of the uterus at the site of a previous cesarean incision, at least two millimeters deep. It is surprisingly common—seen in roughly half of women after cesarean section—and its detection is increasing with better ultrasound and MRI imaging. Women with a niche may notice spotting after their period, chronic pelvic discomfort, or difficulty becoming pregnant. In later pregnancies, a niche can be linked to dangerous problems such as abnormal placenta attachment or uterine rupture. Surgeons can try to trim and repair this defect, but healing is not always satisfactory. This has left clinicians searching for biological explanations beyond surgical technique alone.
Zooming In on Individual Cells in the Scar
The researchers collected tissue from three areas in women undergoing surgery: well-healed cesarean scars, poorly healed niche tissue, and nearby apparently normal uterine muscle. They then used single-cell RNA sequencing, a technology that reads which genes are active in thousands of individual cells, to map the cellular landscape of these regions. In total, they profiled more than 135,000 cells and identified eight major types, including smooth muscle cells, immune cells, blood vessel cells, and fibroblasts—the connective tissue cells that build the scaffolding of a scar. Compared with well-healed scars, niche tissue contained more fibroblasts but fewer endothelial cells, which line blood vessels and help coordinate tissue repair.
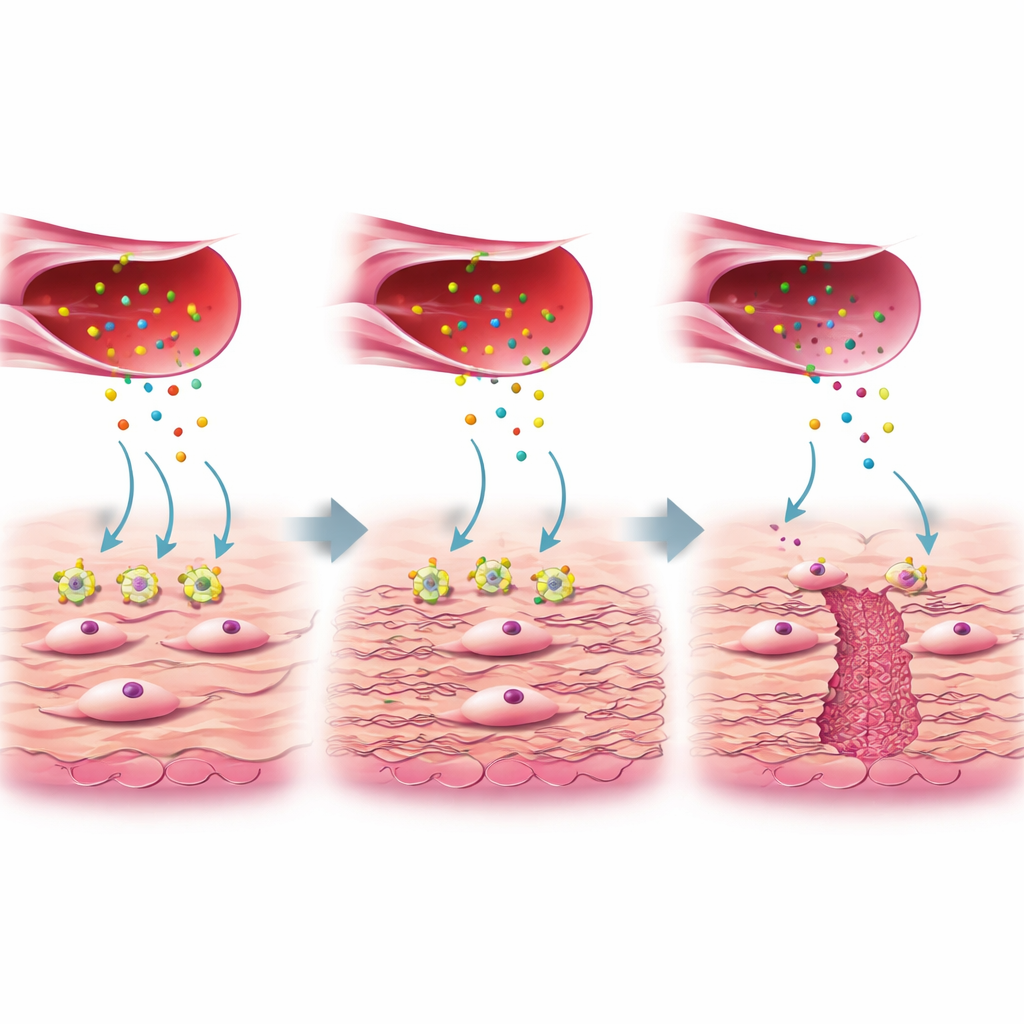
The Missing Signal Between Vessels and Scar Builders
A closer look revealed that not all fibroblasts are alike. One subgroup, called FB3 in the study, showed gene activity linked to collagen production and wound healing and was abundant in well-healed scars but reduced in niche tissue. These helpful FB3 cells normally respond to a protein signal called connective tissue growth factor (CTGF), which is released by a special subgroup of activated endothelial cells. CTGF binds to a receptor on fibroblasts named LRP1. In healthy healing, this CTGF–LRP1 interaction switches on internal pathways (ERK and WNT) that tell fibroblasts to lay down sturdy collagen and other extracellular matrix components, filling in and strengthening the uterine wall. In niche tissue, however, LRP1 levels on FB3 fibroblasts were markedly lower, even though CTGF production from blood vessel cells was similar. The authors showed in cell cultures that when LRP1 was reduced or blocked, fibroblasts failed to respond to CTGF, produced less collagen, and the key repair pathways were blunted.
From Lab Bench to Patients and Animal Models
The team then asked whether this signaling pair could help forecast how well a surgical repair would heal. In samples from 60 women who had transvaginal niche repair, those whose scars later healed well had higher CTGF and LRP1 levels than those whose scars remained problematic, and the combination of both markers best predicted outcome. To test whether boosting CTGF could aid healing, the researchers used a rat model in which a controlled injury was made in the uterus. Rats treated with recombinant human CTGF developed thicker muscle layers, less dense collagen scarring, and better-organized tissue around the wound than untreated animals. Fibroblasts from these rats also produced more collagen when exposed to CTGF—unless LRP1 was experimentally knocked down, again underscoring the importance of the CTGF–LRP1 partnership.
What This Means for Future Mothers
Taken together, the findings paint a picture of niche formation as a breakdown in communication between blood vessels and scar-building cells at the microscopic level. When fibroblasts lack enough LRP1, they cannot properly “hear” CTGF’s call to rebuild, leaving a weak, thinned area in the uterine wall instead of a smooth, solid scar. This work suggests that measuring CTGF and LRP1 around the time of cesarean section or niche repair might one day help doctors predict who is at risk for poor healing. It also raises the possibility that targeted treatments—such as local CTGF delivery or drugs that safely boost the downstream repair pathways—could improve outcomes. While such therapies require careful testing for safety and long-term effects, the study offers a detailed roadmap for turning basic cellular insights into better care for women after cesarean birth.
Citation: He, C., Ge, S., Xia, W. et al. Role of CTGF-LRP1 in impaired healing of cesarean section incisions. Nat Commun 17, 3278 (2026). https://doi.org/10.1038/s41467-026-69747-9
Keywords: cesarean scar defect, wound healing, fibroblasts, growth factor signaling, uterine niche