Clear Sky Science · en
Surface hydroxyl-induced Pt0 clusters on TiO2 for synergistic water gas shift catalysis
Turning Exhaust into Useful Fuel
The water gas shift reaction quietly underpins many clean-energy technologies, helping turn carbon monoxide from industrial gases into extra hydrogen fuel and safer carbon dioxide. This study reports a new way to fine‑tune a common platinum–titania catalyst so it carries out that reaction far more efficiently, by carefully controlling tiny hydroxyl groups—essentially “chemical handles” made of oxygen and hydrogen—on the surface of the support material.
Why This Reaction Matters
Modern hydrogen production and carbon capture often rely on the water gas shift reaction, where carbon monoxide (CO) reacts with water (H2O) to form carbon dioxide (CO2) and hydrogen (H2). Metals such as platinum are excellent at speeding up this chemistry, especially when they are dispersed as very small particles on oxides like titanium dioxide (TiO2). However, the exact way in which the metal and support interact can either help or hinder performance. If platinum becomes too oxidized, it struggles to grab CO; if the oxide surface lacks the right defects, water is slow to split—both of which limit how much hydrogen can be produced.
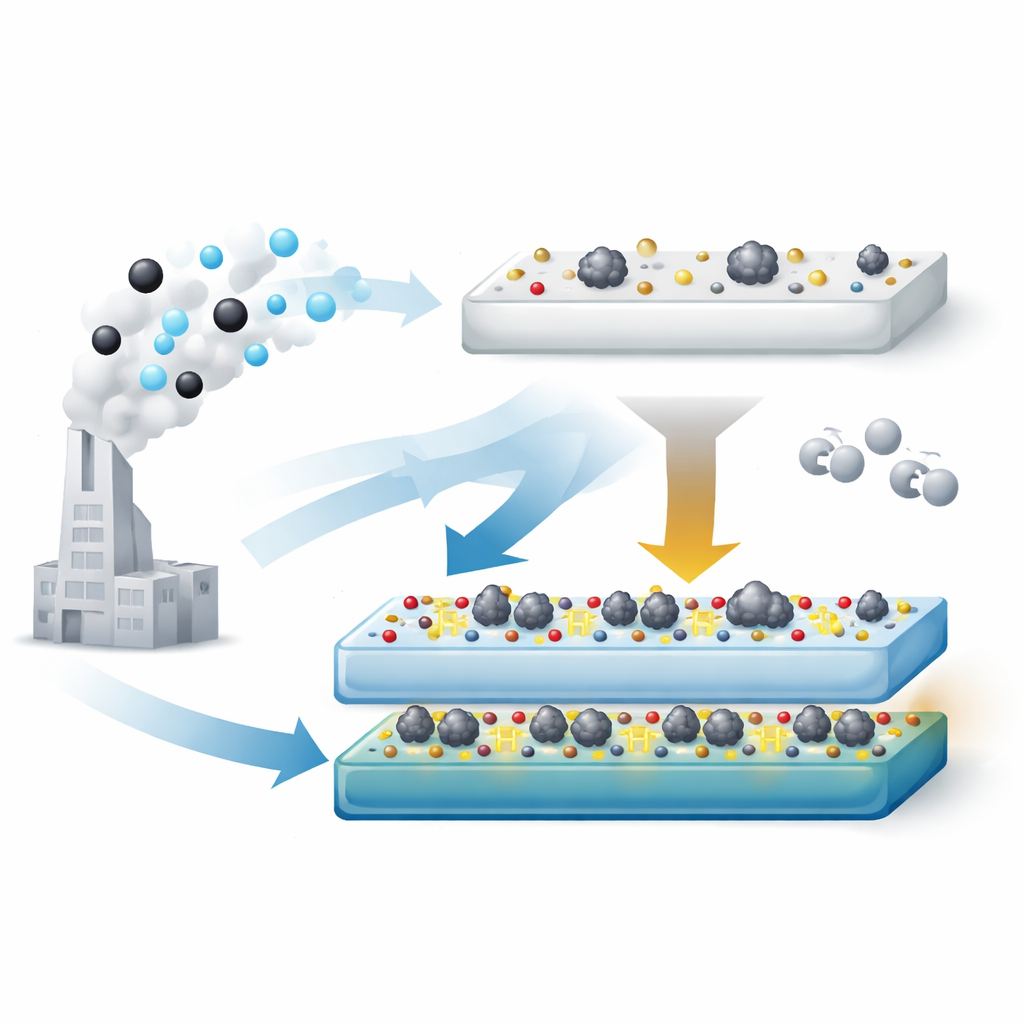
Using Surface “Handles” to Sculpt the Catalyst
The authors devised a two‑step process to engineer the TiO2 support so that it carries an optimal amount of surface hydroxyl groups. First, they convert commercial TiO2 into a titanate phase rich in hydroxyls using a strong alkali treatment. Then, by heating this material in air at different temperatures, they gradually remove some hydroxyls and reshape the solid. At 500 °C they obtain a support (called TiO2‑T‑500) that still carries more hydroxyls than standard TiO2, but is no longer in a tube‑like form that tends to bury the metal. When platinum is added and the catalyst is used, it forms tiny metallic clusters around 2 nanometers in size that stay accessible on the surface.
Making Platinum Better at Grabbing CO
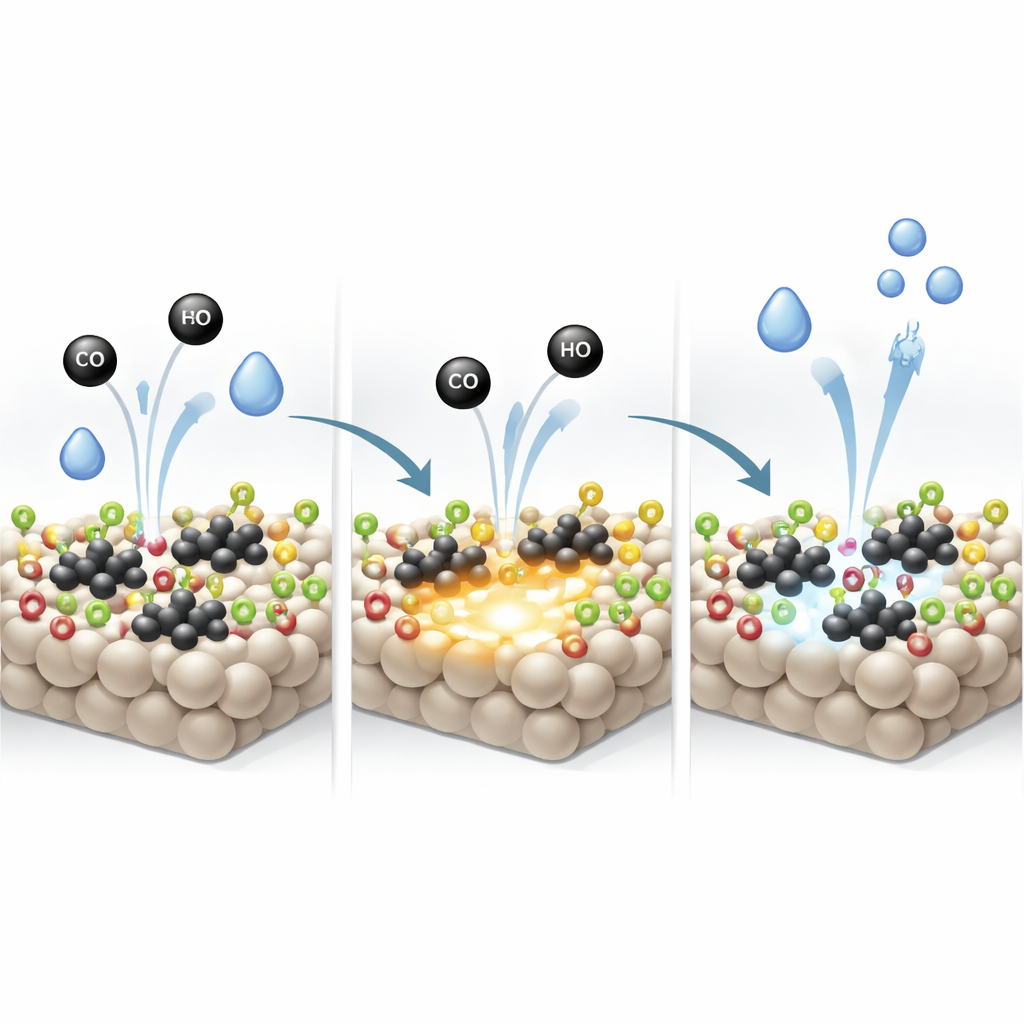
Through a series of spectroscopic measurements, the team shows that these extra surface hydroxyls help drive platinum into a more metallic state (Pt0) after hydrogen treatment, compared with the more oxidized Ptδ+ state on ordinary TiO2. Metallic platinum binds CO more strongly and activates it more readily, which the researchers confirm by monitoring how CO sticks to and leaves the surface under reaction‑like conditions. On the hydroxyl‑rich support, CO remains attached to platinum clusters even after long purging, and reacts more quickly with incoming water to form CO2. Sites near the interface between platinum and TiO2 are especially active, suggesting that both the metal and support participate in the critical steps.
Hydroxyls as Sacrificial Helpers
The hydroxyl groups on TiO2 are not just passive spectators. The study reveals that CO can directly consume these hydroxyls, producing hydrogen and CO2 even before the main reaction fully starts. As this happens, oxygen atoms are removed from the surface, leaving behind oxygen vacancies—missing oxygen sites that act as powerful anchors for splitting new water molecules. Evidence from Raman, infrared, and electron‑spin measurements all points to a strong increase in such vacancies on the engineered support once the reaction is underway. These defects, regenerated and refilled as water repeatedly breaks apart, give the catalyst an enhanced ability to activate H2O without changing the overall reaction pathway.
Faster Reaction and Lasting Stability
Because the hydroxyl‑rich support simultaneously boosts CO activation on metallic platinum and water activation at oxygen vacancies, the overall water gas shift reaction becomes much more efficient. The optimized catalyst achieves reaction rates and per‑site activities roughly two to three times higher than comparable platinum–titania systems reported previously, reaching nearly complete CO conversion at 250 °C. It also remains stable for at least 70 hours of continuous operation, with no loss in performance. In accessible terms, the researchers have found a way to “dial in” just the right amount of surface hydroxyls so that platinum clusters and the oxide surface work together, turning a familiar material into a much more capable engine for clean hydrogen production.
Citation: Wang, CX., Wang, WW., Fu, XP. et al. Surface hydroxyl-induced Pt0 clusters on TiO2 for synergistic water gas shift catalysis. Nat Commun 17, 2757 (2026). https://doi.org/10.1038/s41467-026-69612-9
Keywords: water gas shift, hydrogen production, platinum catalyst, titania support, surface hydroxyls