Clear Sky Science · en
Rational design and in vivo validation of capsid inhibitors for enterovirus D68
Why this matters for parents and doctors
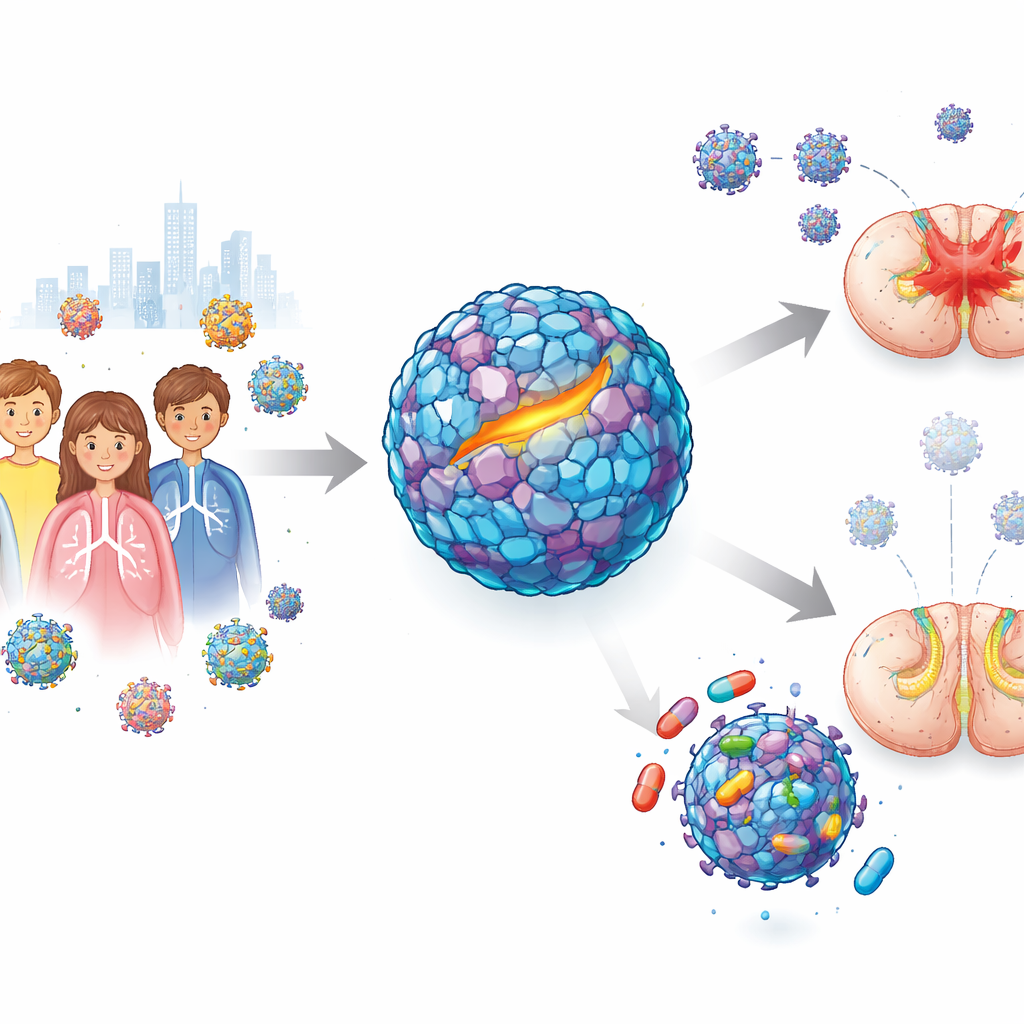
Every few years, a little-known germ called enterovirus D68 sweeps through communities, mostly infecting children. For most, it looks like a bad cold. But in rare cases it attacks the spinal cord, leaving children with sudden, polio-like weakness or even permanent paralysis. There is no approved vaccine or antiviral drug for this virus. This study describes how scientists designed and tested two experimental medicines that block the virus’s outer shell, protect the spinal cord in mice, and may point the way toward future treatments for these frightening episodes.
From mild colds to sudden weakness
Enterovirus D68 usually spreads through coughs and sneezes and causes mild breathing problems. During a large outbreak in 2014, however, doctors noticed a disturbing pattern: some infected children developed acute flaccid myelitis, a condition where the arms or legs quickly become weak or limp. Studies have shown that newer strains of the virus are better able to infect nerve cells than older ones, raising concern that this pathogen has the potential to cause wider and more severe outbreaks. Despite years of effort, earlier drug candidates that looked promising in lab dishes failed to protect animals, especially in models that mimic the neurologic disease. That left an urgent gap: can a drug actually stop this virus once it has already begun to attack the nervous system?
Designing a better lock for the virus shell
The virus is wrapped in a sturdy protein shell, or capsid, that protects its genetic material and helps it latch onto human cells. One of these shell proteins, called VP1, contains a narrow groove known as a canyon, which normally holds a greasy molecule that helps keep the shell stable and ready to infect cells. Previous drugs tried to wedge into this groove but did not work well enough in animals. In this study, the researchers used computer-based screening to search millions of candidate molecules for those that might fit tightly into the canyon. They then refined the best hits through medicinal chemistry, tuning their shapes and chemical groups to grip the pocket more firmly and survive longer in the body. This process yielded two standout compounds, named Jun11695 and Jun11787, which powerfully stopped the virus from damaging cells in culture at extremely low concentrations.
Seeing the drug at work and mapping resistance
Using high-resolution cryo–electron microscopy, the team visualized how the new compounds nestle deep inside the canyon pocket on the viral shell, making many snug contacts with surrounding capsid proteins. This binding appears to lock the shell in a stable form that prevents the virus from carrying out the early steps of infection. Time-of-addition experiments confirmed that the drugs are most effective when present before or during virus entry into cells, but not later, consistent with a capsid-targeting action. When the virus was repeatedly grown in the presence of one compound, Jun11695, rare mutants emerged that were less sensitive to the drug. These mutations mapped to specific positions on the shell proteins that touch the compound, helping confirm the mechanism while also showing that resistance comes with some cost to viral fitness.
Protecting the spinal cord in a mouse model
The crucial question was whether these designer compounds could prevent paralysis in living animals. The researchers used a well-established mouse model in which newborn pups receive the virus in a leg muscle and then develop progressive hind-limb weakness as the virus spreads to the spinal cord and kills motor neurons. Both compounds were absorbed well after injection and stayed in the bloodstream for many hours. When treatment began immediately after infection, mice receiving daily doses of either Jun11695 or Jun11787 showed no sign of paralysis and gained more weight than untreated littermates. Even when dosing was delayed by 24 hours or started only after the first clear signs of limb weakness, the drugs significantly reduced paralysis scores compared with controls. Jun11787, in particular, lowered virus levels in both muscle and spinal cord below the detection limit and preserved motor neurons in the spinal cord, leaving treated animals essentially free of weakness.
What this means for future outbreaks
For families and clinicians, the key message is hopeful but cautious. These experiments show that it is possible to design small molecules that directly target the virus shell, reach the nervous system, and meaningfully blunt or even prevent paralysis—at least in mice. Jun11695 and Jun11787 are early-stage leads, not ready for human use, and important hurdles remain, including testing oral forms and guarding against resistance. Still, the work firmly validates the viral shell as a drug target for enterovirus D68 and offers a concrete blueprint for medicines that might one day protect children facing this rare but devastating form of paralysis.
Citation: Li, K., Rudy, M.J., Klose, T. et al. Rational design and in vivo validation of capsid inhibitors for enterovirus D68. Nat Commun 17, 3052 (2026). https://doi.org/10.1038/s41467-026-69351-x
Keywords: enterovirus D68, acute flaccid myelitis, antiviral drug design, viral capsid inhibitors, neurological infection