Clear Sky Science · en
Inhibition of the cancer stem cell immune checkpoint SOAT1 suppresses regulatory T cell functions through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 pathway in mice
Turning Cancer’s Cholesterol Tricks Against It
Cancer treatment has been transformed by immunotherapy, yet many patients still see little benefit because their tumors find ways to hide from the immune system. This study reveals an unexpected accomplice in that hide‑and‑seek game: a cholesterol‑handling enzyme inside cancer stem cells. By blocking this enzyme with a new experimental drug, the researchers found a way to wake up the body’s defenses and make stubborn tumors in mice far more vulnerable to treatment.
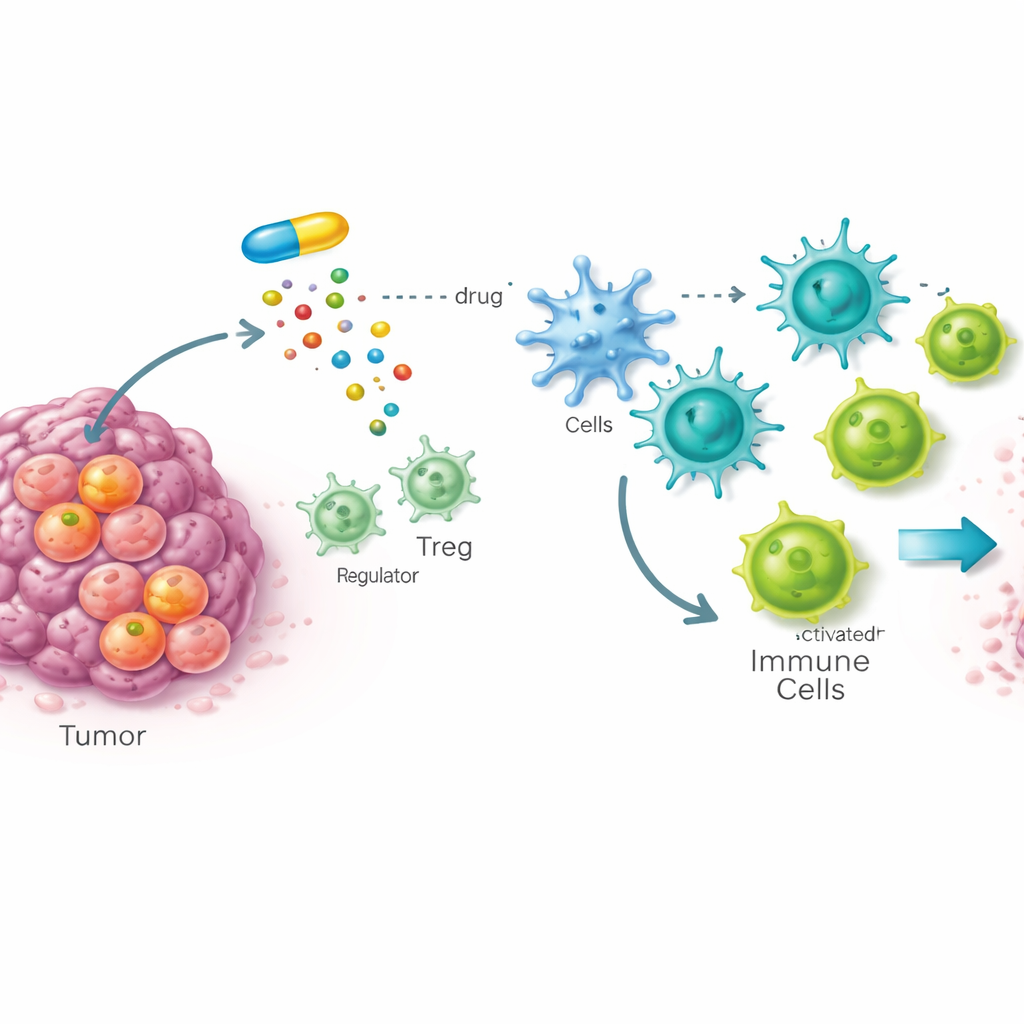
A Hidden Weak Spot in Cancer Stem Cells
Many tumors are driven by cancer stem cells, a small group of cells that can regenerate the tumor and often resist therapy. The team built a whole‑animal drug screen in fruit flies carrying stem cell–like tumors to look for compounds that selectively harm these dangerous cells while sparing normal tissue. From nearly ten thousand candidates, one compound, called STK, stood out. Follow‑up work in mouse and human cancer cells showed that STK binds tightly and specifically to an enzyme named SOAT1, which helps package excess cholesterol into storage droplets. Importantly, SOAT1 turned out to be much more abundant in cancer stem cells than in ordinary cells, and its presence was linked to advanced disease and poorer survival in several human cancers.
Slowing Tumors Across Many Cancer Types
When the researchers treated mice with STK, tumors shrank or grew far more slowly in a wide range of models, including colon, liver, lung, breast, and skin cancers. In genetically engineered mice that develop cancers in their original organs, STK reduced the number of cancer stem cells, decreased tumor burden, and extended survival. In transplanted tumor models, including patient‑derived tumors grown in humanized mice, STK again curbed growth with little apparent toxicity. Genetic experiments confirmed that these benefits depended on SOAT1: when SOAT1 was already silenced in tumor cells, adding STK brought no further advantage. Altogether, the data point to SOAT1 as a common metabolic “checkpoint” that tumors use to thrive and evade control.
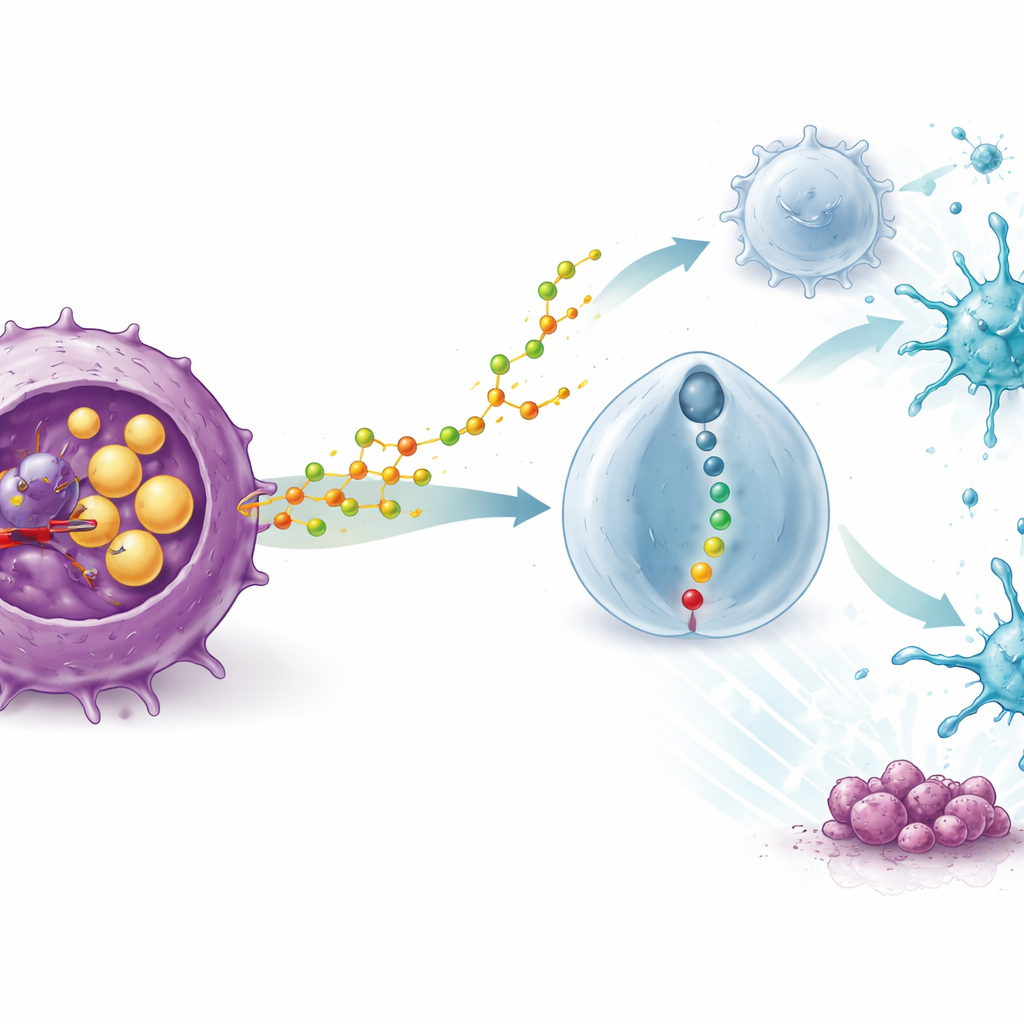
How a Cholesterol By‑Product Rewires Immune Cells
The most striking finding was that STK’s power relied on an intact immune system. In immune‑deficient mice, the drug lost its anti‑tumor punch, and blocking T cells erased its benefits. Single‑cell RNA sequencing of tumor samples showed that STK treatment flooded cancers with helpful immune cells—especially dendritic cells that present tumor material and killer CD8 T cells—while sharply reducing the number and strength of regulatory T cells, or Tregs, which normally act as brakes on immune responses. Mechanistic studies revealed why: blocking SOAT1 in tumor cells caused them to release more of a specific oxidized cholesterol molecule, 20(S)‑hydroxycholesterol. This molecule was taken up by Tregs and activated a receptor on their surface called GPR132, which in turn switched on an internal signaling chain. As this pathway lit up, Tregs became “fragile,” producing fewer suppressive factors and losing their grip on dendritic cells and killer T cells.
From Cold Tumors to Hot Targets
By weakening Tregs in this way, STK effectively rewired the tumor microenvironment from an immune‑silent, or “cold,” state to a highly active, “hot” one. Dendritic cells matured and displayed more activation markers, while CD8 T cells produced higher levels of molecules needed to attack cancer cells. Mice that cleared tumors after STK treatment were better protected against a second tumor challenge, suggesting that lasting immune memory had formed. Crucially, combining STK with existing checkpoint therapies that block PD‑1 or CTLA‑4 further improved tumor control in mouse models, hinting that SOAT1 inhibition could broaden and deepen responses to current immunotherapies.
What This Could Mean for Future Cancer Care
To a non‑specialist, the message is that some cancers shield themselves by reshaping how they use cholesterol, especially within their stem‑like core cells. This work shows that shutting down a key cholesterol‑storage enzyme, SOAT1, not only starves cancer stem cells but also prompts them to release a chemical distress signal that disarms suppressive T cells and rallies the rest of the immune army. In mouse models and early patient‑derived tumors, this strategy makes multiple cancer types more controllable and works especially well alongside existing immunotherapy drugs. While much testing remains before it can be tried widely in people, targeting SOAT1 and the 20(S)‑hydroxycholesterol–GPR132 pathway offers a promising new way to turn the body’s own defenses more reliably against cancer.
Citation: Ding, Y., Fang, W., Xiang, R. et al. Inhibition of the cancer stem cell immune checkpoint SOAT1 suppresses regulatory T cell functions through a trans-cellular 20(S)-Hydroxycholesterol-GPR132 pathway in mice. Nat Commun 17, 4102 (2026). https://doi.org/10.1038/s41467-026-69305-3
Keywords: cancer immunotherapy, cancer stem cells, cholesterol metabolism, regulatory T cells, immune checkpoint therapy