Clear Sky Science · en
SOD1 lactylation impair its enzymatic activity by conformational change to aggravate intervertebral disc degeneration
Why Back Pain Starts So Early
Lower back pain is one of the world’s leading causes of disability, and it often begins surprisingly early in life. At the heart of many cases lies the slow breakdown of the cushions between our vertebrae, known as intervertebral discs. This study uncovers a previously hidden chemical change inside disc cells that links a common metabolic by-product, lactate, to the early aging and failure of these spinal shock absorbers — and it points toward a targeted new treatment strategy.
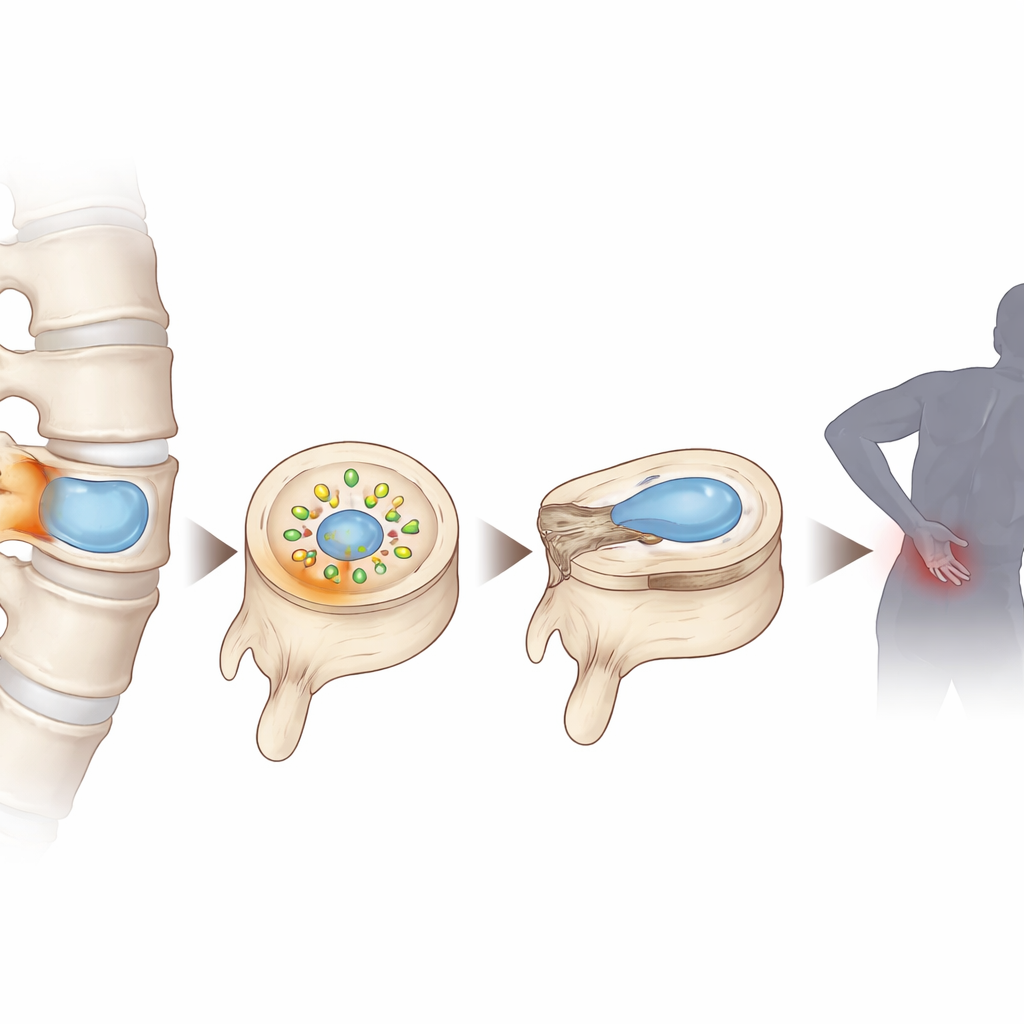
The Hidden World Inside Spinal Cushions
Each intervertebral disc has a soft, gel-like center called the nucleus pulposus, which survives in a low-oxygen, low-nutrient environment because it has almost no direct blood supply. As discs degenerate, this environment becomes even more starved of oxygen and nutrients, causing waste products to build up. By analyzing human disc tissue at different stages of degeneration and combining advanced single-cell sequencing and metabolomics, the researchers found that lactate — the same molecule that accumulates in muscles during intense exercise — rises to unusually high levels inside damaged discs. They also saw that disc cells under these conditions showed signs of oxidative stress, DNA damage, and premature cellular aging.
When Lactate Starts Rewriting Proteins
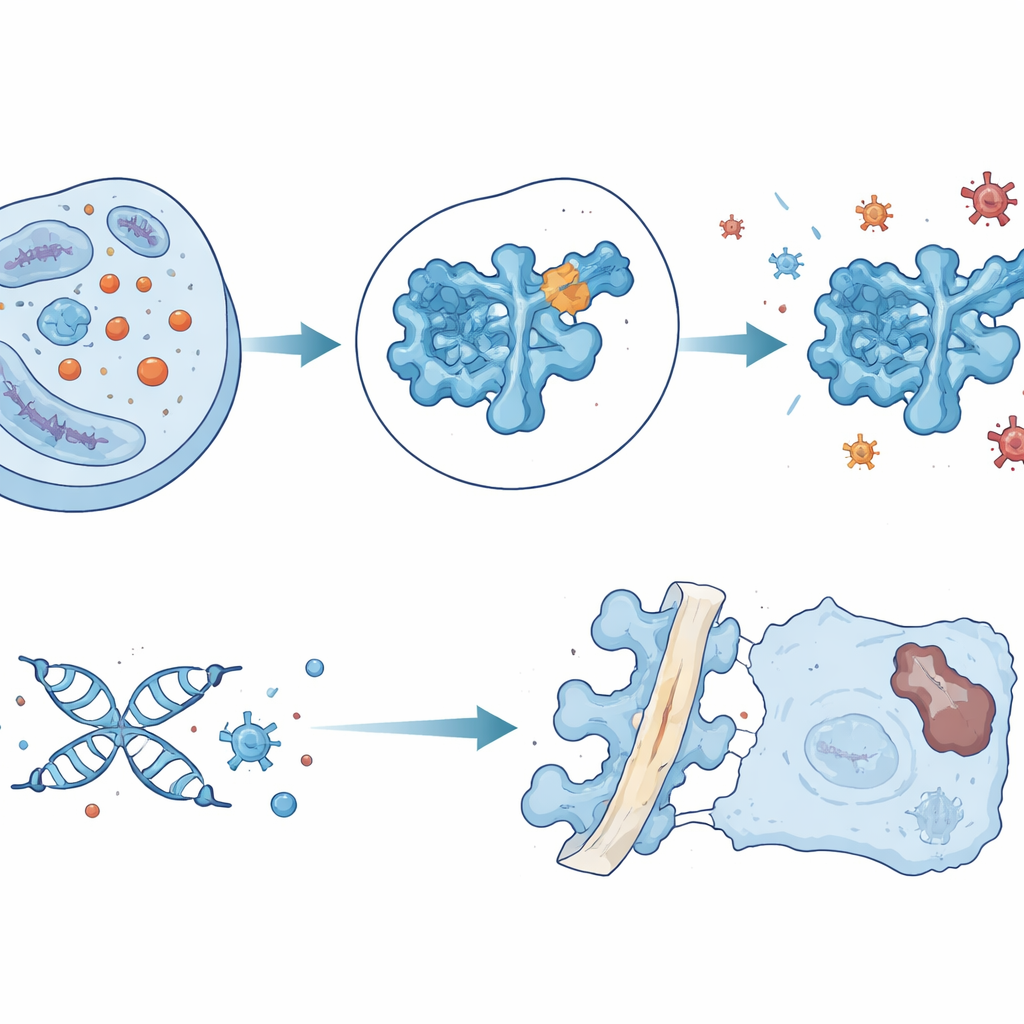
Lactate does more than simply build up: it can chemically modify proteins in a process called lactylation, attaching a small lactate-derived tag to specific sites on them. Using a "lactylome" survey of thousands of protein fragments from rat discs, the team mapped where these tags appear during disc degeneration. Many of the affected proteins are involved in handling reactive oxygen species — unstable molecules that can damage DNA, fats, and proteins. One enzyme in particular, superoxide dismutase 1 (SOD1), stood out. SOD1 normally neutralizes a highly reactive form of oxygen, acting as a crucial antioxidant defense inside disc cells. In degenerating discs, a specific site on SOD1, an amino acid called lysine 123, became heavily lactylated.
How a Tiny Chemical Tag Disarms a Cellular Shield
To understand what this modification does, the researchers created disc cells and rats in which lysine 123 was replaced with another amino acid that cannot be lactylated. These "protected" versions of SOD1 kept their enzyme activity even when lactate levels were high, reduced oxidative stress, and lessened DNA damage and cellular aging in disc cells. In live rats subjected to disc injury, animals carrying the non-lactylatable SOD1 showed milder disc degeneration on MRI and tissue analysis. Computer simulations of SOD1’s 3D structure suggested why: the lactyl tag at position 123 subtly reshapes the enzyme’s surface near its copper-containing active site, weakening its grip on reactive oxygen molecules and making the whole protein less stable.
Turning Off the Damage Switch
The consequences of this structural tweak extend to the disc cell’s genetic control systems. When SOD1 is weakened by lactylation, reactive oxygen accumulates and switches on the p53 pathway, a central regulator of cellular stress and senescence. Cells enter a damage-locked state, no longer functioning properly and contributing to tissue breakdown. By contrast, blocking lactylation at lysine 123 dampens p53 activation and its downstream targets, helping cells maintain redox balance and delay degenerative changes. This positions SOD1 lactylation as a key molecular switch that links metabolic stress to structural failure in intervertebral discs.
A Precision Drug Delivery Idea
Armed with this insight, the team searched through 1.6 million small molecules in silico for compounds that would bind near SOD1’s vulnerable lysine 123 pocket. They identified one, dubbed ZL-01, that interacts directly with that region and reduces SOD1 lactylation without noticeably affecting other common protein modifications. In cultured disc cells, ZL-01 restored SOD1 activity, lowered oxidative stress, and reduced markers of DNA damage and senescence. Injected into injured rat discs, it slowed degeneration, but the drug was cleared quickly from the tissue. To boost staying power and targeting, the researchers loaded ZL-01 into tiny extracellular vesicles engineered with a peptide that binds strongly to collagen type II, a major component of the disc center. This delivery system kept ZL-01 in the disc longer, further decreased SOD1 lactylation and oxidative stress, and significantly improved disc structure and function in the animals.
What This Means for People with Back Pain
Taken together, the work reveals a chain of events in which lactate buildup inside poorly nourished discs chemically disarms a key antioxidant enzyme, SOD1, triggering oxidative damage, stress signaling, and accelerated disc degeneration. By showing that blocking a single lactylation site can protect disc cells and by designing a targeted small molecule to do just that, the study points to a future in which we might treat back pain not just by relieving symptoms or surgically removing damaged tissue, but by directly correcting the underlying molecular missteps that cause discs to wear out too soon.
Citation: Zhang, Y., Zhai, Y., Liu, C. et al. SOD1 lactylation impair its enzymatic activity by conformational change to aggravate intervertebral disc degeneration. Nat Commun 17, 3318 (2026). https://doi.org/10.1038/s41467-026-69127-3
Keywords: intervertebral disc degeneration, lactate, protein lactylation, oxidative stress, SOD1