Clear Sky Science · en
Bioactive lipid-mediated structural and functional regulation of the essential human potassium channel Kir7.1
Why tiny gates in our cells matter
Buried in the membranes of many of our cells are microscopic gates that let potassium ions flow in and out. One of these gates, called Kir7.1, is crucial for healthy vision, brain function, and even the timing of childbirth. When this gate is broken, people can go blind early in life, and animals develop problems with feeding and pregnancy. This study reveals, in atomic detail, how natural fats and hormone-like molecules latch onto Kir7.1 and act as its on–off switches, opening new avenues for drug design.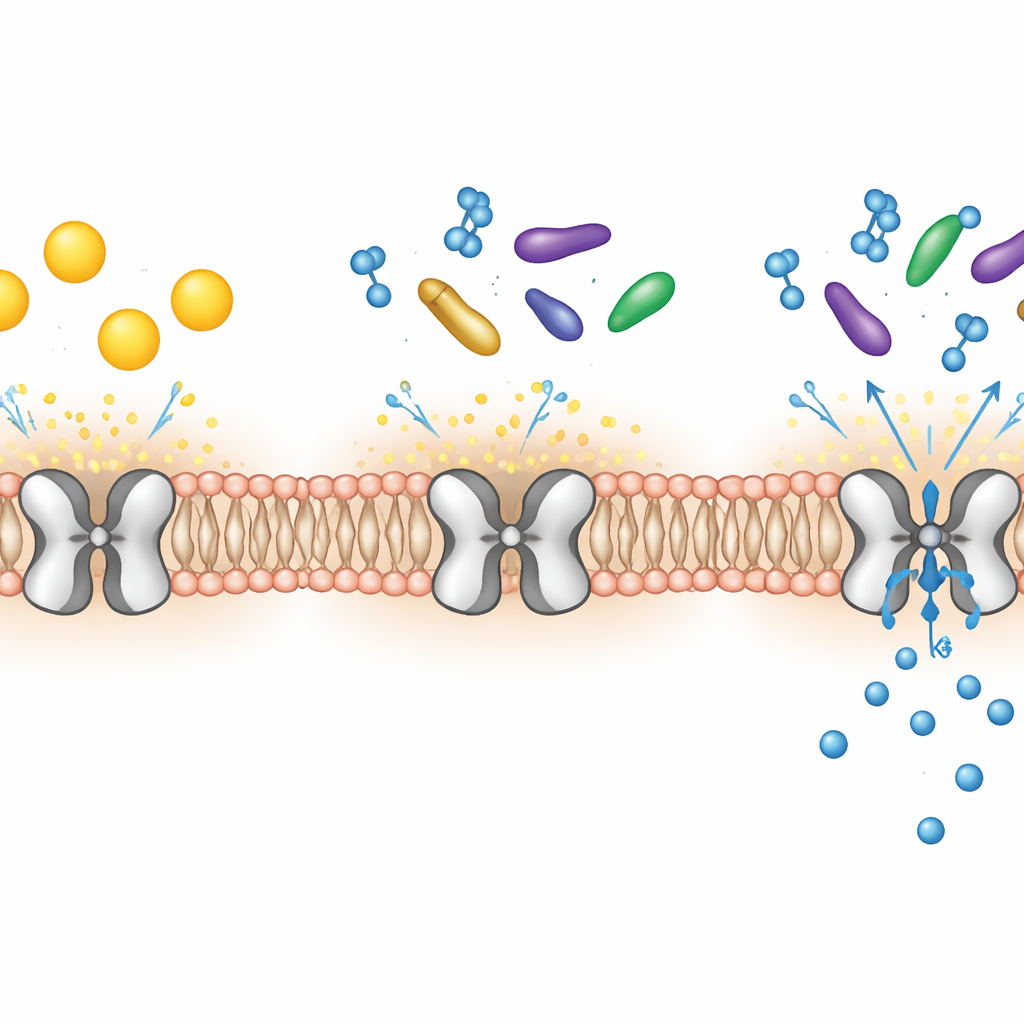
A special gate for electrical balance
Kir7.1 belongs to a family of channels that usually allow potassium ions to move more easily into a cell than out of it, helping to set the electrical balance across the cell membrane. In the eye’s retinal pigment epithelium, Kir7.1 keeps the environment around light-sensing cells stable; without it, photoreceptors slowly die and vision fades. Kir7.1 also shapes nerve circuits that control appetite and helps keep the uterus quiet during pregnancy. These broad roles have made the channel an attractive but poorly understood drug target, especially because its natural regulators, beyond a few known membrane lipids, were mysterious.
Seeing the gate in three distinct poses
To uncover how Kir7.1 works, the researchers produced large amounts of the human protein in mammalian cells, purified it, and imaged it using high-resolution cryo–electron microscopy. They captured the channel in three main shapes. In the “extended” state, the lower, cytoplasmic part of the channel sits further from the membrane and the central tunnel for ions is long and relatively narrow, a likely non-conducting pose. In the “docked” state, this lower domain rotates and moves upward toward the membrane, shortening and slightly widening the tunnel. In a third state, the channel is bound to a powerful synthetic steroid along with a signaling lipid called PIP2, producing a configuration that appears “primed” for opening. A small change in a single amino acid near the selectivity filter helps explain why Kir7.1 can conduct cesium ions, unlike most related channels.
How fats and hormones share control
The structural snapshots revealed that Kir7.1 constantly carries a PIP2 molecule at a conserved site where other family members also bind this lipid. PIP2 alone, however, is not enough to fully open Kir7.1. A second pocket, wedged between its membrane-spanning helices, contains a different kind of lipid. In the resting extended state, this pocket holds cholesterol, a familiar membrane sterol. Electrophysiology experiments showed that adding extra cholesterol dampens Kir7.1 currents, while certain steroids, including the pregnancy hormone progesterone and the neurosteroid DHEA, strongly boost them. The data support a model in which cholesterol acts as a natural brake: activating steroids must first displace cholesterol from the pocket, and when they do so in the presence of PIP2, the channel’s lower domain can rotate and the ion pathway can widen.
Discovering stronger molecular keys
To turn this insight into practical tools, the team screened a wide panel of natural and man-made steroids and their mirror-image forms. They found several new molecules that switch Kir7.1 on, many at low micromolar or even submicromolar concentrations. One standout, an enantiomer of the drug 17-hydroxyprogesterone caproate, bound tightly in the same hydrophobic pocket that normally hosts cholesterol. In the structure with this activator present, the steroid’s polar tip formed a close hydrogen bond with a key threonine side chain, while its oily body nestled against the channel’s helices. This snug fit helps explain its potency and its ability to keep the channel active even in the presence of a standard Kir7.1 blocker.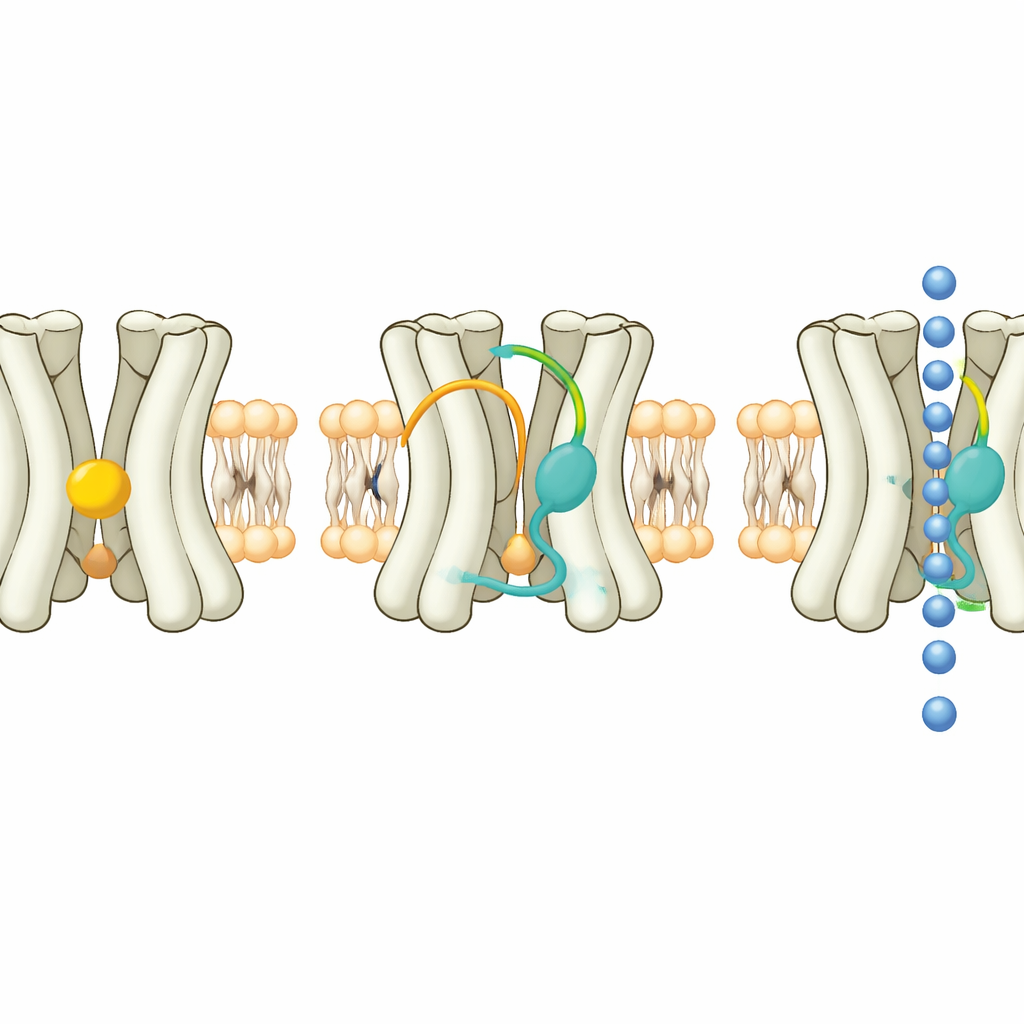
From structural map to therapeutic promise
Together, the work shows that Kir7.1 is governed by a cooperative partnership between PIP2, inhibitory cholesterol, and select activating steroids. PIP2 anchors and partially organizes the channel; cholesterol holds it in an extended, less conductive form; and steroids that evict cholesterol loosen the helices and let the lower domain shift toward a more open, conductive state. By resolving these interactions atom by atom and outlining how small chemical tweaks change steroid potency, the study offers a blueprint for designing new drugs that restore or fine-tune Kir7.1 activity in diseases of the eye, brain, and uterus.
Citation: Niu, Q., Vu, S., Xu, Y. et al. Bioactive lipid-mediated structural and functional regulation of the essential human potassium channel Kir7.1. Nat Commun 17, 2764 (2026). https://doi.org/10.1038/s41467-026-68819-0
Keywords: Kir7.1 potassium channel, cholesterol regulation, steroid modulators, cryo-EM structure, retinal pigment epithelium