Clear Sky Science · en
RUNX1 restrains STAT1-GITRL signaling to shape an immunosuppressive CRC microenvironment
Why this matters for cancer treatment
Colorectal cancer is one of the most common and deadly cancers worldwide, and many patients do not respond well to today’s immunotherapy drugs. This study uncovers a hidden molecular “brake” inside tumor cells that shapes how the immune system behaves in and around colorectal tumors. By understanding this brake, the researchers point to new ways of predicting who might benefit from certain immune‑boosting treatments and how to make those therapies work better.
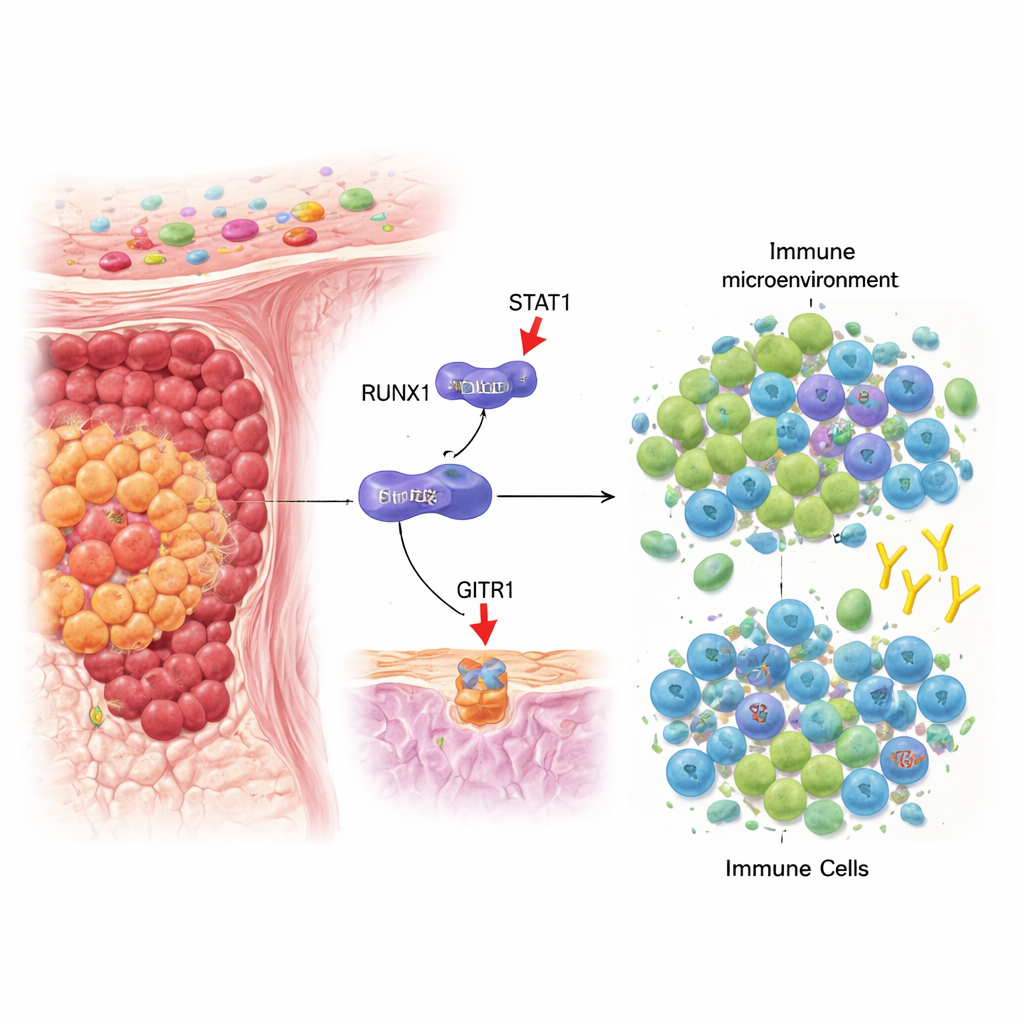
A tug-of-war inside colon tumors
Our immune system constantly patrols for cancer cells, and two key players in this battle are regulatory T cells (Tregs), which calm immune responses, and CD8+ T cells, which can directly kill tumor cells. In colorectal cancer, many tumors develop an environment that favors Tregs over killer T cells, allowing the cancer to grow unchecked. The authors focused on a gene called RUNX1, already known to influence how blood and solid tumors behave, and asked whether it might also affect this immune balance inside colorectal tumors.
A hidden switch that controls immune signals
By analyzing patient tumor samples and large cancer databases, the team found that colorectal tumors with high levels of RUNX1 tended to have low levels of a surface molecule called GITRL and higher numbers of Tregs. GITRL sits on tumor cells and other cells and can interact with a partner molecule, GITR, on T cells. When GITRL engages GITR, it generally weakens Treg activity and boosts CD8+ T‑cell function, tipping the scales toward anti‑tumor immunity. Experiments in cell lines showed that dialing down RUNX1 increased GITRL, while turning RUNX1 up reduced it, suggesting that RUNX1 acts as a negative regulator of this immune‑stimulating signal.
How one protein blocks another
The researchers then asked how RUNX1 lowers GITRL levels. Instead of binding directly to the GITRL gene, RUNX1 physically interacts with another protein called STAT1, a well‑known responder to the immune messenger interferon‑gamma. Normally, when interferon‑gamma is present, STAT1 forms pairs, moves into the nucleus, and switches on genes such as GITRL. The study shows that RUNX1 binds STAT1 in the cell’s fluid compartment, prevents efficient pairing of STAT1, and reduces its movement into the nucleus. As a result, STAT1 has a harder time turning on the GITRL gene, and tumor cells display less GITRL on their surface. This molecular blockage ultimately favors the build‑up of Tregs and weakens CD8+ T‑cell activation in the tumor microenvironment.
From mouse models to therapy potential
To see how this plays out in living organisms, the team used mouse colorectal tumors engineered to alter RUNX1 or GITRL levels. Tumors rich in GITRL grew more slowly, had fewer Tregs, and contained more activated CD8+ T cells. In contrast, tumors with extra RUNX1 grew faster, harbored more Tregs, and had fewer active killer T cells. Importantly, when these RUNX1‑high tumors were treated with an experimental antibody that stimulates GITR—mimicking the effect of GITRL on T cells—the tumors became much more sensitive to therapy and their growth slowed. This suggests that when RUNX1 suppresses tumor‑cell GITRL, it leaves more GITR‑bearing Tregs in place for the drug to target and deplete.
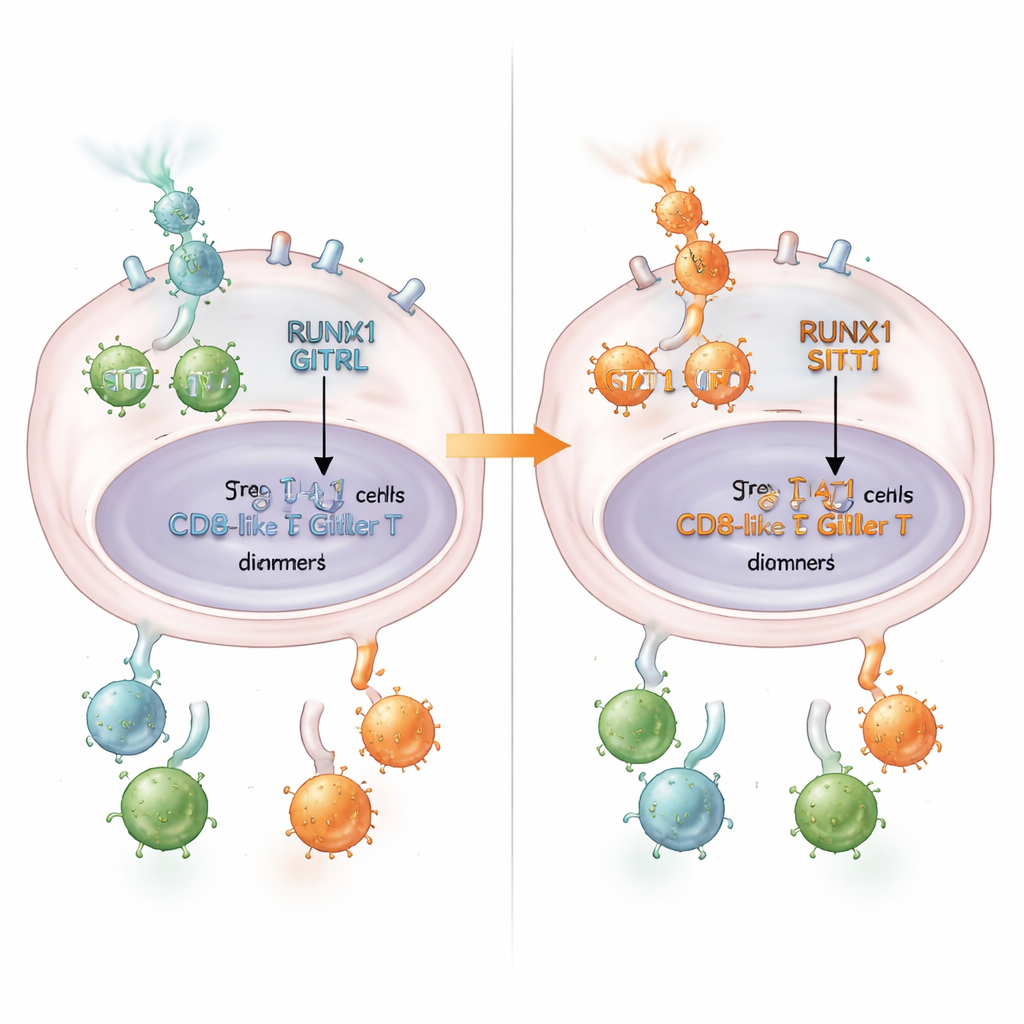
What this means for patients
In accessible terms, this work reveals a chain of events in which RUNX1 acts like a gatekeeper: it holds back STAT1, lowers GITRL, encourages peace‑keeping Tregs, and dampens cancer‑fighting CD8+ T cells. That chain helps explain why some colorectal tumors are more immunosuppressive than others. The findings suggest that measuring RUNX1 and GITRL levels in tumors could help identify patients who are more likely to benefit from drugs that stimulate GITR. In the longer term, therapies that loosen RUNX1’s hold on STAT1—or otherwise restore the RUNX1/STAT1/GITRL axis—might reawaken the immune system inside colorectal tumors and improve the success of immunotherapy.
Citation: He, W., Zheng, L., Huang, W. et al. RUNX1 restrains STAT1-GITRL signaling to shape an immunosuppressive CRC microenvironment. Cell Death Discov. 12, 151 (2026). https://doi.org/10.1038/s41420-026-03053-7
Keywords: colorectal cancer, tumor microenvironment, regulatory T cells, GITR immunotherapy, RUNX1 STAT1 signaling