Clear Sky Science · en
FUT8 reprograms glycolytic metabolism to promote PKM2 lactylation and drive clear cell renal cell carcinoma progression
Why kidney cancer cells change how they burn sugar
Clear cell kidney cancer is one of the most common and deadly forms of kidney cancer. This study explores why these tumors are so addicted to sugar and how a lesser-known enzyme, FUT8, helps them grow, spread, and resist treatment. By tracing how sugar is broken down inside cancer cells and how the resulting waste products can reprogram proteins, the research reveals a new weak point that future drugs might be able to attack.
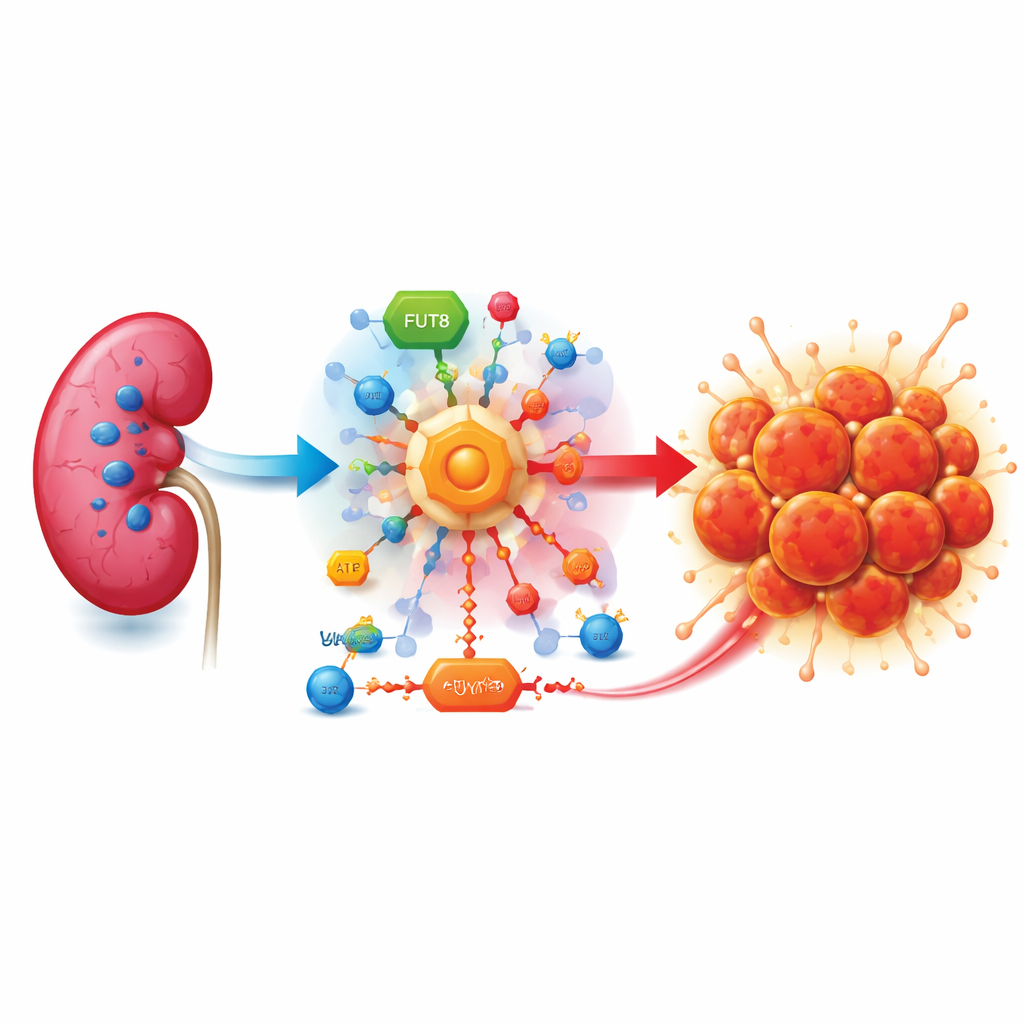
A sugar-hungry tumor
Clear cell renal cell carcinoma often arises when a guardian gene called VHL is lost. Without VHL, cells behave as if they are starved of oxygen, even when oxygen is plentiful. They flip on emergency programs that ramp up sugar burning through a shortcut pathway known as aerobic glycolysis. Instead of fully extracting energy, cancer cells convert much of the sugar into lactate, an acidic by-product that builds up around the tumor and helps it grow, evade the immune system, and spread.
An overlooked sugar decorator steps into the spotlight
The enzyme FUT8 normally adds a small sugar, fucose, onto many cell-surface proteins, tuning how cells respond to growth signals. The authors show that FUT8 itself is strongly elevated in clear cell kidney tumors compared with normal kidney tissue. Patients whose tumors have more FUT8 tend to live for a shorter time, marking FUT8 as a candidate biomarker. When FUT8 is switched off in kidney cancer cells grown in dishes or in mice, tumors grow more slowly, form fewer colonies, move less, and are less able to seed metastases in the lungs. Markers of a more invasive, mobile cell state drop, suggesting that FUT8 helps push tumors toward a shape-shifting, spreading behavior.
Rewiring how cancer cells use fuel
Digging deeper, the researchers discovered that FUT8 boosts the activity of HIF-1α, a master regulator of the low-oxygen response. This, in turn, strengthens glycolysis and weakens the cell’s normal, oxygen-based energy factories in mitochondria. When FUT8 is silenced, cells shift back toward more efficient mitochondrial respiration, their acidification of the surrounding medium falls, and lactate levels inside the cell drop. Knocking down HIF-1α mimics these effects, indicating that FUT8 drives a “Warburg-like” metabolic program largely through this hypoxia pathway. Experiments that add lactate back to FUT8-deficient cells partially restore their growth and movement, showing that lactate is not just waste but an active driver of malignancy.
From waste product to protein switch
The study then asks what lactate actually does inside the cell. Beyond acidifying the environment, lactate can attach to lysine residues on proteins in a modification called lactylation. The authors find that FUT8’s influence on metabolism raises global lactylation levels across many proteins, and that high FUT8 in patient tumors goes hand-in-hand with high overall lactylation. Using a broad protein survey, they identify one key target: PKM2, a central enzyme in glycolysis. In clear cell kidney cancer cells, PKM2 carries a lactylation mark at a specific position, K115, and this mark depends on FUT8 and lactate levels. When the team engineers a version of PKM2 that cannot be lactylated at this site, cancer cells grow more slowly, migrate less, produce less acid, and form smaller tumors in mice. This non-lactylated PKM2 also shows altered activity and location within the cell, linking the chemical tag directly to how the enzyme works.
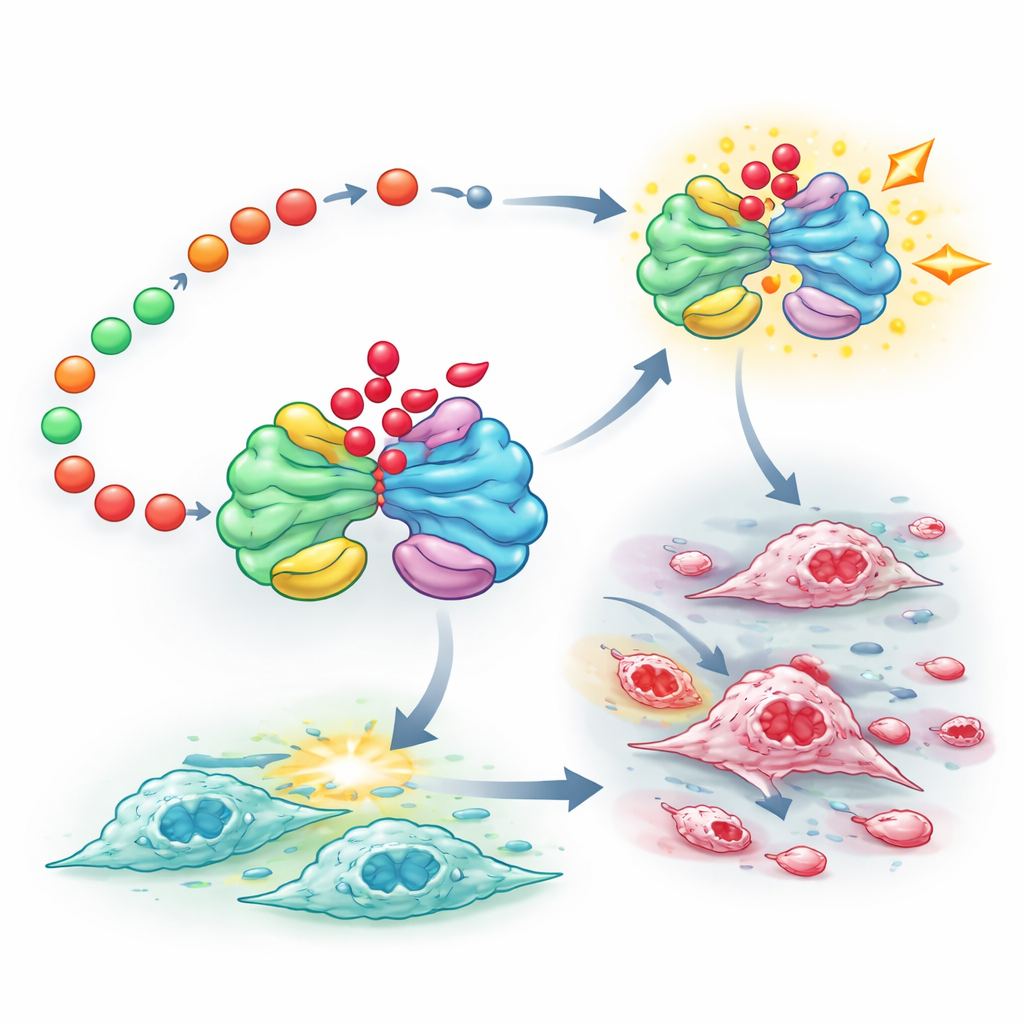
Pairing metabolic and signaling targets
Finally, the researchers test whether blocking both lactate production and FUT8 at the same time can rein in tumors more effectively. In mouse models, a drug that inhibits lactate-forming enzymes and a compound that blocks FUT8 each slow tumor growth on their own, but the combination works best and reduces the overall level of lactylation in tumor proteins. These results suggest that the FUT8–HIF-1α–lactate–PKM2 chain forms a critical backbone that connects altered metabolism to changes in protein behavior and cell identity in clear cell kidney cancer.
What this means for patients
To a layperson, the message is that clear cell kidney tumors co-opt both sugar use and protein decoration to fuel their progression. FUT8 sits at the crossroads of these processes, helping cancer cells overproduce lactate and then use that lactate to flip important protein switches like PKM2. By doing so, it encourages cells to grow faster and become more invasive. Targeting FUT8, lactate production, or the specific lactylation of PKM2 could offer new treatment strategies and help explain why some patients fare worse than others, even when their tumors share the same initial genetic defects.
Citation: Guo, Z., Jiang, H., Wang, X. et al. FUT8 reprograms glycolytic metabolism to promote PKM2 lactylation and drive clear cell renal cell carcinoma progression. Cell Death Discov. 12, 146 (2026). https://doi.org/10.1038/s41420-026-03013-1
Keywords: clear cell renal cell carcinoma, tumor metabolism, lactate signaling, protein lactylation, FUT8