Clear Sky Science · en
Using 3D Invasion properties of RCC Cell Lines In Vitro to predict their Metastatic Potential In Vivo
Why this research matters
When kidney cancer spreads to other organs, it becomes much harder to treat. Doctors would like to know early on which tumors are likely to stay put and which are prone to travel. This study shows that by growing kidney cancer cells in carefully designed three-dimensional mini-tumors in the lab, researchers can forecast how aggressively those cancers will spread in living animals. The work points toward lab tests that might one day spare some patients from unnecessary treatments while flagging others who need fast, intensive care.
Building better mini-tumors
Most cancer research still relies on flat cell layers grown on plastic dishes. These are easy to handle but do a poor job of mimicking how tumors behave inside the body, where cells push through surrounding tissue in three dimensions. In this study, scientists worked with three well-known kidney cancer cell lines, which they already knew carried different genetic and signaling features. They formed each cell type into compact spherical clusters, then embedded these clusters in a soft gel made of collagen and fibronectin that matches the stiffness of real kidney tissue. Over a week, they tracked how far and how fast cancer cells broke away from the spheroid and invaded the surrounding gel, and they mapped the paths of individual cells. 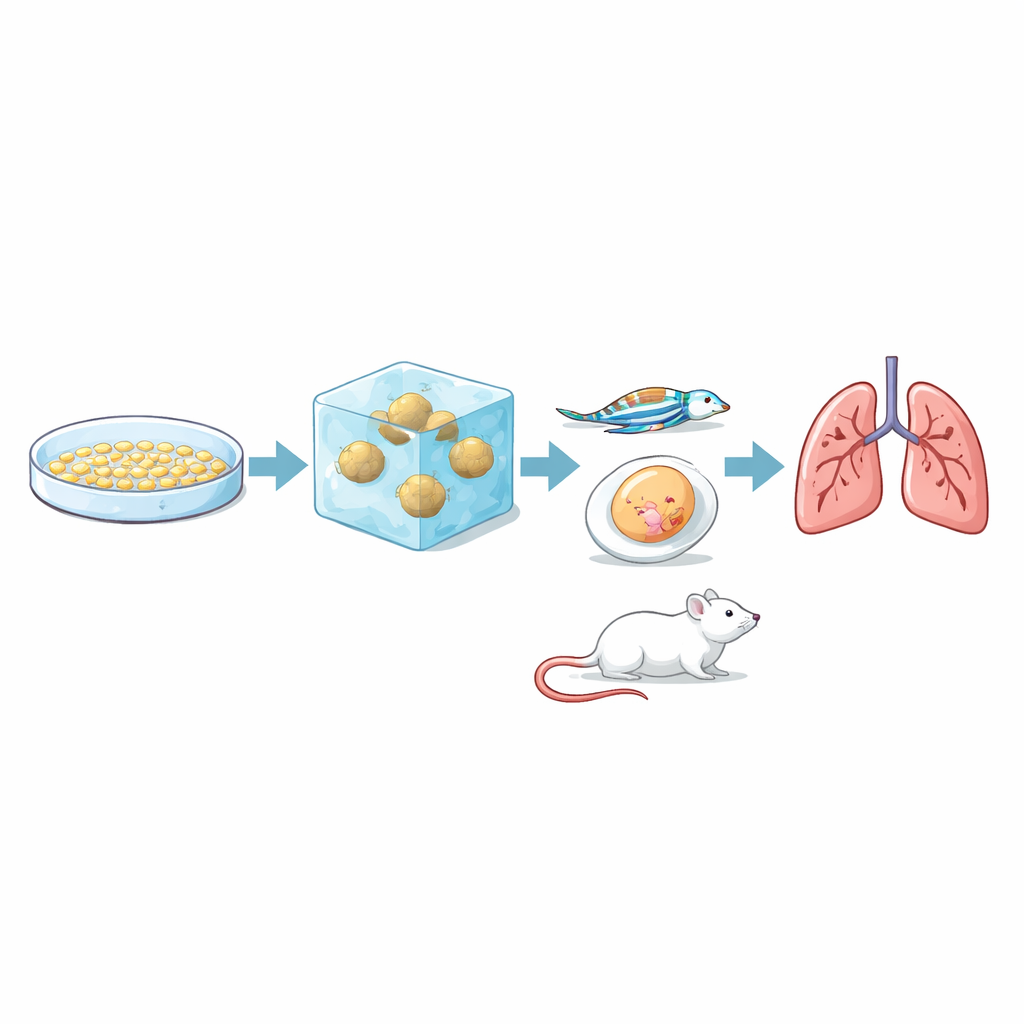
Ranking cancer cells by their urge to invade
The three cell lines showed sharply different behaviors in this 3D setting. One line, called RCC10, stayed mostly as a tight ball with only minimal outward movement. A second line, 786-O, sent cells out into the gel, but only to a moderate degree. The third line, RCC7, was the most aggressive: its cells streamed far into the gel, covered a larger area, and moved at higher speeds. The paths traced by individual RCC7 and 786-O cells were meandering and exploratory, with frequent direction changes, while RCC10 cells tended to move in a straighter, more limited way. Blocking a key signaling route (the TGFβ pathway) in the more invasive spheroids significantly reduced their ability to escape into the gel, confirming that the model responds to drugs that target invasion-related mechanisms.
Testing predictions in living animals
The crucial question was whether this lab-based invasion test actually reflects how tumors behave in real organisms. To find out, the team injected the same three cell lines into several animal models that capture different stages of metastasis. In chicken egg membranes, all three formed tumors, but RCC7 produced larger, blood-filled, and more varied masses. In transparent zebrafish embryos, researchers watched fluorescently tagged cancer cells travel through blood vessels: RCC7 and 786-O cells stuck to vessel walls and slipped out into surrounding tissue more often than RCC10 cells. In mice, RCC7 and 786-O cells reliably formed metastases in the lungs, with RCC7 doing so faster and more extensively, while RCC10 produced no detectable lung metastases within the study period. Taken together, the ranking seen in the 3D gel—RCC7 most invasive, 786-O intermediate, RCC10 least—matched their behavior in animals. 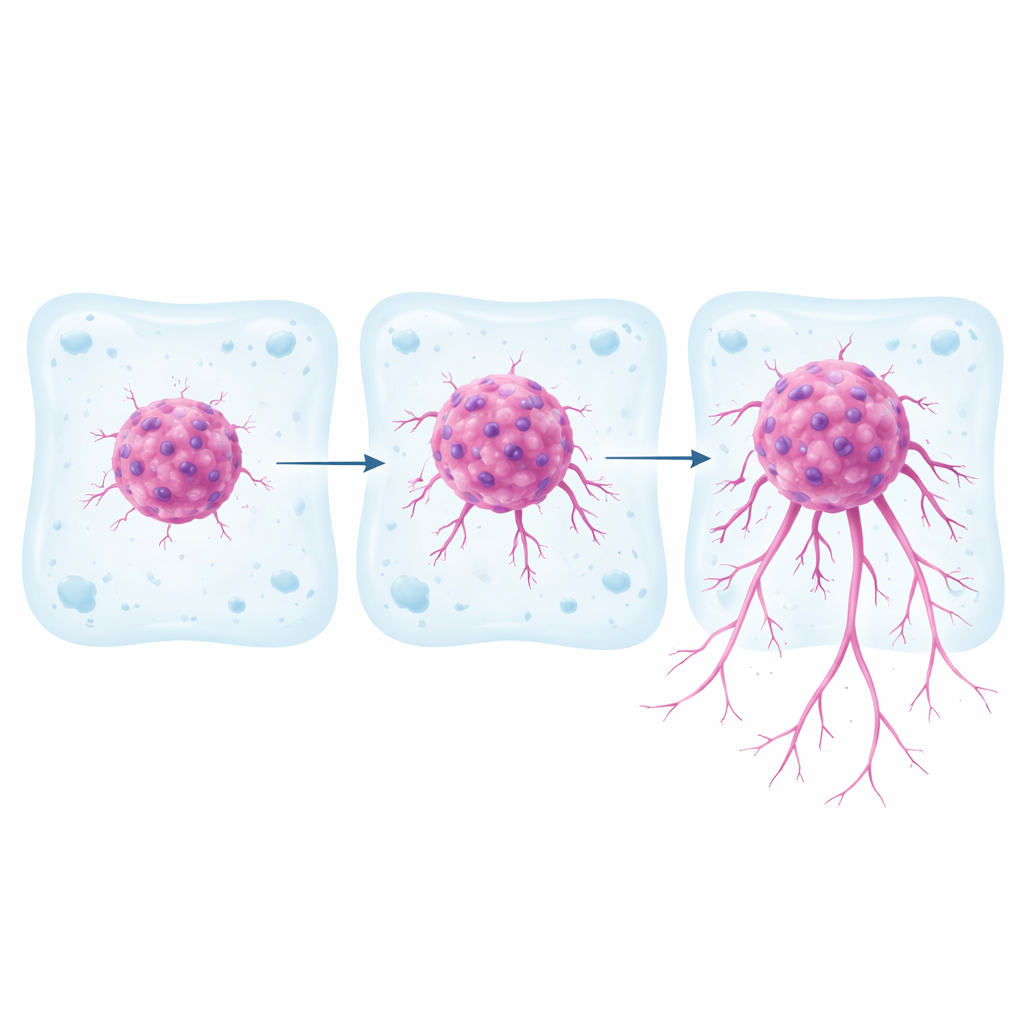
Extending the approach to patient tumors
To move closer to clinical reality, the researchers also created 3D "tumoroids" from fresh tumor samples taken from two kidney cancer patients whose cancers had different standard pathology grades. Grown in the same gel system, tumoroids from the higher-grade tumor invaded quickly and sent out many exploratory branches, while those from the lower-grade tumor remained more compact and slow-moving. Most of the invading cells carried a known kidney cancer marker, confirming they came from the tumor and not from surrounding normal cells. Although this patient sample is small, the results suggest that the 3D invasion behavior of a patient’s tumoroids could reflect the seriousness of their disease.
What this means for patients
This work shows that a relatively simple 3D lab test can mirror how kidney cancer cells spread in living animals. By watching how far and how dynamically cells escape from mini-tumors in a realistic gel, scientists can distinguish between cancers with low, medium, and high metastatic potential. In the future, such 3D tumoroid assays, combined with standard microscopic grading and genetic tests, could help doctors better predict which kidney cancers are likely to metastasize and tailor treatment accordingly. They could also provide an ethical, faster platform to test new drugs aimed at stopping cancer spread before it begins.
Citation: Cesana, B., Nemoz-Billet, L., Azemard, V. et al. Using 3D Invasion properties of RCC Cell Lines In Vitro to predict their Metastatic Potential In Vivo. Cell Death Discov. 12, 122 (2026). https://doi.org/10.1038/s41420-026-02966-7
Keywords: kidney cancer, tumoroids, 3D cell culture, metastasis, renal cell carcinoma