Clear Sky Science · en
Everolimus suppresses glucose transporter 3 membrane trafficking to improve therapeutic efficacy of umbilical cord blood-derived mesenchymal stem cell transplantation in diabetic retinopathy
Why protecting vision in diabetes matters
People with long-standing diabetes often worry about losing their sight. Diabetic retinopathy is a common eye complication where fragile blood vessels and nerve cells in the retina are slowly damaged by high blood sugar. Current drug injections can slow the disease but do not fully restore the eye’s delicate tissue. This study explores a regenerative approach: using stem cells from umbilical cord blood, and shows how a cancer drug called everolimus can make these cells survive better in a sugar-soaked environment like the diabetic eye.
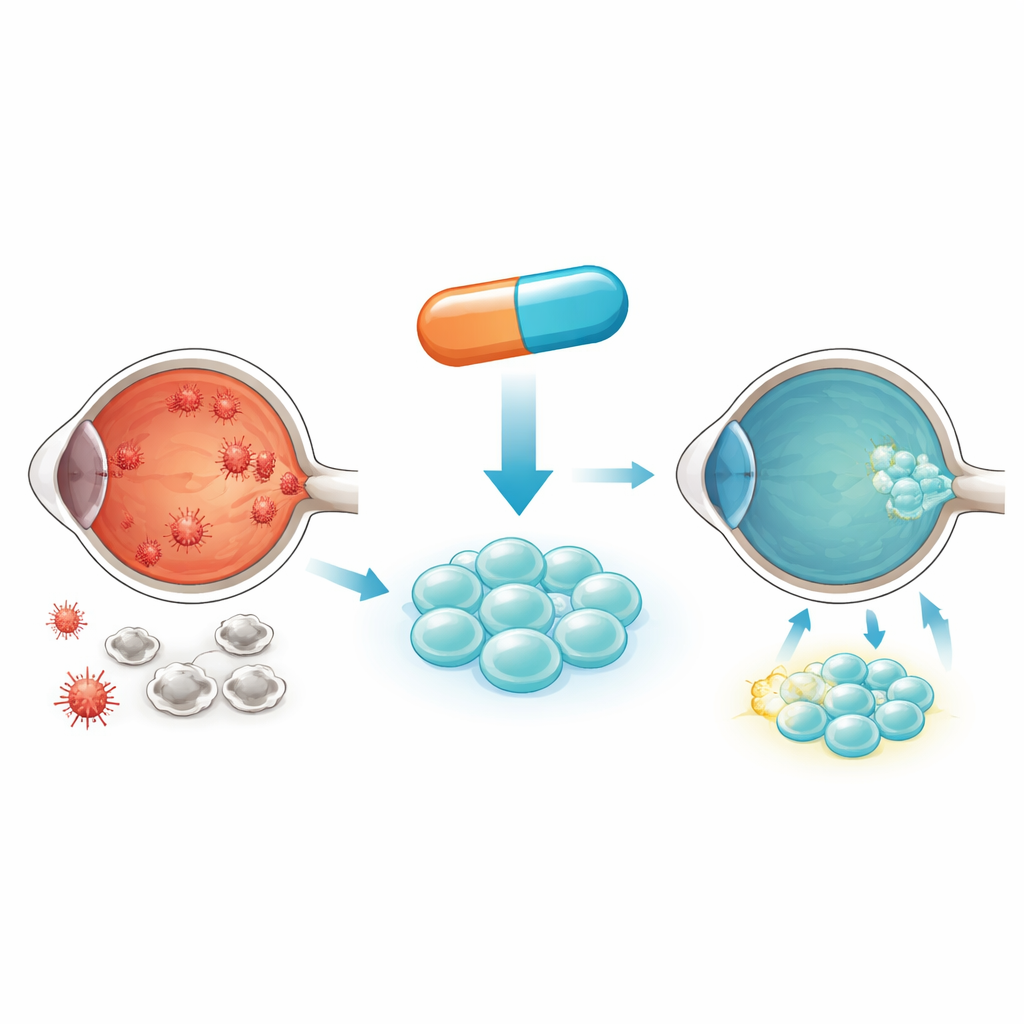
How diabetes harms the eye and incoming stem cells
In diabetes, excess sugar constantly bathes cells. Inside, this sugar is burned in tiny power plants called mitochondria, which can release harmful reactive oxygen species when overloaded. Over time, this oxidative stress pushes cells toward premature aging and death. The authors focus on umbilical cord blood-derived mesenchymal stem cells, which can support and repair damaged retinal tissue. Unfortunately, when these stem cells are placed in high-sugar conditions similar to diabetic blood, they take up too much glucose, generate excessive mitochondrial stress, and start to die. That means fewer healthy cells remain to help rescue the retina.
The sugar doorway that does the most damage
Cells use specialized doorways in their membranes, known as glucose transporters, to let sugar in. The team examined two main types found on these stem cells: GLUT1 and GLUT3. When the researchers exposed the cells to high sugar for many hours, both transporters increased, and glucose uptake rose. But by selectively silencing each doorway with genetic tools, they discovered that GLUT3 played the starring role in driving mitochondrial stress. Blocking GLUT3 sharply reduced the buildup of harmful oxygen species, stabilized mitochondrial function, and lowered overall sugar uptake. When these GLUT3-silenced stem cells were transplanted into rats with diabetic retinopathy, retinal electrical signals and photoreceptor layer thickness improved more than with unmodified stem cells.
A cancer drug repurposed to shield stem cells
The scientists then asked whether a small molecule drug could mimic the benefits of GLUT3 control. They turned to everolimus, which blocks a key growth and metabolism hub in cells called mTOR. In high-sugar conditions, mTOR signaling was strongly activated in the stem cells. Everolimus turned this signal down, reduced glucose entry, and cut mitochondrial oxidative stress more effectively than the older drug rapamycin. At low nanomolar doses, everolimus preserved mitochondrial membrane potential, normalized the cell’s antioxidant defenses, and reduced markers of programmed cell death. In rats with diabetic retinopathy, subconjunctival injections of everolimus-pretreated stem cells led to stronger recovery of retinal electrical responses and better preservation of retinal structure than untreated stem cells.
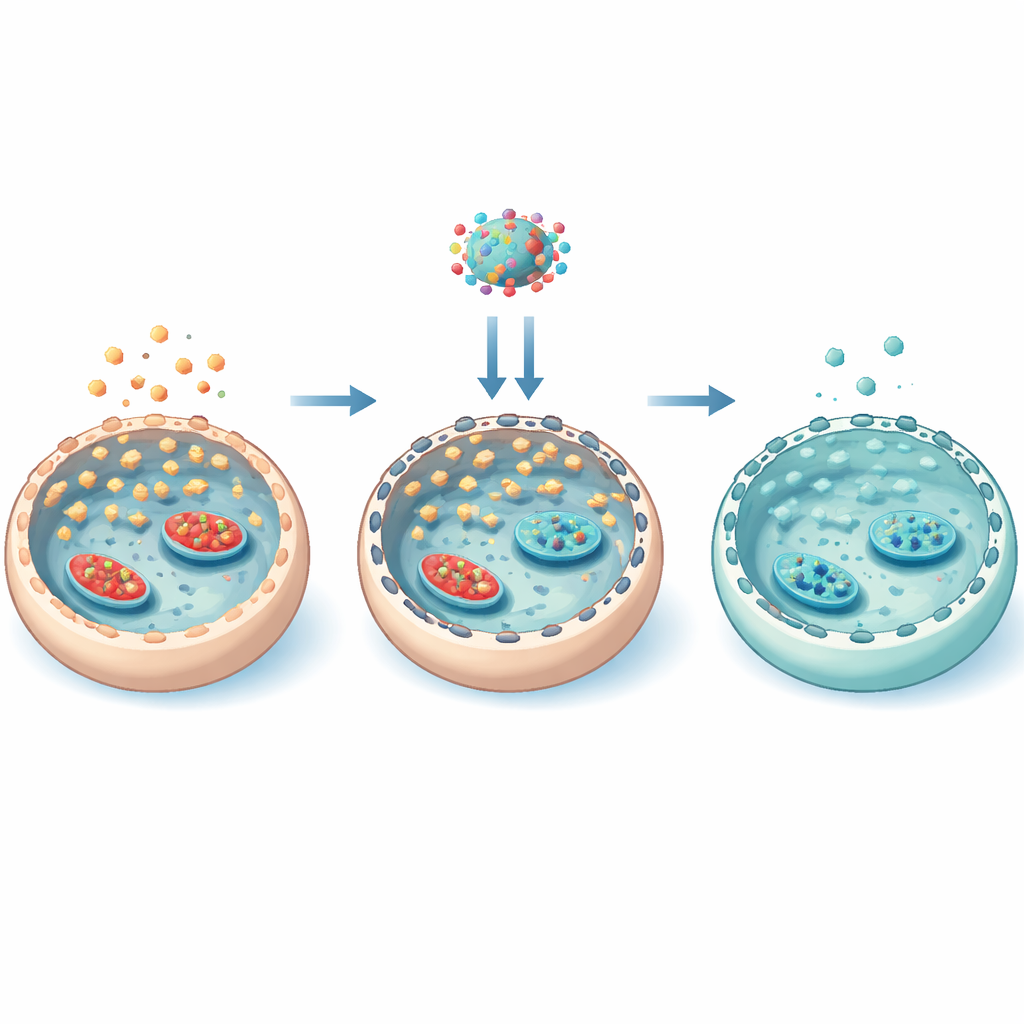
A hidden scaffold that controls sugar entry
To understand how everolimus worked so specifically, the team zoomed in on the cell’s internal scaffold of actin filaments. Under high sugar, mTOR activity increased phosphorylation of a protein called cofilin, which locks actin into more stable filaments. This stabilized scaffold helped move GLUT3 doorways to the cell surface, boosting sugar entry. Everolimus reversed this chain: it reduced mTOR activity, lowered cofilin phosphorylation, loosened the actin network, and pulled GLUT3 away from the membrane. Experiments that either stabilized or disrupted actin confirmed that this physical rearrangement of the scaffold was essential for controlling GLUT3 location. Importantly, everolimus did not broadly rewire all glucose-processing enzymes; instead, it mainly governed how much sugar could get in through transporter positioning.
What these findings mean for future eye treatments
To a lay reader, the bottom line is that the study shows how preconditioning stem cells can help them survive the harsh, sugary environment of the diabetic eye. By using everolimus before transplantation, the researchers were able to close some of the sugar “doors” on the cells, calm their overworked mitochondria, and prevent early cell death. In animal models, these tougher stem cells did a better job of protecting and rebuilding retinal tissue and improving retinal function. While more work is needed before this approach can be tried in patients, the work points to a practical strategy: tune the cells’ sugar intake and stress responses before they are delivered, so they are ready to repair the diabetic retina rather than being overwhelmed by it.
Citation: Jo, H.Y., Jung, J.S., Jo, H.H. et al. Everolimus suppresses glucose transporter 3 membrane trafficking to improve therapeutic efficacy of umbilical cord blood-derived mesenchymal stem cell transplantation in diabetic retinopathy. Cell Death Dis 17, 426 (2026). https://doi.org/10.1038/s41419-026-08673-6
Keywords: diabetic retinopathy, mesenchymal stem cells, everolimus, oxidative stress, glucose transporters