Clear Sky Science · en
Malic enzyme 2 suppresses PINK1-Parkin-mediated mitophagy by stabilizing ATAD3A via competitive interaction with TRIM25
Why tiny powerhouses matter for cancer
Every cell in our body depends on mitochondria, the tiny power stations that make energy. Cells constantly repair and recycle these structures, and when this cleanup process goes wrong, it can contribute to diseases such as cancer and neurodegeneration. This study explores how a metabolic enzyme called malic enzyme 2 (ME2), long known for its role in energy production, unexpectedly acts as a traffic cop for mitochondrial cleanup in liver cancer cells—changing how fast damaged mitochondria are removed and how well tumor cells can grow.
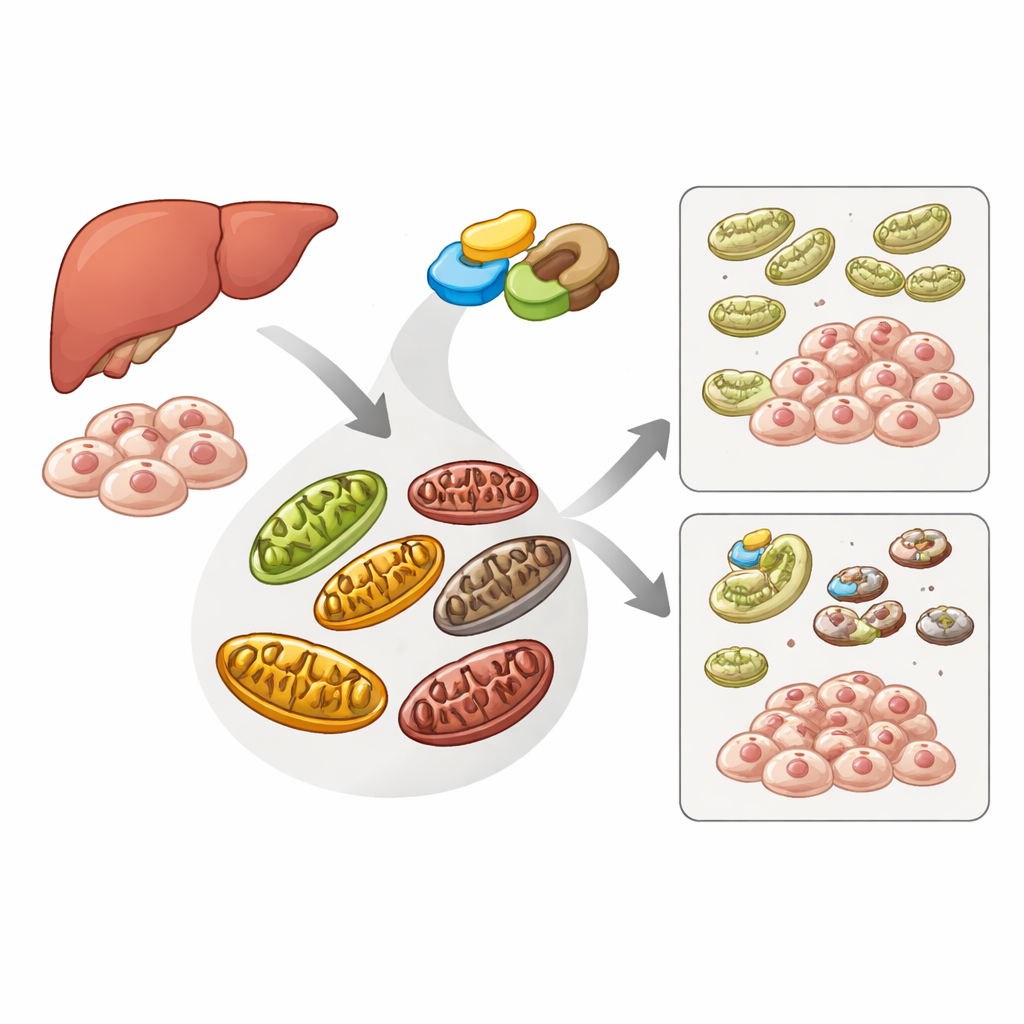
A cell’s cleanup crew for damaged power stations
Cells use a specialized recycling pathway, mitophagy, to identify and remove damaged mitochondria before they harm the cell. A sensor protein called PINK1 and a helper protein called Parkin sit at the heart of this pathway: when mitochondria are unhealthy, PINK1 builds up on their surface, attracts Parkin, and together they tag the faulty organelle for destruction. Another protein, ATAD3A, normally helps ferry PINK1 into mitochondria so it can be broken down, preventing unnecessary cleanup. The authors set out to discover whether ME2, best known for supporting the cell’s main energy-generating cycle, also influences this quality-control system.
When ME2 is lost, mitochondrial cleanup goes into overdrive
Using human cell lines engineered to report mitophagy activity, the researchers reduced ME2 levels and watched what happened. They saw a striking rise in the recruitment of Parkin to mitochondria and a jump in markers of mitophagy, indicating that more mitochondria were being tagged and removed. In liver cancer (HepG2) cells, ME2 loss caused PINK1 to accumulate on mitochondria while key mitochondrial proteins disappeared, consistent with active degradation. Microscopy showed more overlap between mitochondria and autophagosome markers, confirming that damaged power stations were being swallowed by the cell’s recycling machinery far more frequently than normal.
Too much cleanup weakens the cell’s energy system
While some mitophagy is protective, too much can be harmful. In ME2-depleted liver cancer cells, mitochondria became fragmented and sparse, ATP production dropped, and mitochondrial DNA copy number fell. The cells produced more reactive oxygen molecules and had lower membrane potential, both signs of stressed or failing mitochondria. Measurements of oxygen consumption revealed that their respiratory capacity was significantly impaired. Functionally, these cells divided more slowly, formed fewer colonies, and produced smaller three-dimensional spheres. Blocking mitophagy with drugs, or silencing the PINK1 sensor itself, partially rescued cell growth, linking the slowdown in proliferation directly to overactive mitochondrial cleanup.
A protein tug-of-war that decides mitochondria’s fate
To understand how ME2 controls this process, the team searched for proteins that physically associate with it and identified ATAD3A, the mitochondrial gatekeeper that helps regulate PINK1. They showed that ME2 binds ATAD3A and protects it from destruction by TRIM25, a protein that tags other proteins with small "trash me" signals for disposal by the cell’s protein-shredding machinery. When ME2 was absent, TRIM25 attached more tags to ATAD3A, leading to its degradation and less capacity to clear PINK1 from the mitochondrial surface. As a result, PINK1 lingered, mitophagy surged, and mitochondria were excessively removed. Importantly, even mutant versions of ME2 that lacked catalytic activity still restrained mitophagy, indicating that its scaffolding role—holding onto ATAD3A and competing with TRIM25—is what matters here, not its chemistry.
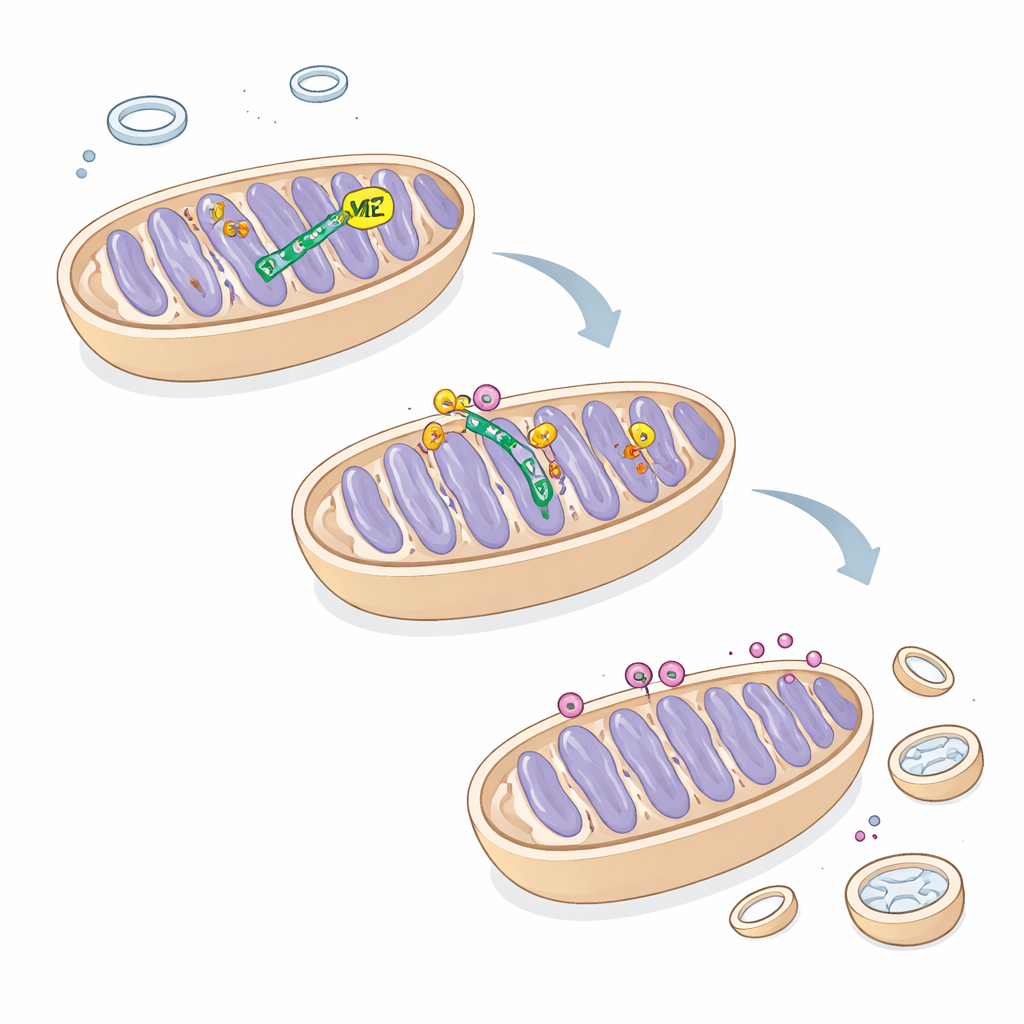
Rebalancing growth and cleanup in liver cancer
By restoring ME2 in cells overloaded with TRIM25, the authors could dampen mitophagy, improve mitochondrial shape and function, and partially restore cell proliferation. These findings reveal a previously hidden circuit in which ME2 shields ATAD3A, allowing proper PINK1 turnover and keeping mitochondrial cleanup in check. For a lay reader, the message is that an enzyme once thought to simply help burn fuel also acts as a structural regulator that balances when mitochondria are repaired versus destroyed. In liver cancer cells, disrupting this balance by losing ME2 pushes the cleanup system into overdrive, stripping the cells of energy-producing machinery and slowing their growth—a vulnerability that could someday be exploited for new therapies.
Citation: Liu, Q., Su, L., Wei, X. et al. Malic enzyme 2 suppresses PINK1-Parkin-mediated mitophagy by stabilizing ATAD3A via competitive interaction with TRIM25. Cell Death Dis 17, 353 (2026). https://doi.org/10.1038/s41419-026-08623-2
Keywords: mitophagy, mitochondria, malic enzyme 2, liver cancer, cell metabolism