Clear Sky Science · en
Mitochondrial DNA release via VDAC1 in keratinocytes: a key driver of innate immunity and vitiligo pathogenesis
Why this skin story matters
White patches of skin caused by vitiligo can be deeply distressing, yet current treatments often only partly work and relapses are common. This study digs into a surprising culprit inside ordinary skin cells: tiny power plants called mitochondria. The authors show how stress inside these structures can spark an alarm signal that rouses the immune system and helps drive the loss of skin color. By tracing this chain of events step by step, they also point to a new kind of therapy that aims not at the immune system itself, but at the way skin cells first call for help.
Skin cells as active alarm senders
Vitiligo has long been viewed as a disease in which immune cells mistakenly attack pigment-producing cells, called melanocytes. However, the outermost skin cells—keratinocytes—vastly outnumber melanocytes and are increasingly recognized as early sentinels in the disease. In people with vitiligo, the skin is under high “oxidative stress,” a chemical strain similar to what happens under strong sunlight or pollution. Keratinocytes under this strain release inflammatory messengers that attract killer T cells to the skin. The key question the authors ask is: what exactly turns this chemical stress into a powerful danger signal that flips on the body’s antiviral and inflammatory machinery?
Power plants that double as warning beacons
Mitochondria carry their own small rings of genetic material, mitochondrial DNA. When this DNA escapes into the main body of the cell, the immune system often treats it as if it were viral or bacterial. The researchers show that exposing keratinocytes to hydrogen peroxide—a classic oxidative stress agent—damages mitochondria without killing the cells outright. Under the microscope, mitochondrial structure becomes distorted, and pieces of mitochondrial DNA appear outside the organelle while the total amount of mitochondrial DNA inside the cell stays roughly constant. This leaked DNA then switches on two powerful sensor systems that normally guard against infections, leading to the production of interferons (proteins that amplify immune responses) and chemokines (signals that call in immune cells). 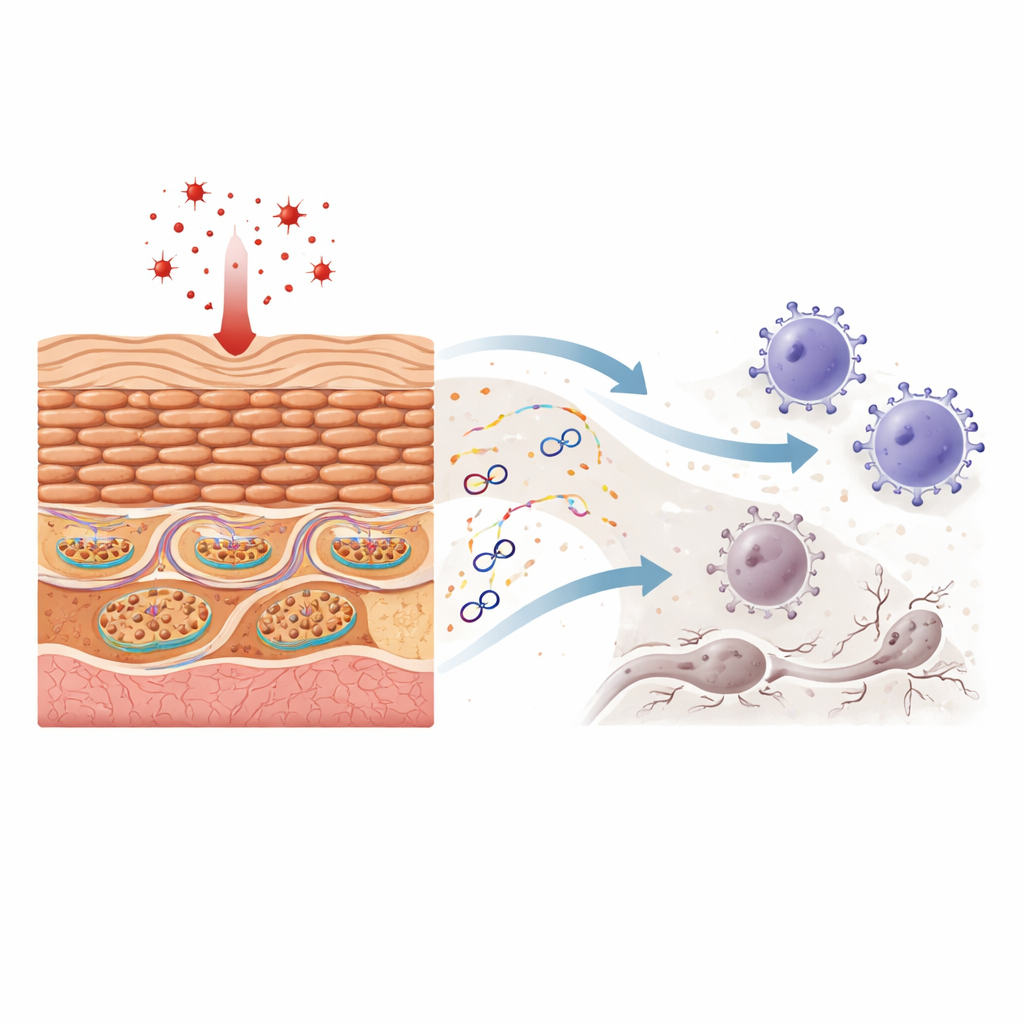
How mitochondrial DNA escapes its cage
To understand how this DNA gets out, the authors focus on two gateways in the mitochondrial membranes. One is the permeability transition pore in the inner membrane, which can open under stress. The other is VDAC1, a channel in the outer membrane that can cluster into larger openings. Using live-cell dyes and molecular probes, they show that oxidative stress first opens the inner pore and then promotes VDAC1 clustering, all without triggering classic cell suicide pathways. Blocking the inner pore prevents DNA from moving from the mitochondrial core into the space between its membranes, while blocking VDAC1 stops it from crossing the outer boundary into the cell interior. Both interventions sharply reduce the amount of mitochondrial DNA in the cytosol, confirming a stepwise escape route that depends on VDAC1.
From internal alarm to fiery cell death
The team then asked whether the stray mitochondrial DNA itself is enough to activate keratinocyte defenses. When they purified this DNA and introduced it directly into keratinocytes, it strongly activated the cGAS–STING system—an internal DNA detector—and another complex called the inflammasome. Together these circuits drove high levels of interferons and chemokines and pushed cells into pyroptosis, an explosive, inflammatory form of cell death that pokes holes in the cell membrane. Blocking cGAS dampened these responses and reduced pyroptosis, showing that this DNA-sensing pathway sits upstream of much of the inflammatory cascade. 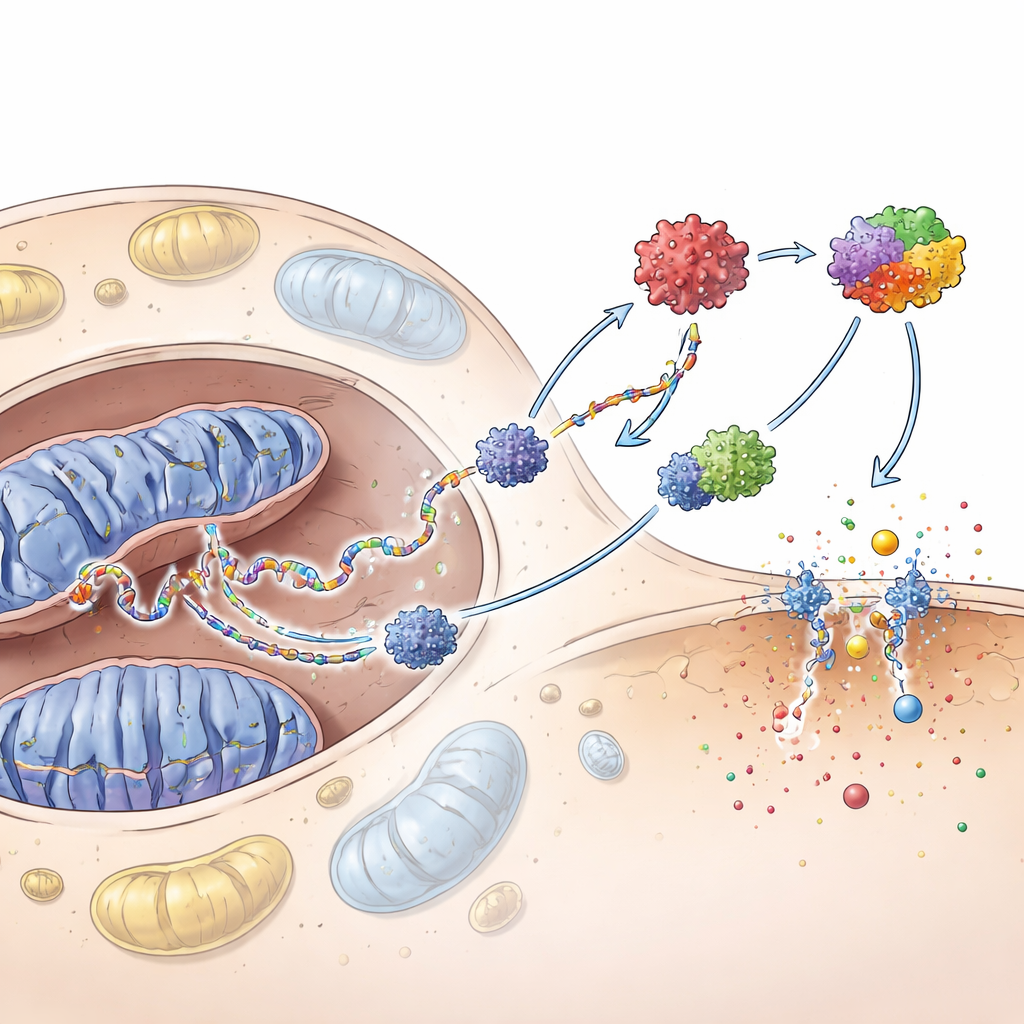
Turning off the gate to calm the skin
Because VDAC1 acts as a key outer gate for DNA escape, the researchers tested whether shutting this gate could ease disease signs. In cell culture, a small molecule called VBIT‑4, which prevents VDAC1 from clustering, cut mitochondrial DNA release and blunted the activation of both cGAS–STING and the inflammasome, lowering chemokine production. Silencing the VDAC1 gene had similar effects. In a mouse model of vitiligo triggered by skin-applied hydrogen peroxide, animals treated with VBIT‑4 developed fewer pale patches, retained more melanocytes, and showed fewer CD8⁺ T cells in the skin. Their skin also displayed lower levels of the inflammatory pathways linked to mitochondrial DNA.
A new way to intercept vitiligo early
To a non-specialist, the takeaway is that ordinary skin cells do not just sit by while the immune system goes rogue—they help start the trouble. Under chemical stress, their mitochondria leak bits of DNA through VDAC1 “gates,” and this leaked DNA acts like a false infection alarm. That alarm wakes up powerful antiviral and inflammatory programs, attracts aggressive immune cells, and promotes destructive forms of cell death, setting the stage for pigment loss. By targeting the VDAC1 gate, drugs like VBIT‑4 might one day quiet this alarm at its source, offering a way to slow or prevent vitiligo before lasting damage to pigment cells occurs.
Citation: Lv, J., Xu, W., Jiang, P. et al. Mitochondrial DNA release via VDAC1 in keratinocytes: a key driver of innate immunity and vitiligo pathogenesis. Cell Death Dis 17, 318 (2026). https://doi.org/10.1038/s41419-026-08585-5
Keywords: vitiligo, mitochondrial DNA, keratinocytes, innate immunity, VDAC1