Clear Sky Science · en
Identification of isoform switching events linked with esophageal adenocarcinoma patient survival informs novel prognostic and therapeutic targets
Why this research matters
Esophageal adenocarcinoma is a type of cancer of the food pipe that is becoming more common yet still has a very low survival rate. Current screening tests often miss people at risk, and standard treatments like chemotherapy and surgery help only a fraction of patients. This study asks a fresh question: instead of looking only at which genes are on or off in tumors, what if we look more closely at which versions of those genes are used? By focusing on these "switched" versions, the researchers uncover new warning signs of poor survival and point to potential treatment strategies that may spare healthy cells.
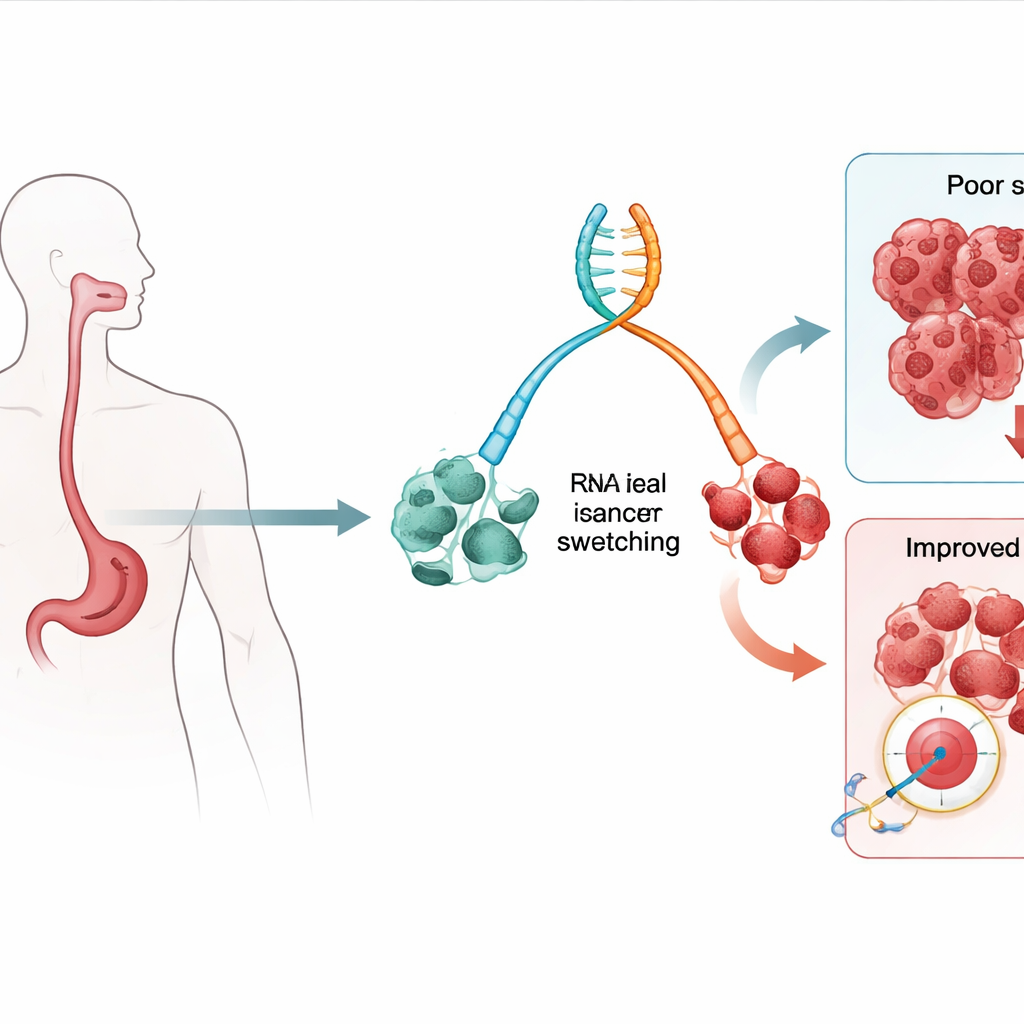
Shifts in gene messages as early warning signs
Our genes can produce multiple slightly different messages, known as isoforms, from the same stretch of DNA. The team examined RNA, the molecule that carries these messages, from tissue samples taken from people with Barrett’s esophagus (a known pre-cancer condition) and from those who had already developed esophageal adenocarcinoma. They looked for isoform “switches,” where one version of a gene becomes more common and another less common as tissue progresses from low-risk Barrett’s to high-risk disease and cancer. Using statistical models that relate these switches to patient outcomes, they identified dozens of isoforms whose presence or absence was linked to higher chances of death from all causes and from cancer specifically.
Two gene versions that spell trouble
Among the many isoforms uncovered, two stood out: particular versions of genes called TTLL12 and HM13. These specific versions were more common in high-grade precancerous lesions and cancers than in low-grade Barrett’s tissue, and patients whose tumors relied heavily on these isoforms tended to have worse survival. Importantly, overall levels of the TTLL12 and HM13 genes did not tell the full story; it was the shift to these particular isoforms that carried the warning signal. The researchers confirmed these patterns not only in patient samples, but also in a rat model of reflux-driven esophageal cancer and in an independent human cancer dataset, suggesting that the findings are robust and biologically meaningful.
Turning harmful isoforms against the cancer
To test whether these isoforms are merely markers or actual drivers of disease, the scientists used small RNA tools to selectively silence the risky versions of TTLL12 and HM13 in two esophageal cancer cell lines grown in the lab. Blocking either isoform sharply reduced cancer cell growth and slowed their ability to migrate—a key step in spread—while having only minor effects on normal esophageal cells. Even more striking, when the isoforms were silenced, standard chemotherapy drugs (paclitaxel and carboplatin) became more effective, working together with the knockdown in a synergistic way to kill many more cancer cells than either approach alone.
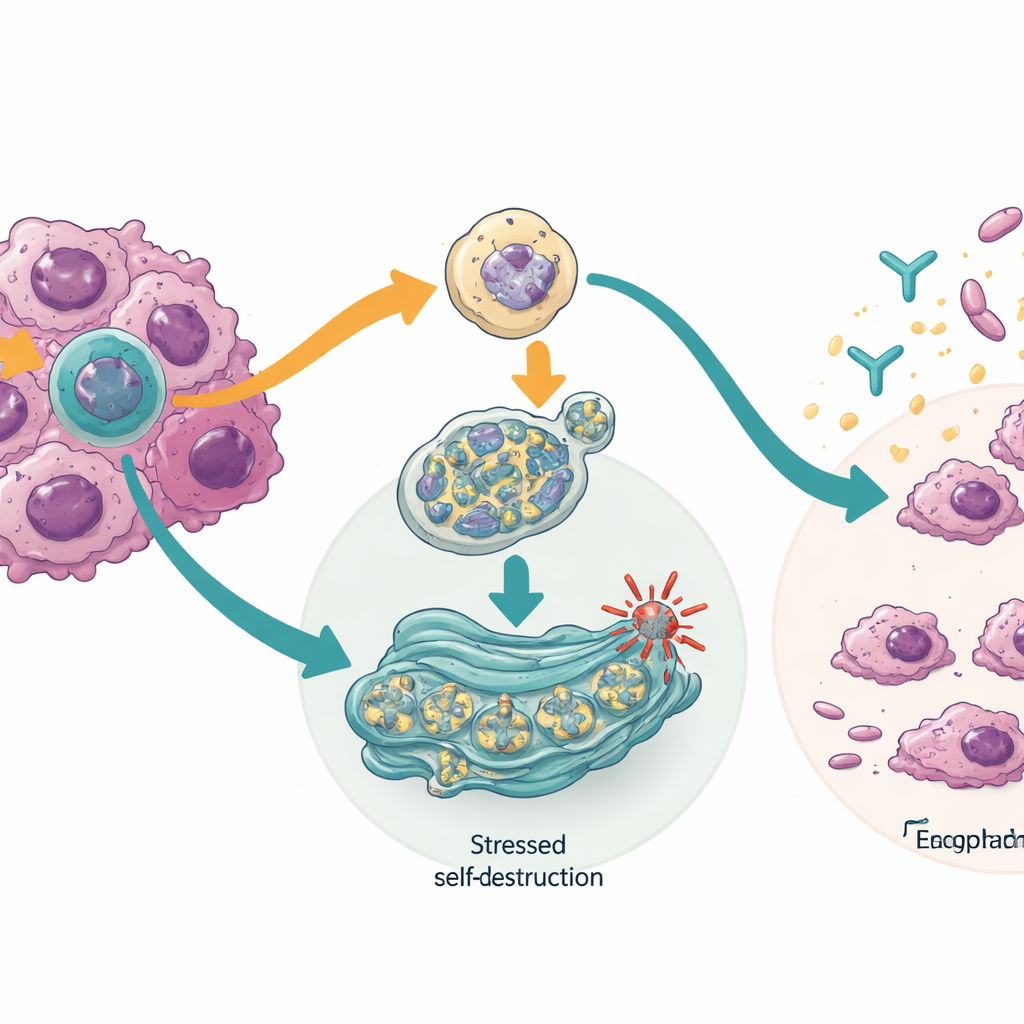
How the cancer cells are pushed toward self-destruction
Digging into how this works, the team found that blocking the TTLL12 isoform switched on a quality-control system inside cells called chaperone-mediated autophagy. This system helps break down damaged or dangerous proteins. In the cancer cells, its activation lowered levels of proteins that help tumors survive DNA damage, including CHK1 and mutant forms of the well-known guardian gene TP53. Blocking the HM13 isoform, on the other hand, stressed the cell’s protein-folding factory, the endoplasmic reticulum, triggering a response pathway that can halt protein production, activate recycling of faulty cell parts, and ultimately drive the cell into programmed death. In some cancer cells, this same change also made them more vulnerable to an immune-based drug that targets PD-L1, suggesting that isoform switching may help tumors hide from the immune system.
What this could mean for patients
Together, these findings suggest that paying attention to which isoforms a tumor uses could improve how doctors predict who is at highest risk and who might respond best to certain treatments. The specific versions of TTLL12 and HM13 highlighted here do more than signal danger; they appear to help cancer cells survive, move, and resist therapy, yet can be targeted without much harm to normal esophageal cells in the lab. While this work is still at an early, experimental stage and larger patient studies are needed, it opens the door to future tests that track isoform patterns and to new drugs or RNA-based therapies designed to selectively shut down the most harmful gene versions in esophageal cancer.
Citation: Zhang, Y., Ntsiful, D.A., Israel, R. et al. Identification of isoform switching events linked with esophageal adenocarcinoma patient survival informs novel prognostic and therapeutic targets. Cell Death Dis 17, 305 (2026). https://doi.org/10.1038/s41419-026-08542-2
Keywords: esophageal cancer, Barrett’s esophagus, RNA isoforms, targeted therapy, autophagy