Clear Sky Science · en
Therapeutic advances with KRASG12C inhibitors and combination strategies in non-small cell lung cancer brain metastases
Why This Matters for Patients and Families
Brain metastases from lung cancer are often devastating, leading to serious neurologic problems and a short life expectancy. This article reviews a new wave of precision medicines that target a specific genetic change, KRAS G12C, in non-small cell lung cancer (NSCLC). These drugs are designed not only to shrink lung tumors but also to cross into the brain and attack metastatic tumors there—an area where older chemotherapies and many targeted drugs have struggled. For patients and families, this work points toward treatment plans that are more effective, longer-lasting, and better tailored to the biology of each person’s cancer.
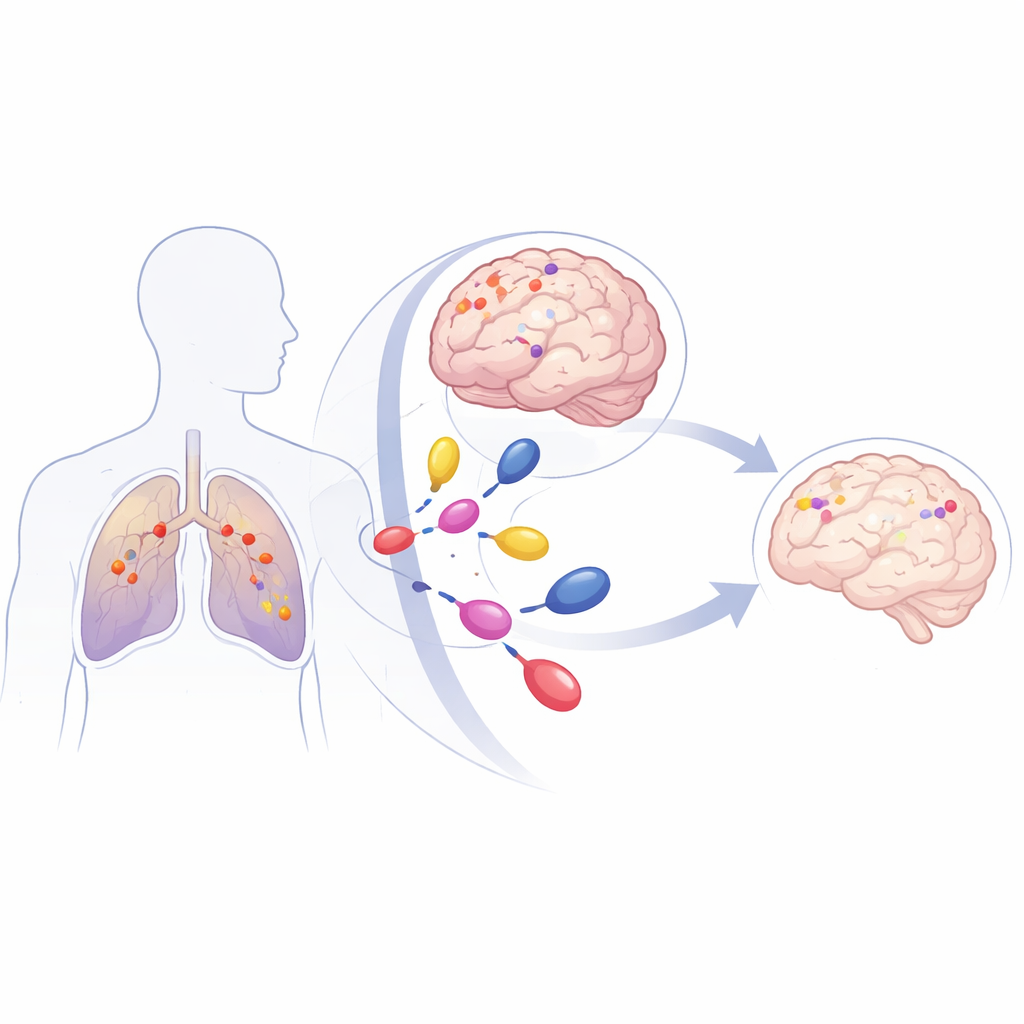
A Common Lung Cancer with a Dangerous Pattern
NSCLC is the most common form of lung cancer and a leading cause of cancer death. Up to 40% of patients will develop metastases in the brain, which are difficult to treat because they sit behind the brain’s protective filter, the blood–brain barrier. Many NSCLC tumors carry mutations in a gene called KRAS, which acts like a stuck accelerator for cell growth. One particular version, called KRAS G12C, makes up about 40% of KRAS-mutant NSCLC. Patients with this mutation have a high risk of developing brain metastases early in the course of their illness, and traditional treatments such as whole-brain radiation or focused radiosurgery usually control disease for only a few months.
How KRAS G12C Fuels Tumor Growth
KRAS normally switches between “on” and “off” states to regulate cell signals for growth, survival, and metabolism. The G12C mutation locks KRAS in its active form, continuously sending growth signals through major pathways that promote cell division, resistance to cell death, new blood vessel formation, and an immune-suppressive environment around the tumor. This constant signaling not only accelerates tumor growth in the lung but also helps cancer cells spread and adapt to new sites like the brain. Co-mutations in other genes, such as CDKN2A, KEAP1, LKB1, and SMARCA4, can make tumors even more aggressive and more likely to recur in the brain after local treatments.
New Drugs That Reach the Brain
For decades, KRAS was considered “undruggable,” but several classes of small molecules now specifically target KRAS G12C. Some bind the inactive form of the protein and keep it switched off, while newer agents also target the active form. Drugs such as sotorasib and adagrasib were the first to reach the clinic and have shown clear benefit in advanced NSCLC, including in patients with brain metastases. Adagrasib in particular has strong evidence that it crosses the blood–brain barrier and produces shrinkage of untreated brain lesions in a substantial fraction of patients, with many achieving months of controlled intracranial disease. Next-generation agents—such as olomorasib, fulzerasib, garsorasib, divarasib, D3S-001, and RMC‑6236—are being tested for greater potency, better brain penetration, and broader activity against various KRAS mutations.
Combining Forces to Overcome Resistance
Even with these advances, most patients treated with KRAS G12C inhibitors alone see their cancer progress again within several months. The review highlights how tumors adapt—through secondary KRAS mutations, reactivation of growth pathways, and changes in the tumor environment—and describes combination strategies designed to block these escape routes. Pairing KRAS inhibitors with immunotherapy can boost the immune system’s attack on tumors, while combinations with drugs that target MEK, CDK4/6, or SHP2 further shut down downstream signaling. In preclinical models and early clinical studies, these combinations have led to deeper and more durable responses, including in brain metastases. Researchers are also testing whether adding KRAS inhibitors to radiation can sensitize tumors, allowing lower radiation doses while maintaining or improving control in the brain.
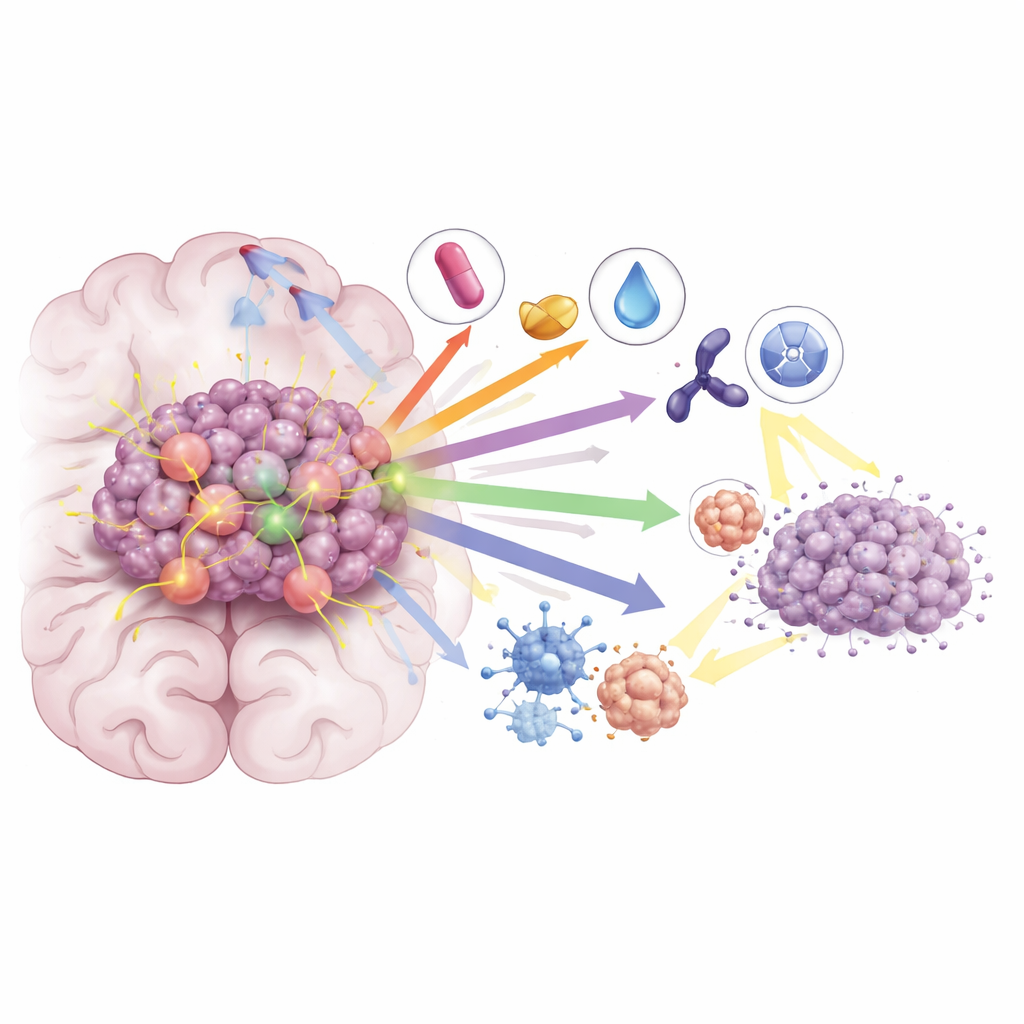
What This Means for the Future
Overall, the article concludes that KRAS G12C is no longer an untouchable target. A growing toolkit of brain-penetrant inhibitors, especially when combined thoughtfully with immunotherapy, other targeted drugs, and radiation, is beginning to change the outlook for NSCLC patients with brain metastases. While challenges remain—particularly drug resistance and the need for longer-lasting benefit—the authors argue that rational combination strategies focused on both the cancer cell and its surrounding environment offer the best hope for turning short-lived responses into more durable control of disease and longer survival.
Citation: Bhattacharya, D., Roman, B. & Reddy, S. Therapeutic advances with KRASG12C inhibitors and combination strategies in non-small cell lung cancer brain metastases. Cancer Gene Ther 33, 323–337 (2026). https://doi.org/10.1038/s41417-026-01003-0
Keywords: KRAS G12C, non-small cell lung cancer, brain metastases, targeted therapy, immunotherapy combinations