Clear Sky Science · en
Attenuation of the CpG island methylator phenotype and lack of WNT signalling activation restrains Kras mutant intestinal neoplasia
Why some bowel growths turn dangerous
Colon cancer often starts from tiny growths in the gut, but not all of these growths behave the same way. This study asks a subtle yet important question: when two closely related cancer‑linked genes misfire, why does one change push cells strongly toward cancer while the other often stalls? By following these gene changes in mice over many months, the researchers uncover why some early serrated polyps in the intestine are far more likely to progress to cancer than others.
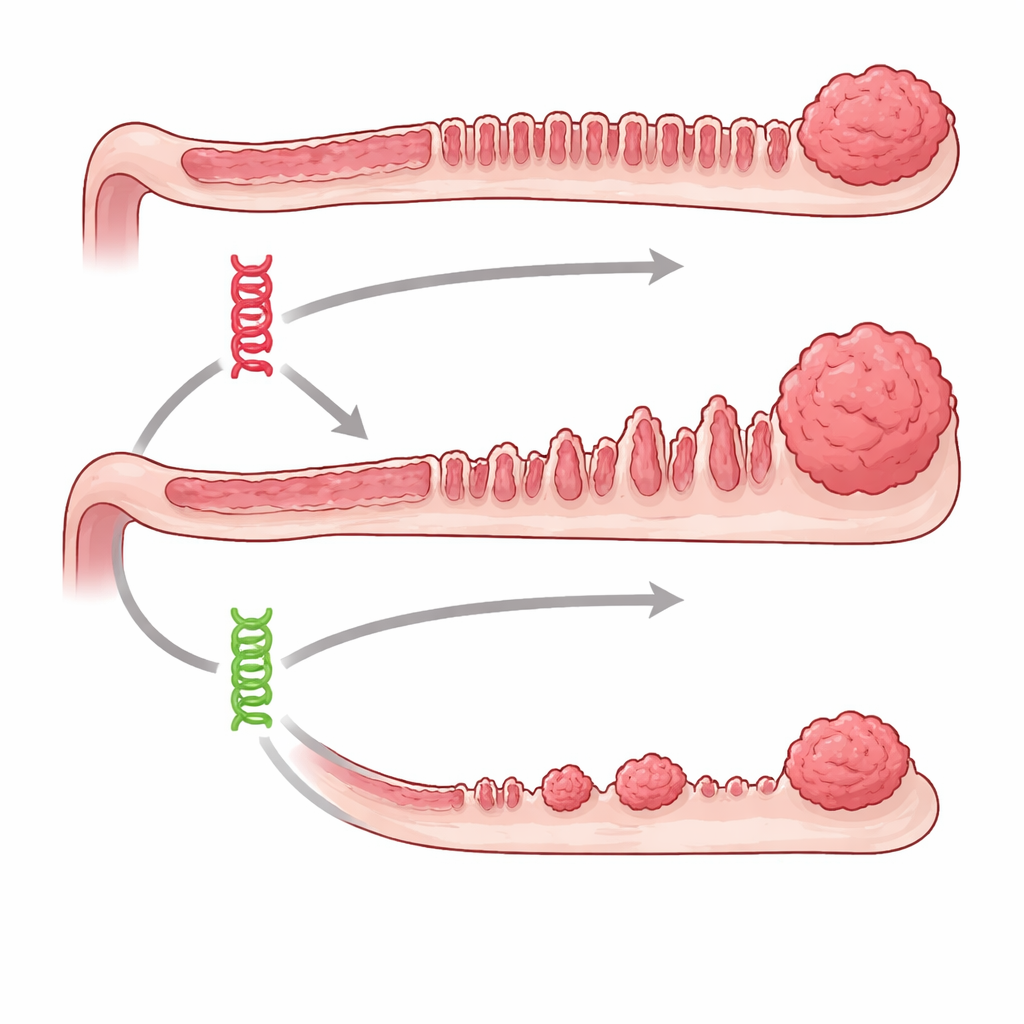
Two faulty switches with very different outcomes
The team focused on two well‑known cancer drivers, BRAF and KRAS, which sit in the same growth‑control pathway inside cells. In people, errors in these genes are common in colon tumours, especially those that arise from serrated, saw‑toothed looking polyps. Using engineered mice, the researchers could flip on either the Braf or Kras mutation specifically in the intestinal lining at a chosen time, then track what happened for up to a year and a half. Both mutations did eventually produce serrated‑type lesions, but Braf‑driven mice developed many more precancerous growths, and they did so much earlier and more consistently than Kras‑driven mice. Advanced serrated tumours and invasive cancers were common in the Braf group but noticeably rare when only Kras was mutated.
Growth patterns and sex differences in risk
Looking at the tissue under the microscope, the researchers saw that Braf‑mutant intestines became longer and more thickened, with enlarged, mucus‑filled crypts typical of serrated disease. Kras‑mutant intestines showed less dramatic structural change, even though individual crypts actually divided more briskly, as measured by a cell‑division marker. This suggests that simple fast growth is not enough to make dangerous lesions. Strikingly, sex also mattered: in the Kras model, advanced serrated lesions arose almost exclusively in male mice, whereas Braf‑mutant mice of both sexes were similarly affected. This mirrors human patterns, where certain BRAF‑mutant colon cancers are more frequent in women and KRAS‑mutant serrated cancers are relatively rare.
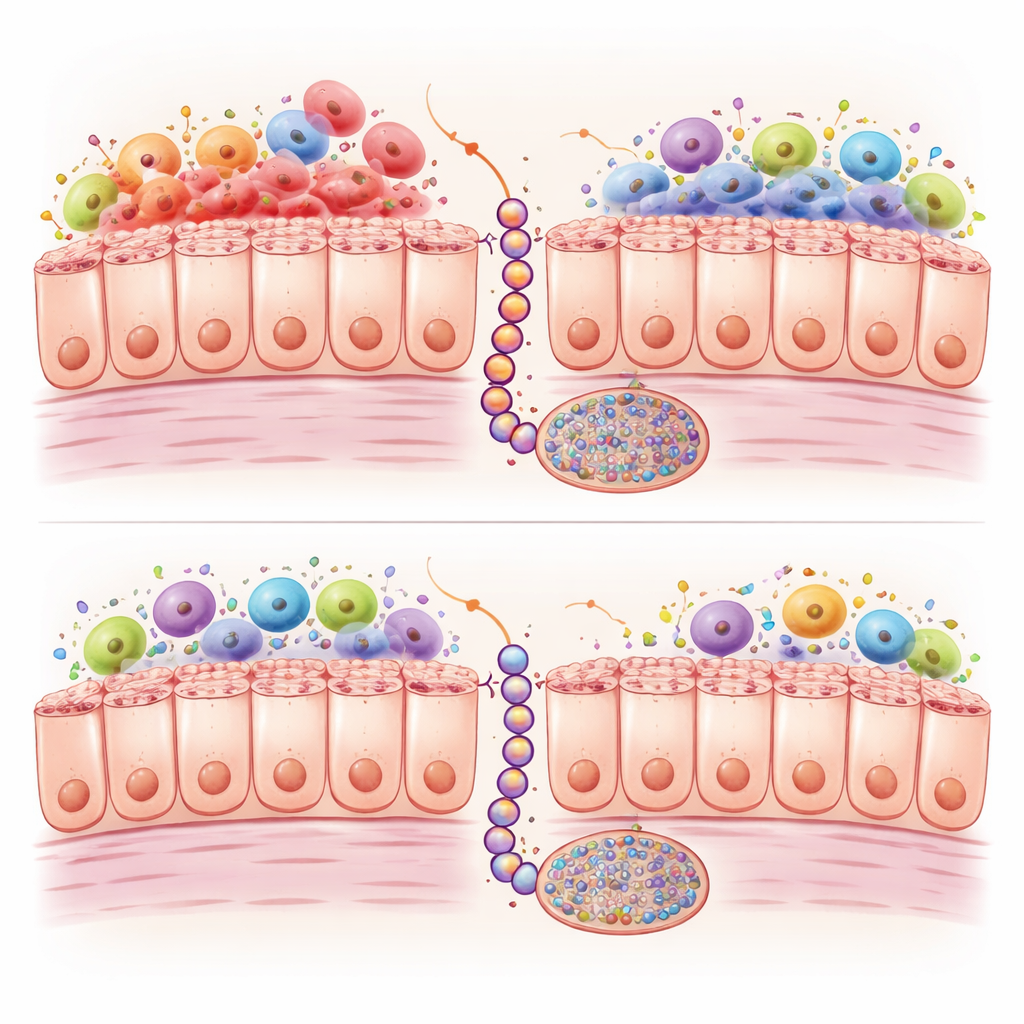
Inflammation and chemical tags on DNA
Digging deeper, the scientists compared gene activity and chemical marks on DNA in the two models over time. Braf‑mutant intestines showed strong activation of immune and inflammatory programs, with an especially high presence of M1‑type macrophages, a variety of white blood cell that releases harsh inflammatory signals. Kras‑mutant intestines had fewer of these cells and weaker immune activation. At the same time, both mutations caused extra chemical tags, called methyl groups, to accumulate on many DNA sites; in human cancers, this so‑called CpG island methylator phenotype, or CIMP, can shut down protective genes. However, in the Kras mice this methylation built up more slowly and to a lesser degree at over a thousand sites, particularly in genes tied to cell growth control, cell‑to‑cell contacts, and WNT signalling, a pathway crucial for pushing serrated lesions toward full‑blown cancer.
A key pathway that stays mostly quiet
One of the clearest differences involved the WNT pathway, which often becomes overactive in colon tumours. In the Braf‑mutant mice, previous work had shown that many lesions eventually acquire changes that drive WNT into overdrive, visible as the protein β‑catenin moving into cell nuclei. In the current study, when the team stained Kras‑mutant serrated lesions for β‑catenin, almost all showed only the normal pattern, with just a single lesion displaying faint abnormal nuclear staining. This means that in the Kras setting, the crucial WNT “second hit” that usually propels serrated polyps to cancer is rarely achieved, likely because the weaker DNA methylation and different gene‑expression environment make such changes less likely to take hold.
What this means for bowel cancer risk
Taken together, the findings show that even though BRAF and KRAS sit in the same signalling pathway, they set the intestinal lining on very different long‑term paths. Braf mutation couples strong inflammatory cues, heavy DNA methylation, and frequent WNT activation, creating fertile ground for serrated lesions to progress to cancer. Kras mutation alone can produce serrated growths, but with slower onset, lower frequency, fewer DNA methylation changes, and little WNT activation, so most lesions remain restrained. For patients, this work helps explain why BRAF‑mutant serrated polyps are considered higher risk, while KRAS‑mutant serrated lesions are both rarer and less prone to turn malignant unless additional damaging events occur.
Citation: Fennell, L., Liu, C., Kane, A. et al. Attenuation of the CpG island methylator phenotype and lack of WNT signalling activation restrains Kras mutant intestinal neoplasia. Br J Cancer 134, 1230–1239 (2026). https://doi.org/10.1038/s41416-025-03271-3
Keywords: colorectal cancer, serrated polyps, BRAF and KRAS, DNA methylation, WNT signalling