Clear Sky Science · en
Defibrotide for prophylaxis of sinusoidal obstruction syndrome/veno-occlusive disease (SOS/VOD) in pediatric high-risk patients: consensus guidelines from the European Society for Blood and Marrow Transplantation (EBMT)
Why Protecting Young Livers Matters
For children with serious blood and immune diseases, a stem cell transplant can be lifesaving—but it also carries dangerous side effects. One of the most feared is a type of liver injury called sinusoidal obstruction syndrome, where tiny blood vessels in the liver become clogged and damaged. This condition can quickly lead to swelling, pain, organ failure, and even death. The article explains why some children are at particularly high risk and presents expert guidelines on using a protective medicine, defibrotide, to prevent this complication before it begins.
What This Dangerous Liver Problem Looks Like
After a stem cell transplant, powerful chemotherapy and radiation can injure the delicate inner lining of the liver’s small blood channels. Inflammation and clotting follow, blood flow slows, and pressure builds up. Children may develop a swollen and painful liver, yellowing of the skin and eyes, rapid weight gain from fluid, and a need for frequent blood and platelet transfusions. In the most severe cases, several organs fail. Children are affected more often than adults—historically up to one in five, and in some high-risk groups far more—because of differences in their developing bodies, their underlying diseases, and the treatments they receive.
Why Some Children Face Much Higher Risk
The risk is not the same for every child. Certain diseases and medical histories are tied to strikingly high rates of liver injury. These include blood disorders that require years of transfusions, such as transfusion-dependent thalassemia; rare bone and immune diseases like malignant infantile osteopetrosis and congenital macrophage activation syndromes; aggressive cancers such as high-risk neuroblastoma; and any child with prior liver damage or a past episode of this same liver complication. Very young children under two years old are especially vulnerable, in part because they often receive intense drug combinations and because key medicines, such as the chemotherapy busulfan, behave very unpredictably in their bodies.
How Doctors Try to Spot Trouble Early
For many years, doctors relied on simple rules to diagnose this liver problem, such as waiting for bilirubin—a yellow pigment measured in blood—to rise above a fixed level. The new guidelines show that this approach can miss nearly a third of cases in children, who may never become obviously jaundiced. Instead, pediatric experts have refined their criteria to look for earlier and more subtle warning signs, especially a pattern called refractory thrombocytopenia, where platelets are consumed so quickly that transfusions are needed more and more often. Using these pediatric-specific rules, doctors can recognize the condition two to three days sooner, allowing treatment to start before damage becomes irreversible.
Using a Protective Drug Before Damage Occurs
Defibrotide is a drug that acts mainly on the cells lining blood vessels, calming inflammation and helping to balance clotting and breakdown of clots. It is already an approved treatment once severe liver injury is diagnosed. Because it works at the level of the blood vessel lining, many experts believe it makes the most sense to use it preventively in children whose risk is very high, rather than waiting until swelling, jaundice, and organ failure appear. Past studies—including a large pediatric trial and a meta-analysis of several reports—suggest that giving defibrotide before transplant can cut the chance of liver injury and reduce related problems such as kidney damage, even if it does not always change the final death rate in every study. Conflicting trial results in mixed adult–child populations, however, led European regulators to caution against routine preventive use, creating confusion for pediatric teams.
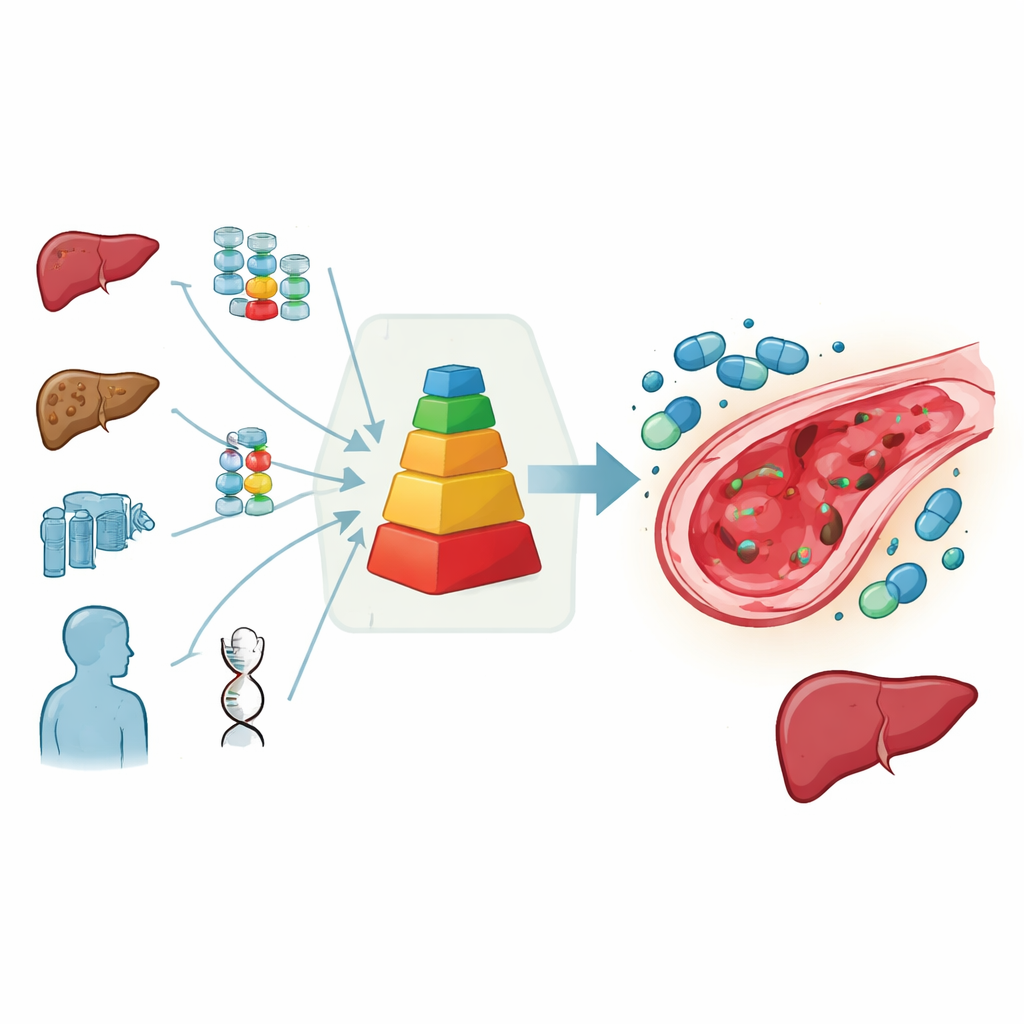
A Simple Score to Guide Tough Decisions
To help resolve this impasse, the European pediatric transplant group assembled experts to weigh all available data and their clinical experience. They created a practical scoring system that sums up each child’s risk: points are assigned for high-risk diseases, very young age, previous liver injury, signs of iron-related liver scarring, and especially hazardous treatments such as busulfan-based intensive chemotherapy, total body irradiation, repeat transplants, or certain antibody drugs used before transplant. The higher the total score, the greater the expected chance of developing liver injury. In this framework, defibrotide is strongly recommended when the predicted risk is clearly above about 15 percent, considered advisable at intermediate risk, and not recommended when risk appears low.
What This Means for Families and Care Teams
For families facing a stem cell transplant, these guidelines offer a clearer, evidence-based way to weigh the benefits and burdens of adding a preventive drug. The message is that preventing or softening this liver complication can spare children prolonged hospital stays, intensive care, and severe illness, even when death is ultimately avoided. The authors argue that, given defibrotide’s safety profile, targeted preventive use in the highest-risk children is a sensible and humane strategy. They present their recommendations as a living document that will evolve as new data emerge, but their core conclusion is straightforward: by carefully identifying children at greatest risk and protecting the liver’s fragile blood vessels in advance, doctors can make a dangerous yet lifesaving procedure measurably safer.
Citation: Corbacioglu, S., Bajwa, R., Antmen, A.B. et al. Defibrotide for prophylaxis of sinusoidal obstruction syndrome/veno-occlusive disease (SOS/VOD) in pediatric high-risk patients: consensus guidelines from the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 61, 417–425 (2026). https://doi.org/10.1038/s41409-025-02793-x
Keywords: pediatric stem cell transplantation, sinusoidal obstruction syndrome, defibrotide prophylaxis, liver complications, treatment risk scoring