Clear Sky Science · en
Preventing trogocytosis by cathepsin B inhibition augments CAR T-cell function
Why this research matters for future cancer therapies
CAR T-cell therapy has transformed treatment for certain blood cancers, sending many patients into deep remission. Yet a large fraction of those patients eventually relapse, often because cancer cells find ways to hide from these engineered immune cells. This study uncovers a previously murky escape route—"trogocytosis," a process where cancer cells give up key surface markers to CAR T cells—and shows how blocking a single enzyme can make CAR T cells more durable and effective.
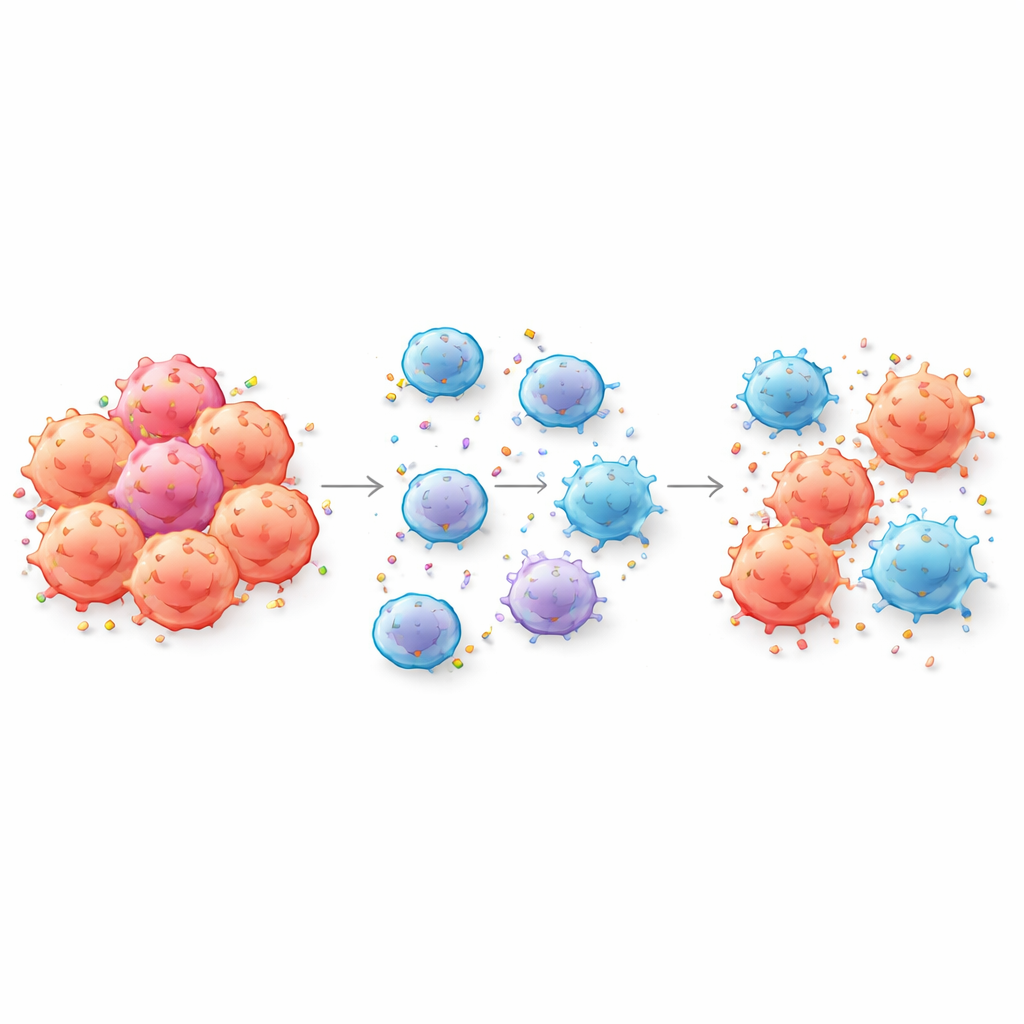
When cancer cells hand off their ID tags
CAR T cells are designed to recognize cancer by grabbing onto specific markers, or antigens, on the tumor cell surface. The authors show that, during close contact, CAR T cells can literally tear off pieces of the tumor’s membrane and incorporate those antigens into their own surface—a process called trogocytosis. As this happens, tumor cells lose the very markers CAR T cells need to detect them, while CAR T cells themselves become coated in tumor antigens. The team confirmed this exchange for several clinically important targets, including CD19 and BCMA, in both blood cancers and solid tumor models, and even detected it in patients’ blood after CAR T treatment.
Friendly fire: how trogocytosis weakens CAR T cells
Once CAR T cells carry tumor antigens on their own surface, they start to recognize each other as targets. This leads to “fratricide,” where CAR T cells kill fellow CAR T cells, and to chronic self-stimulation that drives them into an exhausted, less functional state. To prove trogocytosis was the direct cause, the researchers engineered a “trogocytic antigen degrader” system that selectively destroys the transferred antigen on CAR T cells without affecting their normal targets. Removing these acquired antigens sharply reduced fratricide and the build-up of exhaustion markers, and allowed CAR T cells to control leukemia cells more effectively over repeated challenges in the lab.
Tracking and dissecting the antigen-stealing process
To study trogocytosis in real time, the team built a split-luciferase assay in which light is produced only when antigen fragments move from tumor cells into CAR T cells. This sensitive readout enabled rapid screening of cellular processes that might drive antigen transfer. Using small-molecule inhibitors, they discovered that the enzyme cathepsin B, a protease normally involved in breaking down proteins, is a key player. Cathepsin B rapidly moves to the contact zone where CAR T cells engage tumor cells, and its activity outside the cell is sufficient to promote the peeling and uptake of antigen-rich membrane fragments. Remarkably, blocking cathepsin B strongly reduced trogocytosis without significantly impairing the CAR T cells’ ability to kill tumor cells.
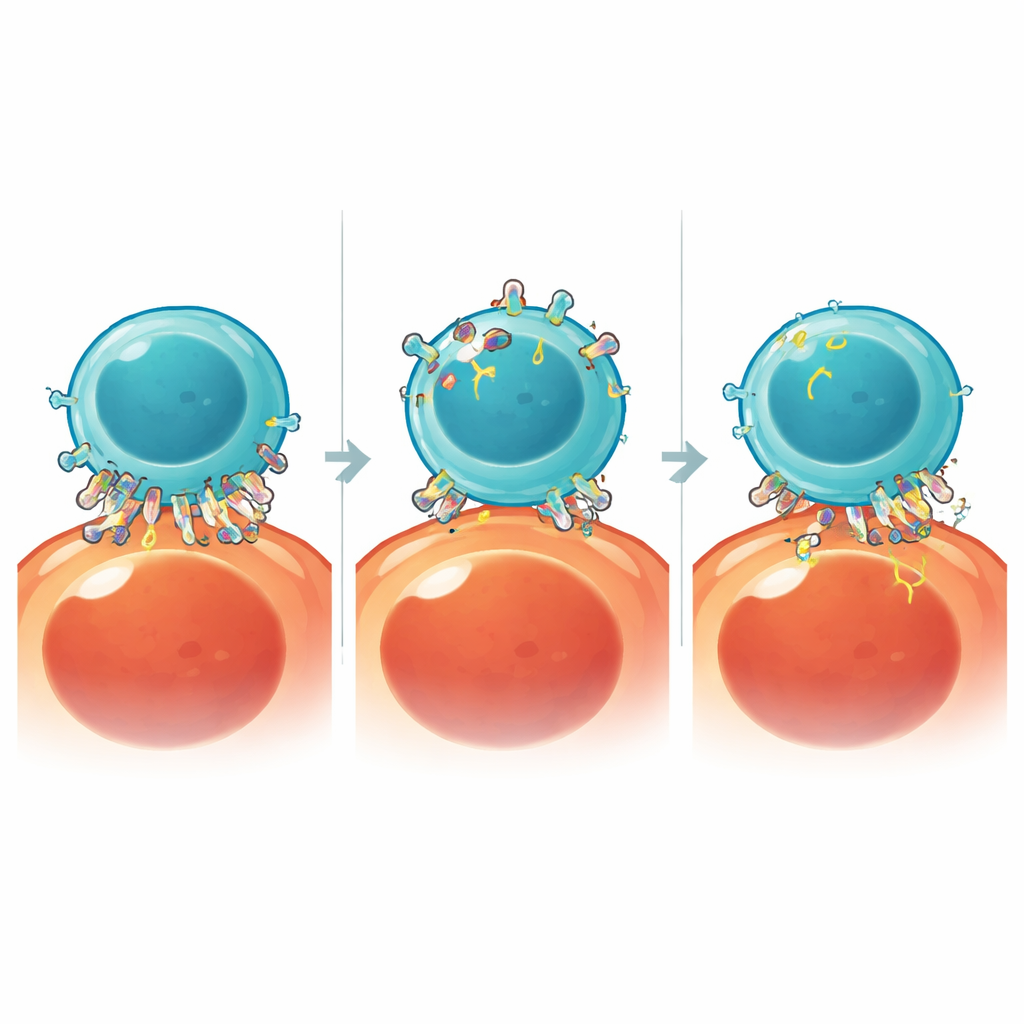
Using natural brakes to protect CAR T cells
The body already contains built-in inhibitors of cathepsin B, known as cystatins. The researchers boosted one of these, cystatin A, inside CAR T cells and showed that this safely dialed down cathepsin B activity and sharply reduced trogocytosis across multiple tumor models, including primary leukemia cells from a patient. With less antigen theft, CAR T cells survived better, accumulated in greater numbers, and maintained stronger long-term tumor control in both lab dishes and mouse models. However, chronic cystatin A expression unexpectedly triggered a separate internal brake—upregulation of the regulatory protein CISH—which promoted signs of T-cell exhaustion. By knocking out CISH while overexpressing cystatin A, the team could retain the benefits of reduced trogocytosis and fratricide while minimizing exhaustion, further extending CAR T persistence.
What this could mean for patients
This work provides clear evidence that antigen stealing by CAR T cells is not just a curious side effect but a direct driver of treatment failure, causing both tumor escape and self-destruction of therapeutic cells. It also shows that trogocytosis depends on cathepsin B and can be selectively uncoupled from cancer-killing activity using either drugs or genetic tricks such as cystatin overexpression. In the future, CAR T products that are pre-engineered to limit trogocytosis—or combined with smart cathepsin B inhibitors—could remain active for longer, keep better pressure on cancer cells, and reduce the chances of relapse.
Citation: Dietze, K.A., Nguyen, K., Pathni, A. et al. Preventing trogocytosis by cathepsin B inhibition augments CAR T-cell function. Sig Transduct Target Ther 11, 149 (2026). https://doi.org/10.1038/s41392-026-02654-z
Keywords: CAR T-cell therapy, trogocytosis, cathepsin B, cystatin A, cancer immunotherapy