Clear Sky Science · en
Endoplasmic reticulum stress in disease pathogenesis: its implications for therapy
When Cells’ Factories Go Wrong
Every cell in your body contains a tiny factory called the endoplasmic reticulum, or ER, where most proteins are folded into their working shapes. This review explains what happens when that factory is overloaded or damaged—a state known as ER stress—and how it quietly contributes to many major illnesses, from cancer and heart disease to Alzheimer’s, diabetes, and autoimmune disorders. Understanding this hidden stress response not only helps explain why such different diseases share common features like inflammation and cell loss, but also points to new kinds of treatments that fine‑tune the cell’s own repair systems.
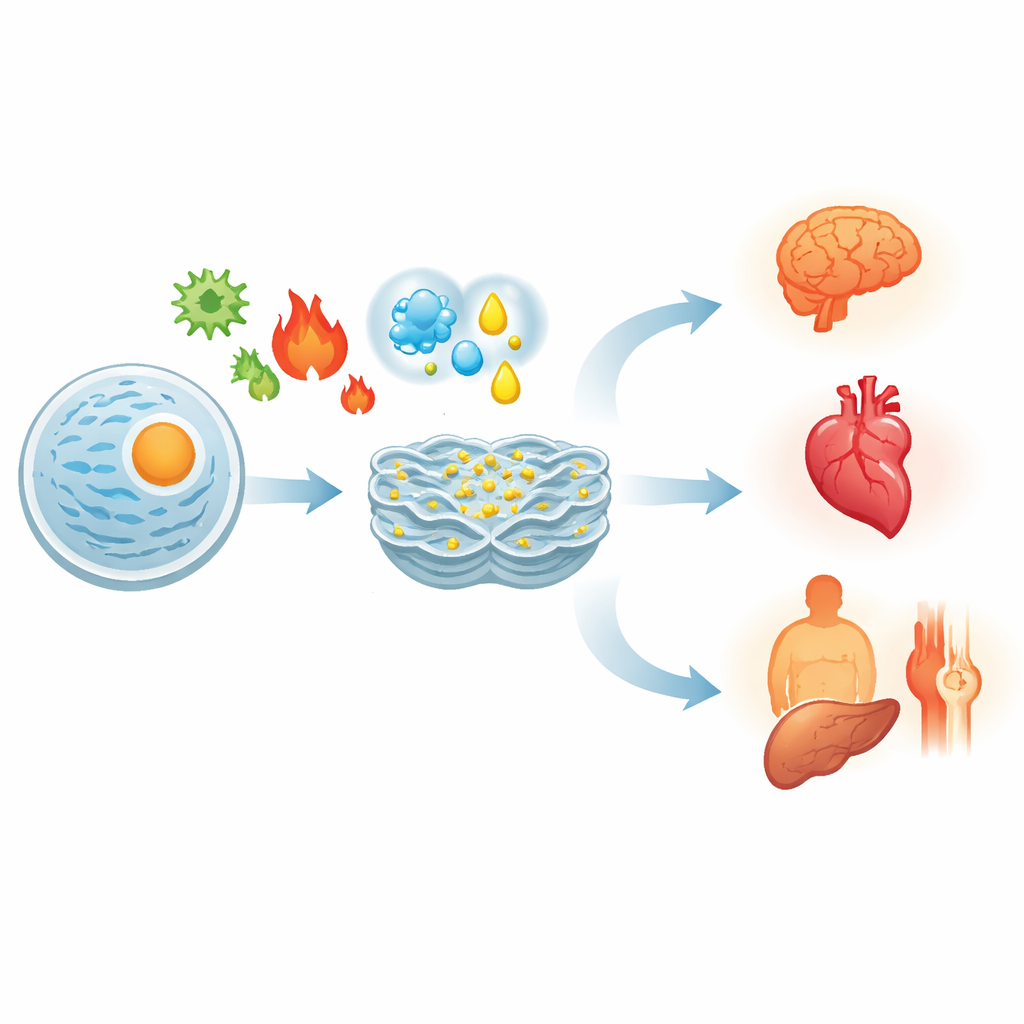
How Cells Sense Trouble Inside
Proteins are made as floppy chains that must be folded with care. Heat, lack of oxygen, excess nutrients, toxins, or genetic mutations can all cause misfolded proteins to pile up in the ER. To cope, cells trigger a coordinated defense program called the unfolded protein response (UPR). Three sensor proteins embedded in the ER membrane—IRE1, ATF6, and PERK—act like alarms that flip on when they sense misfolded proteins. Together they slow the influx of new proteins, boost the production of folding helpers and quality‑control factors, and increase the disposal of defective proteins. If this response succeeds, balance is restored and the cell survives.
When Adaptation Turns into Damage
The same safety system can become harmful when stress is too intense or lasts too long. Under chronic pressure, the UPR shifts from being protective to pushing cells toward self‑destruction. Pro‑death signals are turned on, reactive molecules build up, and communication with other cell parts such as mitochondria breaks down. This prolonged ER stress can kill vulnerable cells outright or leave survivors altered in ways that drive disease—changing how they grow, use energy, or talk to the immune system. The review traces these step‑by‑step changes, showing how the ER becomes a control hub for cell fate, metabolism, and inflammation.
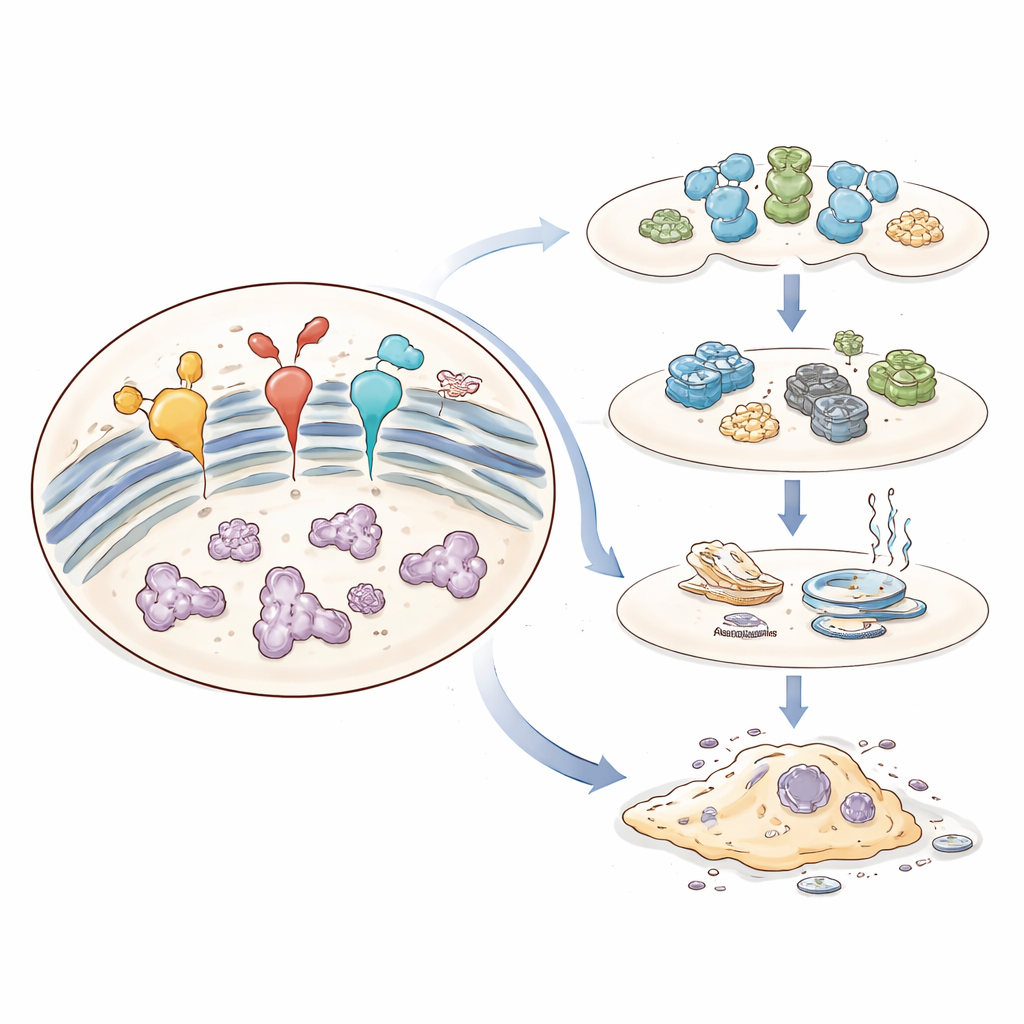
A Common Thread Across Many Diseases
Because all organs depend on properly folded proteins, ER stress shows up in very different conditions. In cancers, tumor cells live on the edge of constant ER stress due to rapid growth and poor blood supply. They exploit the UPR to keep dividing and to evade immune attack, yet can be pushed into death if the stress is tipped further. In the heart and blood vessels, ER stress contributes to clogged arteries, damage after a heart attack, and heart failure by harming muscle and vessel cells. In the brain, it is linked to the toxic protein clumps seen in Alzheimer’s, Parkinson’s, and Huntington’s disease, and to the gradual loss of neurons. In obesity and type 2 diabetes, ER stress in the liver, fat, and pancreas disrupts insulin action and hormone secretion, fueling high blood sugar. In autoimmune diseases such as rheumatoid arthritis or inflammatory bowel disease, ER stress in immune and barrier cells amplifies inflammation and tissue injury.
Turning a Weakness into a Target
Because ER stress sits at the crossroads of so many pathways, it offers multiple entry points for therapy. Drug developers have designed molecules that either dial down overactive stress signals or deliberately exaggerate them in cancer cells to trigger their death. Some compounds aim at the three main sensors; others boost folding helpers like GRP78, enhance the clearance of faulty proteins, or protect mitochondria. The review also catalogues early clinical trials of existing drugs—such as bile acid derivatives, diabetes medicines, and cholesterol‑lowering agents—that appear to ease ER stress in people. At the same time, new tools such as fluorescent probes, nanogels, and experimental cancer vaccines are being built to track or manipulate ER stress more precisely inside tumors and other tissues.
The Road Ahead for ER‑Based Treatments
The authors conclude that ER stress is both an early driver and a sustaining force in many chronic diseases. However, because the UPR can help or harm depending on dose and timing, future treatments must be carefully tuned rather than simply switched on or off. Better animal models that combine multiple diseases, together with "omics" technologies that measure stress signals across whole organs, should clarify when intervention will help. If these challenges can be met, therapies that re‑balance the cell’s internal factory may complement existing drugs, offering new ways to slow cancer, protect the heart and brain, improve metabolism, and calm misdirected immune responses.
Citation: Wei, S., Zhang, N., Zhang, H. et al. Endoplasmic reticulum stress in disease pathogenesis: its implications for therapy. Sig Transduct Target Ther 11, 136 (2026). https://doi.org/10.1038/s41392-026-02600-z
Keywords: endoplasmic reticulum stress, unfolded protein response, cancer therapy, neurodegenerative disease, metabolic disorders