Clear Sky Science · en
Advanced in vitro cardiac models for drug evaluation: integration of organoids, engineered tissues, and microphysiological systems
Why Growing Mini Hearts Matters
Heart disease is the world’s leading killer, yet many promising drugs fail late in development because today’s lab tests and animal studies often miss harmful effects on the human heart. This article explains how scientists are building increasingly realistic "mini hearts" in the lab—from flat cell layers to tiny beating tissues, organoids, and heart-on-a-chip systems—to predict how our hearts will respond to new medicines more safely and accurately.
From Simple Cell Layers to Tiny Beating Tissues
Researchers now routinely grow human heart muscle cells from reprogrammed adult cells, called induced pluripotent stem cells. These cells can be spread as thin sheets or guided into specific shapes using microscopic patterns. Even at this basic level, controlling cell shape, alignment, and the softness of the surface changes how strongly the cells beat and how electrical signals travel between them. These flat cultures are easy to grow in large numbers and to measure with automated cameras and sensors, so they are widely used for the first pass of safety testing, especially to detect drugs that may disturb the heart’s rhythm.
Building Three-Dimensional Heart Muscle and Chambers
To more closely mimic a real heart, scientists assemble three-dimensional heart tissues by mixing heart cells with gel-like materials that resemble the natural scaffold of the heart. These engineered heart tissues are often cast around posts or rings so their tiny contractions can be measured as they pull on the supports. By applying regular stretching and electrical pulses, the tissues gradually develop more adult-like structure and pumping behavior. Larger patches and chamber-shaped constructs can even imitate how a ventricle fills and ejects fluid, allowing measurement of pressure–volume changes similar to those used in the clinic. Adding blood vessel cells and support cells helps these tissues survive longer and integrate better when transplanted into animal hearts.
Self-Assembling Mini Hearts and Early Development
Another branch of research focuses on organoids—self-organizing clusters of cells that resemble early stages of a developing heart. Starting from stem cells, researchers adjust timing and strength of growth signals so the cells spontaneously sort into layers and form hollow, beating structures with early chamber-like regions, outer covering cells, and primitive blood vessels. Some organoids even generate blood-forming and liver-like tissues alongside the heart regions, offering a window into how different organs co-develop. These models are especially powerful for studying birth defects, inherited heart conditions, and how different cell types in the heart communicate during development.
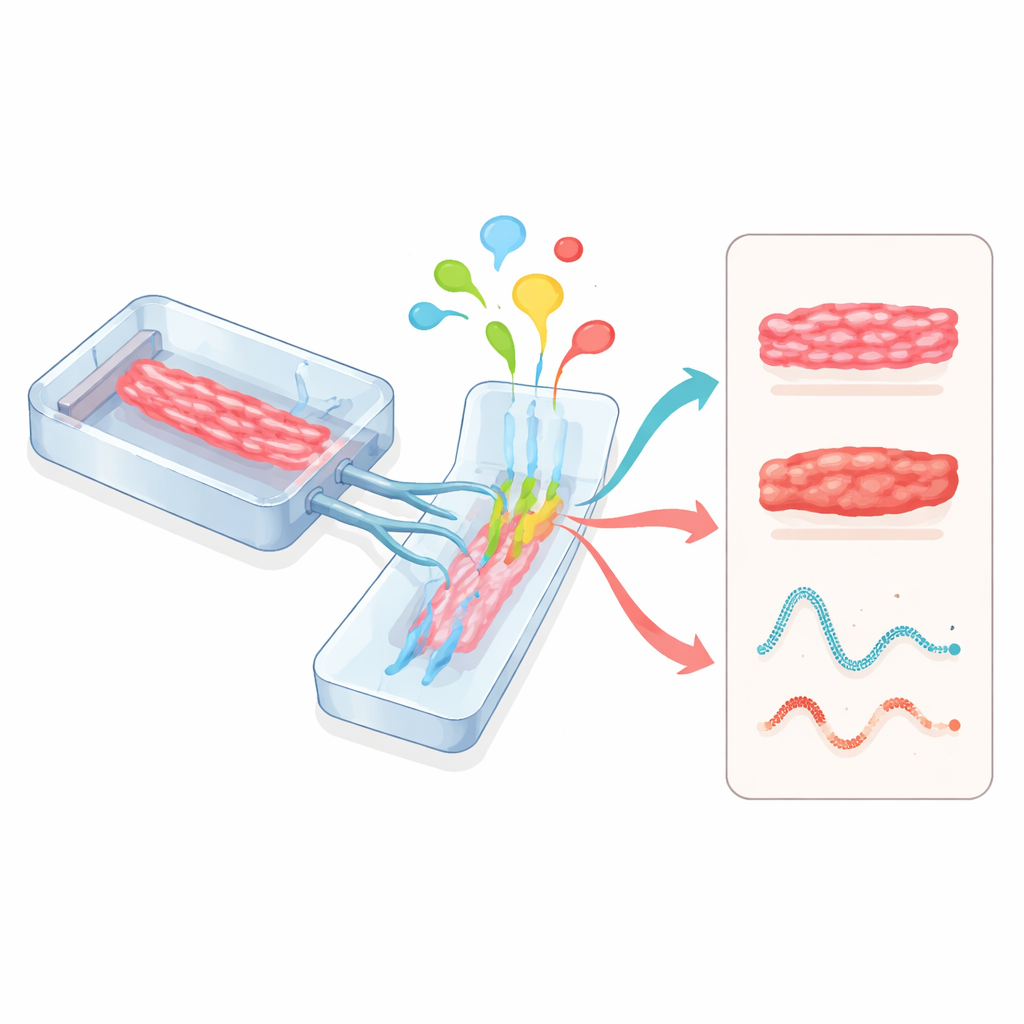
Hearts-on-Chips and Multi-Organ Connections
Microphysiological systems, often called organs-on-chips, take yet another approach. Heart tissues are grown inside small, transparent devices etched with channels for fluid flow and flexible walls that can be stretched. These chips can precisely control nutrients, oxygen, mechanical strain, and electrical pacing while sensors track beating strength and electrical activity in real time. When heart modules are linked with liver, blood vessel, or other organ modules on the same circuit, scientists can see how drugs processed by the liver affect the heart, or how inflammation and immune cells influence heart function. Regulators such as the U.S. Food and Drug Administration are beginning to recognize these human-based chips as promising tools for drug safety evaluation.
What This Means for Future Medicines and Therapies
Together, flat cell layers, engineered tissues, organoids, and organs-on-chips form a toolkit that brings heart testing much closer to human reality than traditional animal models alone. Each model balances realism and practicality in a different way, and combining them offers a more complete picture of how a drug might help or harm the human heart. Although challenges remain—such as fully maturing these mini hearts, supplying them with realistic blood flow, and standardizing methods across labs—these advances are laying the groundwork for safer drugs, more personalized treatments, and eventually lab-grown heart tissues that could help repair damaged hearts.
Citation: Kim, Y.H., Son, Y.H., Choi, Y. et al. Advanced in vitro cardiac models for drug evaluation: integration of organoids, engineered tissues, and microphysiological systems. Microsyst Nanoeng 12, 162 (2026). https://doi.org/10.1038/s41378-026-01249-6
Keywords: heart-on-a-chip, cardiac organoids, engineered heart tissue, drug cardiotoxicity, hiPSC cardiomyocytes