Clear Sky Science · en
Functional drug screening of tumor organoids on an active-matrix digital microfluidic chip for cancer precision medicine
Bringing Cancer Treatment to the Lab-on-a-Chip
Cancer drugs do not work the same way in every patient, even when tumors look similar under the microscope. This study describes a tiny “lab-on-a-chip” that can quickly test how a person’s own mini-tumors respond to medicines, using only very small amounts of tissue. The goal is to help doctors choose the right drug, at the right dose, for each individual, instead of relying on trial and error.
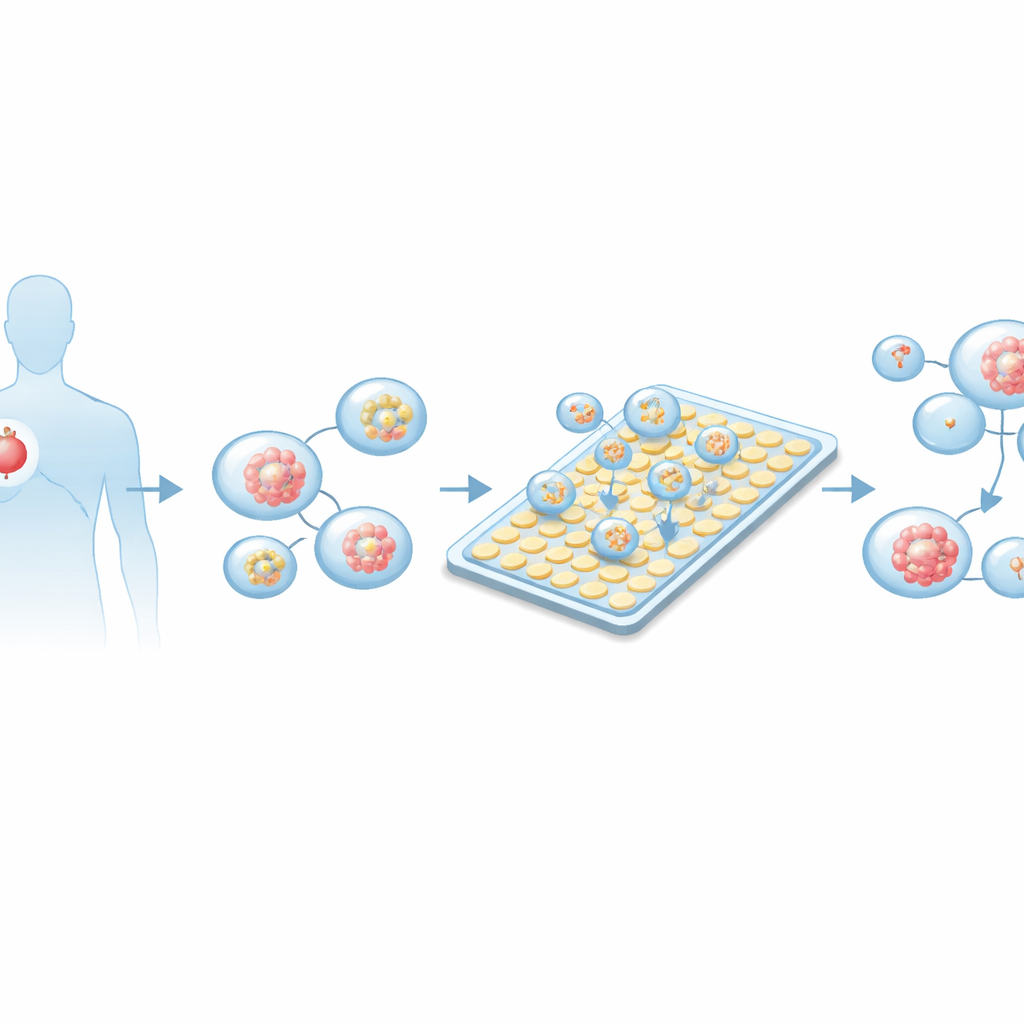
Why Tiny Tumors Can Guide Big Decisions
Traditional lab tests for cancer drugs usually rely on flat layers of cells grown in plastic plates or on animal models. These approaches are slow, require many cells, and often fail to mimic how a real human tumor behaves. In recent years, three-dimensional “organoids” – tiny, spherical clumps of cells grown from a patient’s tumor – have emerged as more faithful stand-ins. They preserve many features of the original cancer, including its genetic diversity and structure, and they often mirror how the patient actually responds to treatment. However, organoids are precious: biopsy samples yield few cells, making it difficult to run many drug tests with conventional, large-volume methods.
A Smart Chip That Moves Droplets Like Pixels
The researchers built on a technology called active-matrix digital microfluidics. Instead of pushing liquids through narrow channels, this system moves tiny droplets across a flat grid of electrodes, a bit like shifting pixels on a screen. By turning electrodes on and off, the chip can generate, split, move, and mix droplets with volumes down to a few nanoliters – thousands of times smaller than a typical lab test. The active-matrix design uses thin-film transistors beneath each electrode, so a large number of “pixels” can be controlled with relatively few wires. The complete platform adds precise temperature control, timing electronics, and integrated imaging, allowing the chip to act as an automated, self-contained experiment station.
Growing Patient Tumors and Loading the Chip
To test the platform, the team grew organoids from lung adenocarcinoma samples taken from multiple patients. Tumor tissue was gently broken down into single cells, embedded in a gel that supports three-dimensional growth, and cultured until small spherical structures formed and matured. Microscopy and fluorescent staining confirmed that these organoids expressed markers consistent with lung tumors and lacked markers of other cancer types. In parallel, the researchers optimized how many tumor spheroids should be suspended in liquid for chip loading, so that most droplets generated on the device would contain one to a few organoids of an appropriate size – large enough to be mature, but not so big that the centers became unhealthy.
High-Speed, High-Precision Drug Testing
Once organoid-containing droplets were on the chip, the system automatically split and rearranged them into regular arrays. Additional droplets containing culture medium and a common chemotherapy drug, cisplatin, were generated and combined in a stepwise fashion to form a series of precisely controlled concentrations. The organoid droplets were then merged with these drug droplets and kept on the chip for up to three days at body-like temperature. An integrated microscope captured the same organoids over time, revealing early shrinkage at higher drug doses as soon as 12 hours, and more severe breakdown of structure after 72 hours. Live/dead fluorescent stains showed that stronger cisplatin exposure led to more damaged cells, seen as an increase in red signal relative to green.
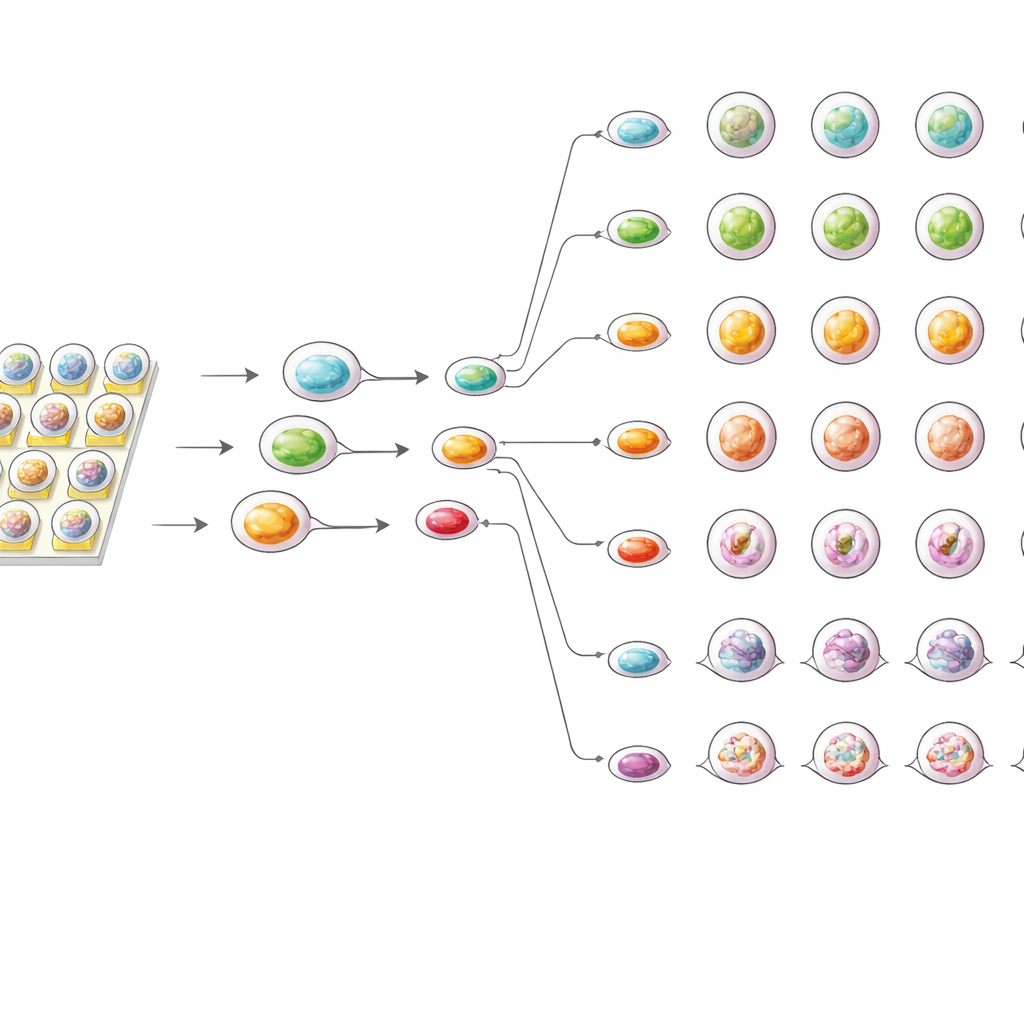
Sharper Sensitivity with Less Sample
To see how the chip compared with standard methods, the team also tested the same organoids in conventional 96-well plates. In both setups, drug effects increased with dose, and each patient’s organoids followed a distinctive response pattern. However, the on-chip tests consistently showed slightly stronger inhibition of organoid growth and lower effective dose values than the plate-based assays. The authors suggest that the tiny droplet environment allows drug molecules to reach organoids more evenly and quickly, and prevents buildup of survival signals that can occur in larger wells. At the same time, the chip needed far fewer cells and reagents and produced highly reproducible results across repeated experiments.
From Prototype to Personalized Cancer Care
In everyday terms, this work shows that a palm-sized device can grow miniature versions of a patient’s tumor and expose them to many doses of a cancer drug, all while using only a sliver of tissue. By watching how these mini-tumors change shape and stay alive or die over time, the platform can provide a detailed, functional readout of drug sensitivity that complements genetic tests. Although further validation with more drugs and longer culture times is needed, this active-matrix microfluidic chip offers a promising route toward faster, more accurate, and more patient-specific decisions in cancer treatment.
Citation: Sun, R., Feng, Z., Wu, T. et al. Functional drug screening of tumor organoids on an active-matrix digital microfluidic chip for cancer precision medicine. Microsyst Nanoeng 12, 135 (2026). https://doi.org/10.1038/s41378-026-01215-2
Keywords: cancer precision medicine, tumor organoids, microfluidic chip, drug sensitivity testing, lung adenocarcinoma