Clear Sky Science · en
Metabolic oxidoreductases: central regulators of the epigenetic landscapes in stemness
How Cell Fuel Shapes Cell Identity
Every cell in your body carries essentially the same DNA, yet neurons, blood cells, and skin cells look and act completely differently. This article explores a surprising link between how cells process fuel and how they decide what type of cell to become. It explains how a class of enzymes called oxidoreductases, which normally handle energy and nutrients, also help write and erase the chemical marks on DNA and proteins that control which genes are turned on. Understanding this connection could reshape how we think about stem cells, aging, and even cancer.
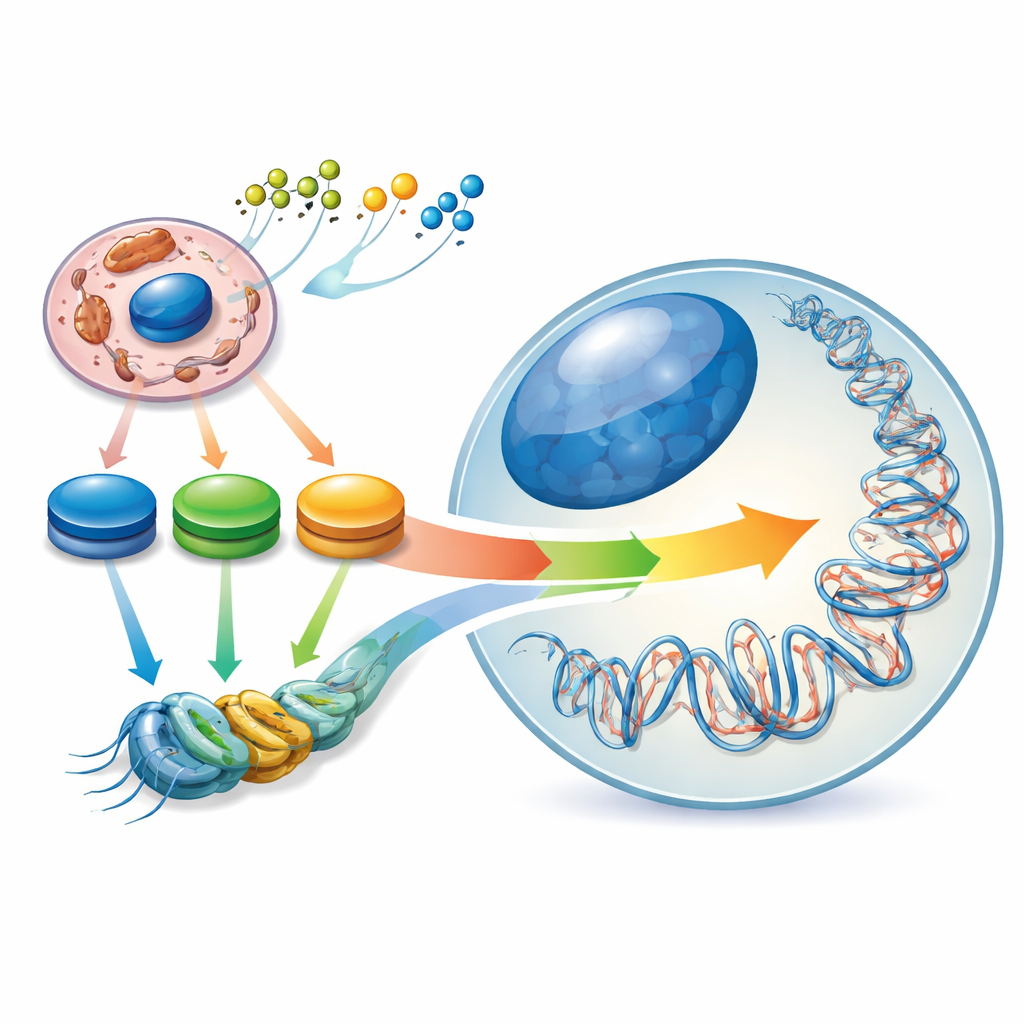
The Hidden Landscape Inside Our Genes
Stem cells are special because they can either renew themselves or mature into many different cell types. They manage this balancing act through an internal “epigenetic landscape” – patterns of chemical tags on DNA and its packaging proteins that act like dimmer switches for genes. The authors describe this landscape as being molded by an “energy contour” inside the cell: peaks and valleys in the levels of key metabolic molecules such as acetyl-CoA, NAD⁺/NADH, and α-ketoglutarate. When a stem cell shifts from a naive state to a more committed one, its central carbon metabolism is rewired, and the epigenetic landscape shifts with it. Thus, changes in cellular fuel use are not just consequences of cell identity – they actively help determine it.
Enzymes That Turn Food Into Epigenetic Signals
Oxidoreductases are enzymes that catalyze oxidation–reduction reactions and sit at key junctions of metabolism. Many of them generate “epigenetic metabolites,” molecules that serve as raw material or cofactors for enzymes that tag DNA and histones. For example, dehydrogenases in glycolysis and the TCA cycle produce acetyl-CoA for histone acetylation, NAD⁺/NADH and NADPH that influence deacetylases, and α-ketoglutarate that powers DNA and histone demethylases. By controlling how much of these metabolites are available, oxidoreductases set the range of possible epigenetic changes. Different stem cell states rely on different metabolic mixes: naive pluripotent stem cells tap both sugar and fat, primed pluripotent cells lean heavily on glycolysis and glutamine, and many adult stem cells favor fatty acid breakdown and oxidative phosphorylation.
Direct Editors of DNA, RNA, and Histones
Some oxidoreductases go beyond supplying ingredients and directly rewrite epigenetic marks. Iron- and α-ketoglutarate–dependent enzymes such as TET proteins and JMJD demethylases remove methyl groups from DNA and histones, reshaping which genes are active. Related enzymes, ALKBH5 and FTO, erase methyl marks on RNA, influencing how long messages last and how efficiently they are translated. Another group, LSD1 and LSD2, use a flavin cofactor to demethylate specific histone lysines. Because all of these enzymes require metabolic cofactors and oxygen, their activity is exquisitely sensitive to the cell’s energy and redox state. The review shows how shifts in α-ketoglutarate, succinate, or mutant metabolites in cancer can tip the balance between open, flexible chromatin and tightly locked, repressed states.
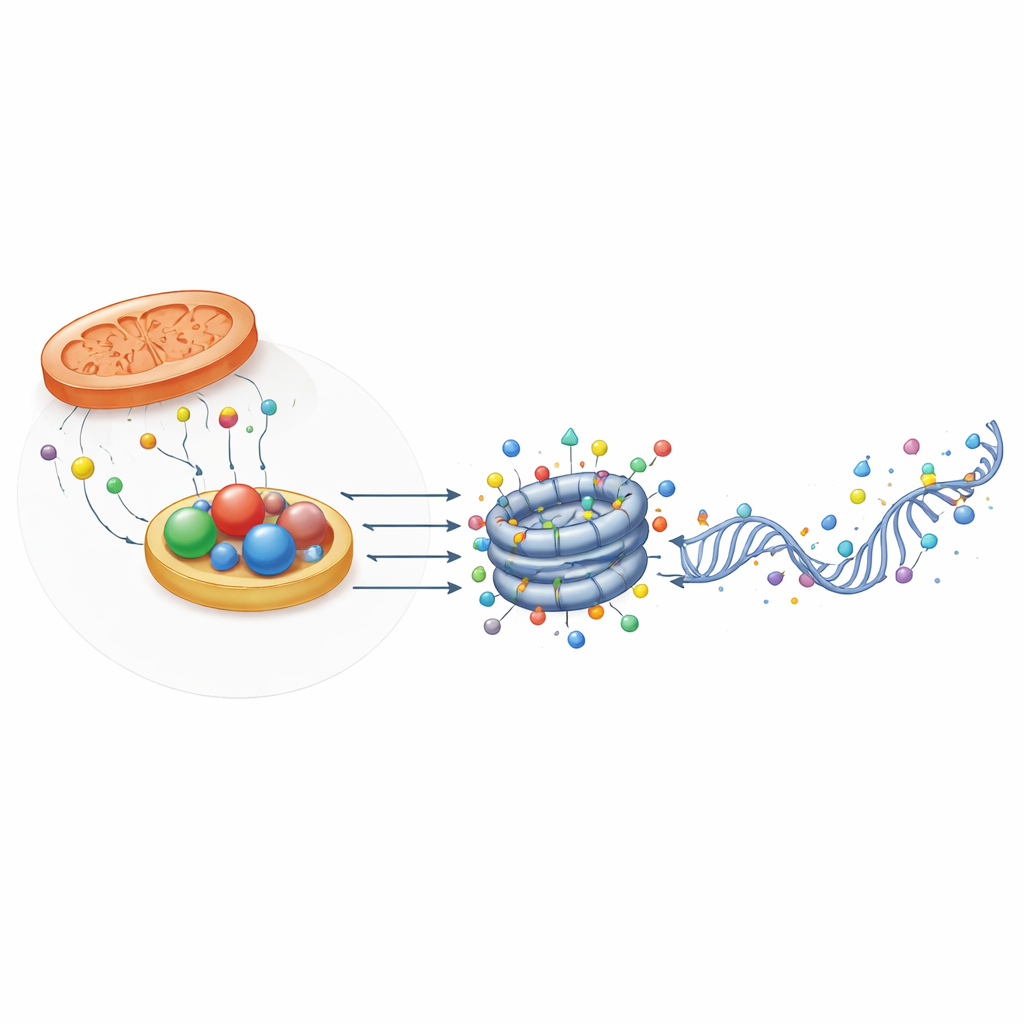
Structural Helpers That Sense Redox State
Intriguingly, not all oxidoreductase-related proteins act as enzymes. Some, such as CTBP and NPAC/GLYR1, retain the structural fold that binds NADH but have little or no catalytic activity. Instead, they form multi-unit scaffolds that bring together transcription factors and chromatin modifiers at specific gene regions. Their assembly depends on NAD(H) binding, meaning they can act as physical sensors of the cell’s redox state. In mouse embryonic stem cells, CTBP2 helps poise pluripotency genes for shutdown during differentiation by recruiting complexes that remove acetyl and methyl marks, while NPAC/GLYR1 teams up with LSD2 and RNA polymerase II to coordinate transcriptional elongation and alternative splicing. These non-enzymatic roles add another layer by which metabolism influences gene control.
Why This Matters for Health and Disease
The authors conclude that oxidoreductases knit together metabolism and gene regulation at three levels: they generate key metabolites, directly erase chemical marks on DNA, RNA, and histones, and act as scaffolds that reorganize chromatin machinery. Together, these functions turn a cell’s fuel usage into durable instructions about which genes to express, helping to maintain stemness or push differentiation. This framework helps explain why metabolic mutations can reprogram cells toward cancer-like states, and why adjusting nutrients or metabolic enzymes can alter stem cell behavior. As researchers probe these links in more detail, oxidoreductases are emerging as attractive targets for regenerative therapies and anticancer strategies that aim to remodel the epigenetic landscape by rewiring how cells handle energy.
Citation: Lee, HT., Roe, JS. & Youn, HD. Metabolic oxidoreductases: central regulators of the epigenetic landscapes in stemness. Exp Mol Med 58, 1017–1037 (2026). https://doi.org/10.1038/s12276-026-01687-2
Keywords: stem cell metabolism, epigenetic regulation, oxidoreductase enzymes, chromatin remodeling, cancer stem cells